Clear Sky Science · en
Structural insights into the Thermus thermophilus type IV pilus machinery assembling two distinct pili
Tiny Bacterial Hairs with Big Jobs
Bacteria may be microscopic, but many are equipped with remarkable surface “hairs” that let them move, cling to surfaces, and even pull in stray DNA from their surroundings. In this study, researchers examine how one heat-loving bacterium, Thermus thermophilus, builds and operates a sophisticated nano-machine that pushes these hairs—called pili—through its cell envelope. Understanding this machinery not only reveals how microbes adapt and evolve but also offers inspiration for future nanotechnology and new ways to disarm harmful bacteria.
Two Different Hairs from One Machine
Thermus thermophilus produces two distinct types of pili: a wider, thicker filament and a narrower, slimmer one. Earlier work showed that these filaments are made of different building blocks and likely serve different purposes, such as movement along surfaces or pulling in DNA. Yet both are constructed by the same multi-part machine that spans from the inner to the outer surface of the cell. The central question of this study is how a single system can assemble two different filaments and push them through the cell’s protective layers to the outside world.
Mapping the Hidden Hardware
To uncover how the machine is put together, the researchers used cryo-electron tomography, a technique that freezes cells rapidly and images them in 3D at very low temperatures. They combined these snapshots with targeted genetic changes that removed or altered specific parts of the machine. By comparing normal cells with mutants lacking certain components, they could see which blurred shapes in their 3D maps corresponded to which proteins. Advanced structure prediction tools were then used like molecular blueprints, helping to fit these protein models into the observed densities and assemble a hypothetical full-length model of the machinery.
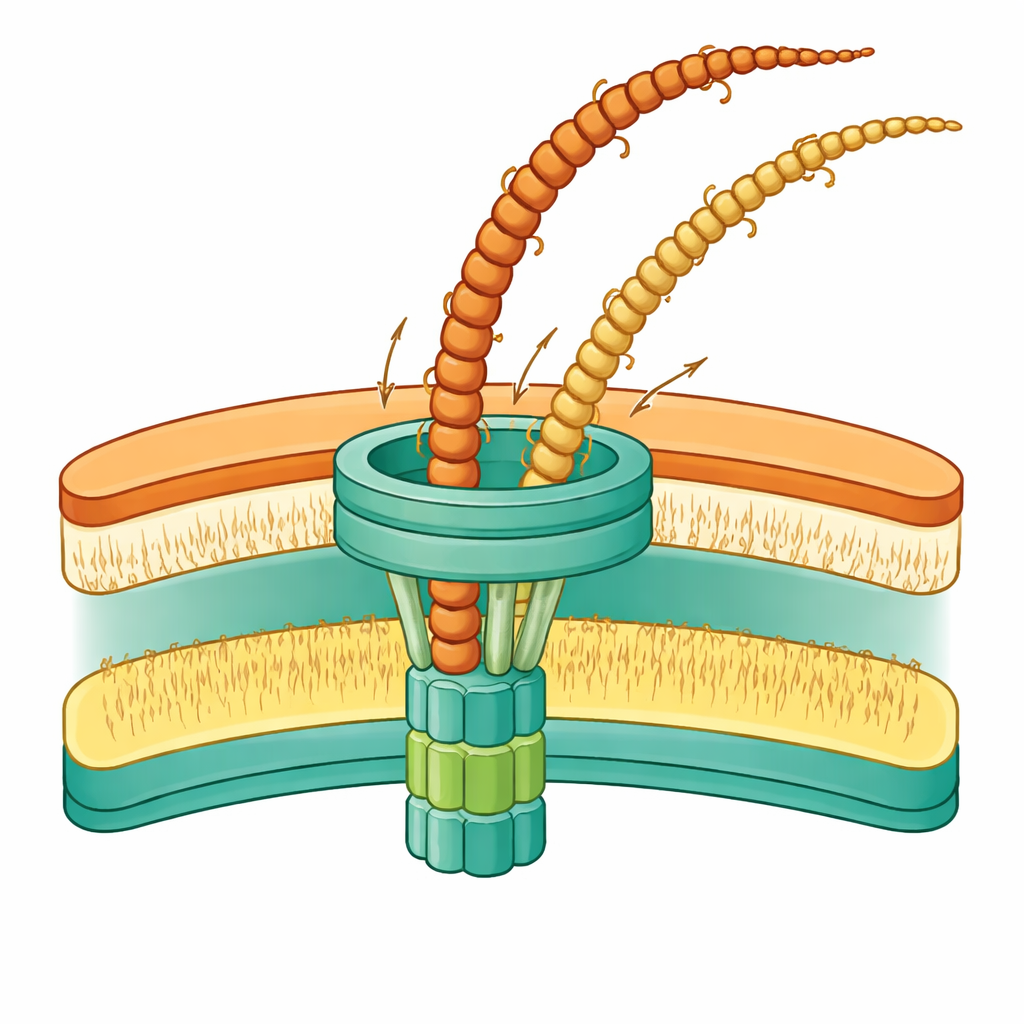
A Flexible Link That Holds It All Together
One standout result concerns a protein called PilW. This component sits between an outer membrane gate and an inner platform anchored in the cell’s inner membrane. When PilW is missing entirely, pili fail to reach the outside and instead pile up in the space between the membranes. When only part of PilW is trimmed, the machine still works, but its inner components shift position and the surrounding membranes bend inward. These observations suggest that PilW behaves like a flexible tether that links the outer gate to the inner platform, adjusting its length and shape as the machine cycles between idle and active states. The team’s models indicate that this flexibility allows the system to cope with the unusually wide gap between the two membranes in Thermus, while still aligning all parts well enough to assemble and eject pili.
Watching the Filaments and Their Sugar Coats
Alongside the in-cell imaging, the researchers also isolated the two pilus types and examined them in greater detail using single-particle cryo-electron microscopy. This higher-resolution approach revealed the precise arrangement of the building blocks and, crucially, allowed the team to model the sugar molecules that decorate the filaments’ surfaces. The wider pilus carries three sugar attachment sites per building block, producing a dense halo of carbohydrates. The narrower pilus has only one such site, but its sugar chain sticks out farther, making the overall filament appear larger than its protein core would suggest. Computer simulations then explored how these sugars bend and sway as the filament passes through the outer membrane gate.
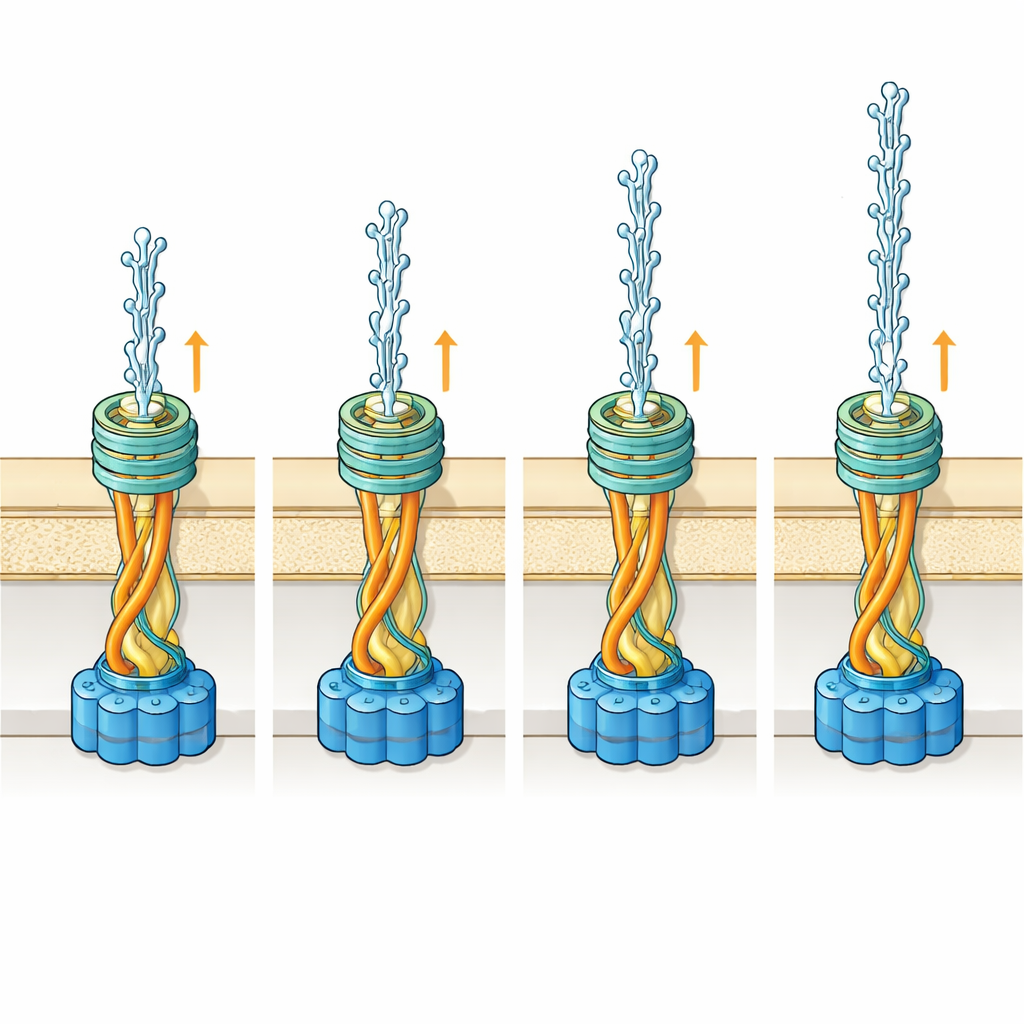
A Shared Gate for Two Very Different Cables
By docking their detailed pilus structures into a model of the outer membrane gate, called PilQ, the team found that both filament types can, in principle, slide through the same opening. For the wider filament, its multiple sugars have enough room to explore many shapes as they move through the gate. For the narrower filament, space becomes tight at one point in the channel, so some sugar conformations would clash with the walls. Simulations suggest that in this case the sugar chain likely hugs the filament more closely while inside the gate and fans out only once outside the cell. Rather than evolving a larger, more expensive gate to fit both filaments comfortably, the bacterium seems to rely on the natural flexibility of these sugar chains to make the system work efficiently.
What This Means for Microbial Life
Taken together, the study paints a coherent picture of how a single, adaptable machine builds and exports two very different pili in a hot-spring bacterium. A flexible linker protein appears to keep the inner and outer parts aligned as the motor cycles and the structure shortens slightly during active filament growth. At the same time, the sugar coatings on the pili give them both protection and reach, while remaining flexible enough to squeeze through a relatively narrow exit channel. For non-specialists, the key message is that even simple microbes rely on highly coordinated, moving assemblies to survive and evolve—molecular devices whose elegance and efficiency rival, and sometimes surpass, human-made machines at the nanoscale.
Citation: Neuhaus, A., McLaren, M., Isupov, M.N. et al. Structural insights into the Thermus thermophilus type IV pilus machinery assembling two distinct pili. Commun Biol 9, 474 (2026). https://doi.org/10.1038/s42003-026-09762-0
Keywords: type IV pili, bacterial nanomachines, cryo-electron microscopy, protein glycosylation, Thermus thermophilus