Clear Sky Science · en
Glycosylation as a dynamic regulator of RLR and cGAS-STING innate immune signalling pathways
How Sugar Tags Help Cells Sense Viruses
Our cells are constantly on watch for invading viruses, and much of this defense relies on tiny sugar tags attached to proteins. This review explains how these sugar tags, known as glycosylation, act like dimmer switches rather than simple on/off buttons for key antiviral pathways. Understanding this sugar-based fine-tuning could open new ways to boost vaccines, treat viral infections, and even harness the immune system against cancer.
The Cell’s Early Warning Alarm
When viruses enter a cell, they leave behind telltale strands of RNA or DNA. Specialized “alarm” systems patrol the cell’s interior for this genetic debris. One system, called the RIG-I-like receptor (RLR) pathway, detects viral RNA. Another, known as the cGAS–STING pathway, senses DNA that appears in the wrong place inside the cell. Once triggered, both pathways switch on chemical cascades that end in the release of type I interferons and inflammatory molecules—powerful signals that warn neighboring cells and rally immune defenses. Because too much or too little of this response can be dangerous, the cell must carefully tune these alarms, and glycosylation is one of the key ways it does so. 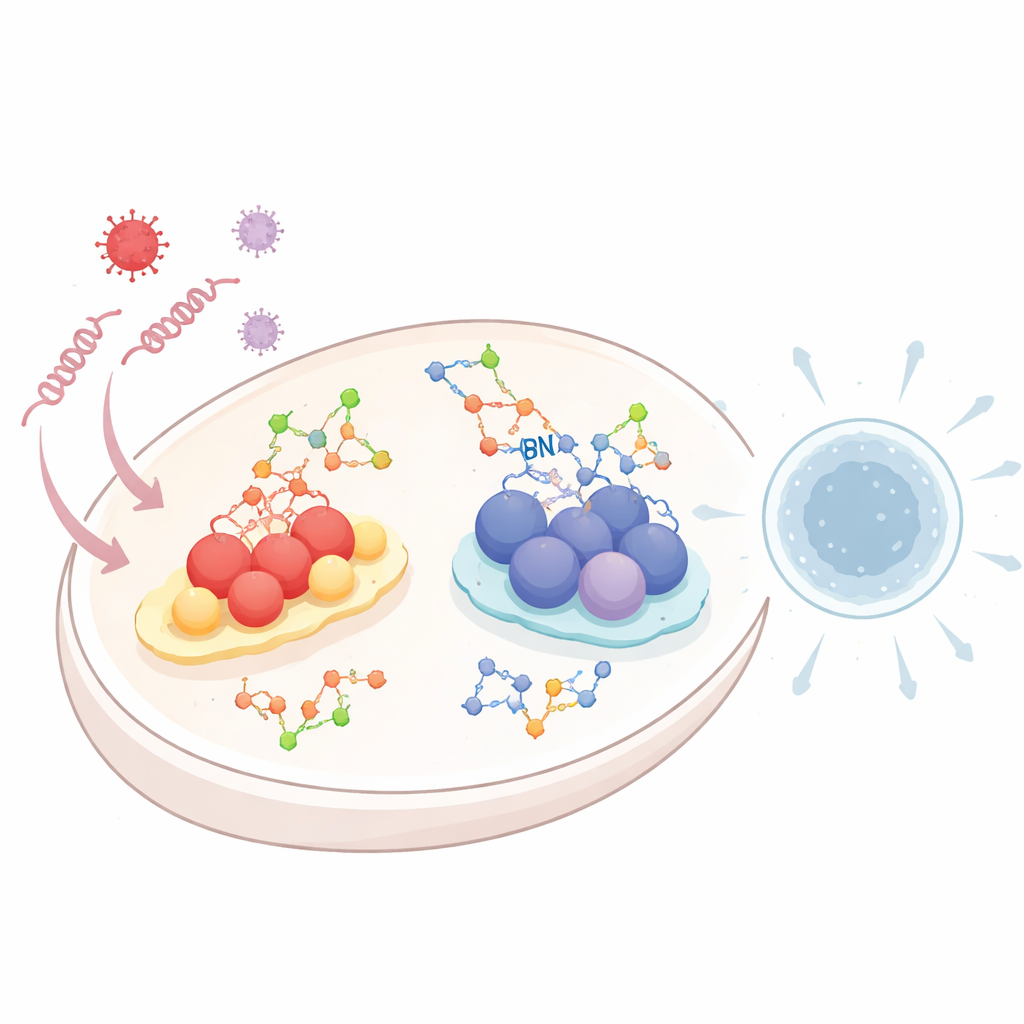
Sugar Coats as Precision Controls
Glycosylation adds small sugar units to proteins in different cellular locations. In the cell’s internal shipping network, the endoplasmic reticulum and Golgi, bulky N-linked sugar chains help proteins fold properly, stay stable, and travel to the right membrane. In contrast, a leaner form called O-GlcNAcylation places a single sugar on proteins in the cytoplasm and nucleus. This minimalist tag is highly dynamic, turning over quickly as two enzymes add or remove it in response to nutrient levels and stress. The review describes how these different sugar types do not simply turn immune sensors on or off; instead, they adjust how easily sensors cluster, how long they last, and how strongly they signal. In this way, glycosylation links the cell’s metabolic state directly to its readiness to fight infection.
Tuning the RNA Sensor Pathway
Within the RNA-sensing RLR system, a central relay protein called MAVS sits on mitochondria and acts as a key hub. The review shows that O-GlcNAcylation on MAVS can both accelerate and brake antiviral signaling, depending on where the sugar is attached. Sugar tags at certain sites promote further protein decorations, such as a specific type of ubiquitin chain, that help MAVS form large signaling clusters and drive strong interferon production against RNA viruses. Other sugar sites keep MAVS quietly dispersed and inactive under normal conditions, preventing needless inflammation. Viral infection and changes in the cell’s sugar-producing metabolic pathway can shift the balance between these activating and inhibiting tags. Related modifications on another factor, IRF5, can push the immune system toward damaging “cytokine storms,” highlighting that more sugar is not always better. In parallel, viruses can exploit N-linked sugars on cell-surface proteins such as the growth factor receptor EGFR to mark upstream sensors like RIG-I for degradation, blunting antiviral defenses.
Tuning the DNA Sensor Pathway
The DNA-sensing cGAS–STING pathway is also strongly shaped by glycosylation. STING, a membrane protein in the endoplasmic reticulum, needs N-linked sugar chains to fold correctly, remain stable, and assemble into signaling clusters once activated by DNA-derived messengers. Without these sugars, STING fails to form the higher-order structures and traffic through the cell compartments required for robust interferon release. At the same time, O-GlcNAcylation of STING at a particular site enhances a different set of chemical tags that promote its clustering and movement, amplifying antiviral signals against DNA viruses. Sugary chains called sulfated glycosaminoglycans, built in the Golgi, further assist by helping activated STING polymerize into long structures that recruit downstream enzymes. Viruses counterattack by altering their own glycoproteins or by triggering degradation pathways that target STING, often in the same cellular spaces where these sugar decorations are added or processed. 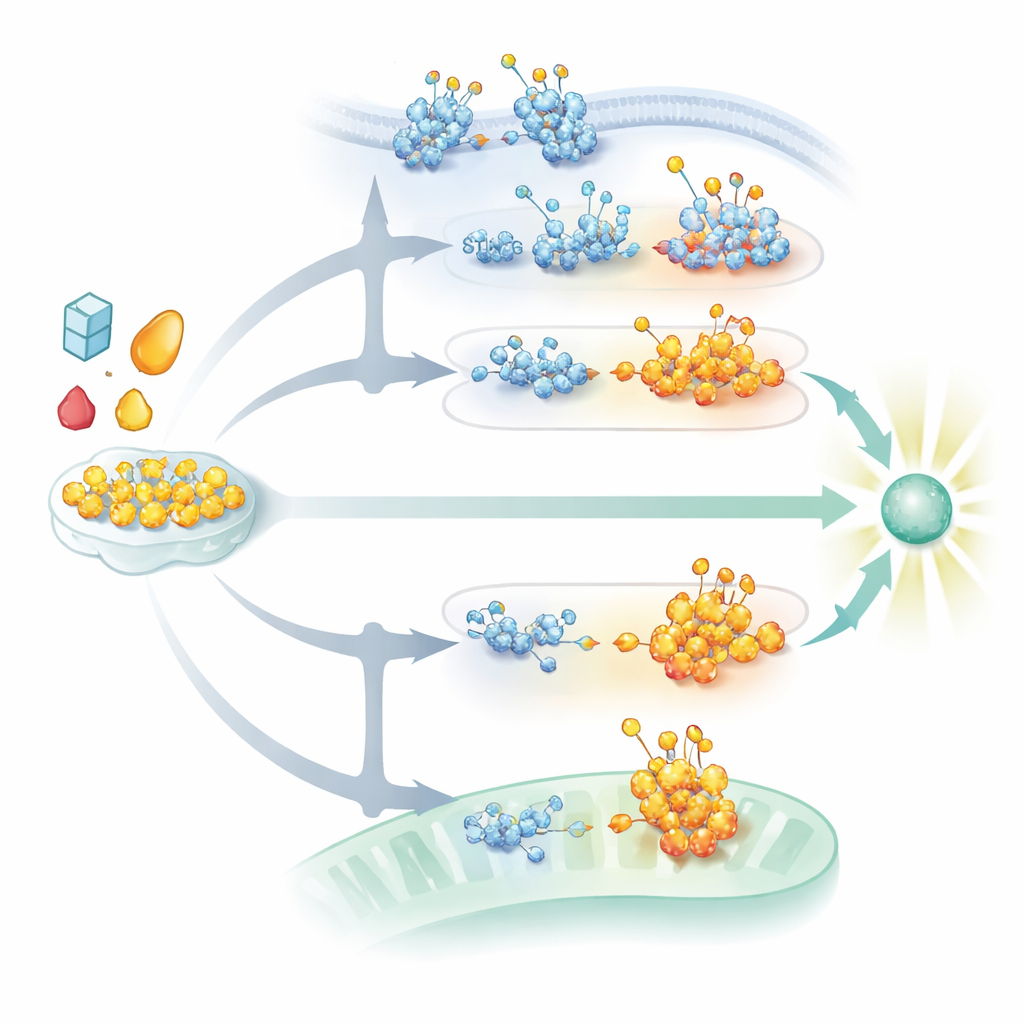
Metabolism, Disease, and Future Therapies
Because the main sugar donor for O-GlcNAcylation is made by a metabolic route known as the hexosamine biosynthetic pathway, shifts in nutrient use directly shape antiviral signaling. Many viruses drive this pathway to their advantage, while experimental boosting or blocking of hexosamine flux can strengthen or weaken antiviral responses in animal models. The review connects these mechanisms to human disease: tumors frequently rewire glycosylation to dampen interferon signaling and escape immune attack, while chronic metabolic stress or inherited defects in glycan processing can tip the balance toward harmful autoimmunity. The authors argue that enzymes controlling glycosylation—especially those that add or remove O-GlcNAc or build key N-linked structures—are promising but challenging drug targets. Future therapies will likely need to act in a site-specific and context-aware fashion, nudging the “glyco-code” toward better protection against infections, cancer, and inflammatory disorders without triggering new forms of immune imbalance.
The Big Picture for Everyday Health
In everyday terms, this article reveals that the body’s frontline antiviral alarms do not operate in isolation—they are wired into the cell’s energy use and sugar chemistry. Tiny sugar tags on a handful of critical proteins decide whether a stray viral genome sparks a measured warning, a full-blown defense, or a dangerous overreaction. By decoding and eventually learning to adjust this sugar-based control system, researchers hope to design treatments that make vaccines more effective, help clear stubborn infections, unmask tumors to the immune system, and calm runaway inflammation in autoimmune disease.
Citation: Tong, J., Zhang, W., Xue, M. et al. Glycosylation as a dynamic regulator of RLR and cGAS-STING innate immune signalling pathways. Commun Biol 9, 422 (2026). https://doi.org/10.1038/s42003-026-09767-9
Keywords: innate immunity, glycosylation, RLR pathway, cGAS-STING, O-GlcNAcylation