Clear Sky Science · en
Growth rate-driven modelling suggests that phenotypic adaptation drives drug resistance in BRAFV600E-mutant melanoma
Cancer Cells That Learn to Live With Drugs
Targeted drugs have transformed treatment for many people with melanoma, a serious skin cancer. Yet these drugs almost always stop working: tumors that once shrank begin to grow again. This study asks a simple but powerful question: instead of relying only on genetic mutations, can melanoma cells "learn" to survive treatment by changing how they behave—and could this learning explain why some ways of giving drugs work better than others?
From Sensitive Cells to Survivors
The researchers focused on melanoma cells carrying a common change in a gene called BRAF, which makes them especially sensitive to a type of drug known as a BRAF inhibitor. In lab experiments from earlier work, these cells were exposed to the drug encorafenib at different doses and for different lengths of time. By carefully measuring how quickly the cells grew or died under each condition, the authors split the cells into two broad categories: drug-naive cells that had never seen the drug, and drug-adapted cells that had lived in the drug for at least a week. Drug-naive cells grew well without treatment but were strongly slowed or killed by the drug. Drug-adapted cells, in contrast, grew better in the presence of the drug but lost that advantage when the drug was removed—hinting that their survival was linked to a flexible, reversible change in state rather than permanent genetic damage.
A Map of Hidden Cell Behaviors
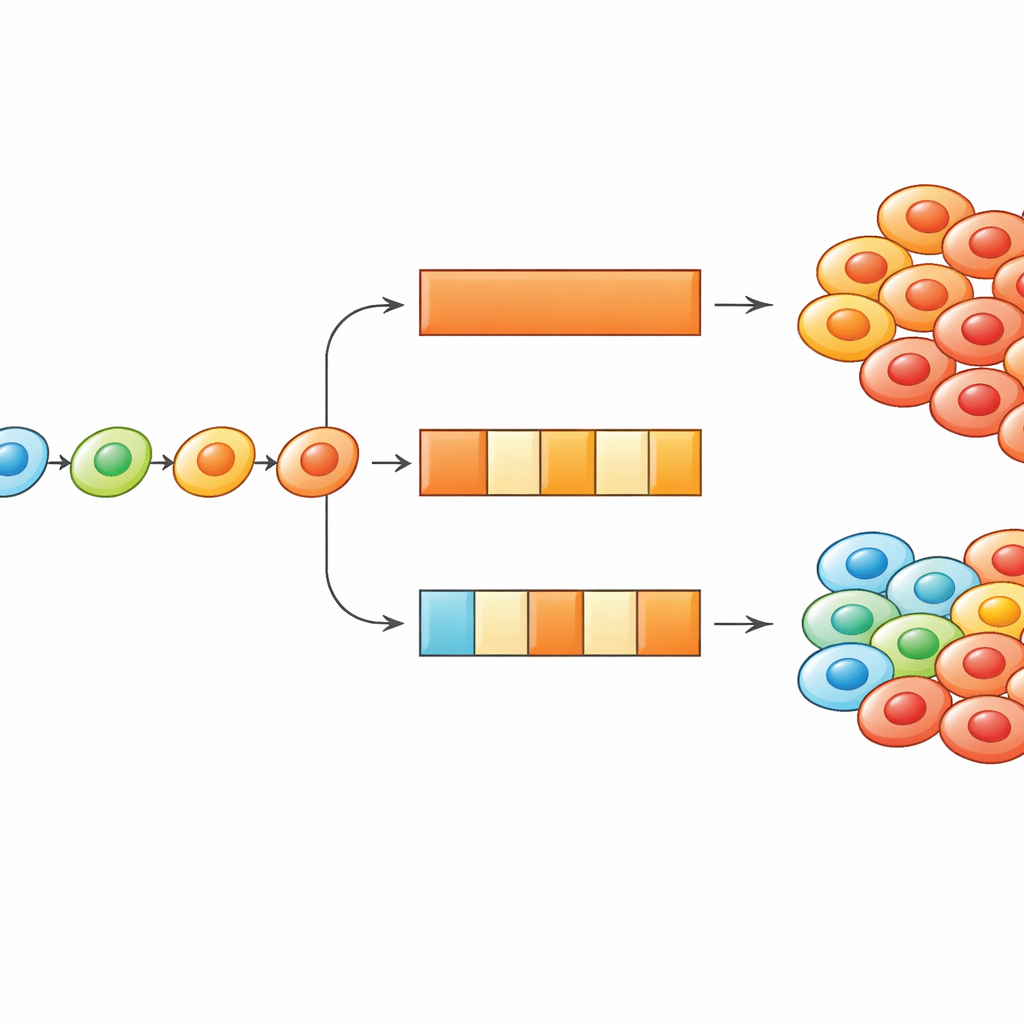
To make sense of these changes, the team built a mathematical model in which each cancer cell is assigned a "phenotype state"—an abstract position on a scale from very drug-sensitive to very drug-resistant. Instead of assuming only two types of cells, they allowed many intermediate states, reflecting gradual shifts in behavior observed in biological markers of resistance. For every combination of state and drug dose, they estimated a net growth rate, which could be positive (more cell divisions than deaths) or negative (more deaths than divisions). All of these rates were arranged into a grid, or "fitness matrix," that acts like a landscape: some states at some doses are good places for cells to thrive, others are deadly. Cells in the model can move step by step across this landscape as treatment is turned on or off.
Do Cells Drift or Climb Toward Advantage?
The key unknown was how cells move across this landscape. The authors tested four possible strategies. In one, cells never change state, so any resistant cells must be present from the beginning. In another, cells wander randomly between neighboring states, like a drunkard’s walk, regardless of whether their situation improves. In the two remaining strategies, cells tend to move toward states where their growth rate is higher—either noisily, with some missteps, or strongly, always climbing toward better survival. When the team ran computer simulations and compared them with real lab data, especially experiments where the drug was given either continuously or in an on–off pattern (one week on, one week off), only the strategies where cells climb toward higher fitness could reproduce what was observed: intermittent treatment at a high dose suppressed cell numbers better than continuous treatment with the same total time and a higher overall amount of drug.
Using On–Off Dosing to Turn Adaptation Against Cancer
Diving deeper, the researchers used both simulations and simpler equations to explore how often cells are allowed to change state and how long drug-on and drug-off periods last. They found that when cells adapt in a directed way, treatment schedules can be tuned to either slow or exploit this adaptation. Shortening how frequently cells can update their state—conceptually, slowing the biological mechanisms that enable phenotypic change—tends to make intermittent treatments more powerful, because cells cannot so quickly climb to highly resistant states. Conversely, if cells begin in a strongly resistant state, drug holidays can give them time to slide back toward more sensitive states, so that reintroducing the drug triggers a surge in cell death. The study shows how the timing of dosing and the speed of cellular adaptation together shape which schedule works best.
What This Means for Future Cancer Care
To a layperson, the central message is that cancer cells are not just fixed enemies; they are shape-shifters. In this melanoma system, the cells appear to actively move toward behaviors that help them grow under whatever conditions they face. The authors’ model suggests that this directed adaptation alone—without invoking different permanent clones—can explain why on–off drug schedules sometimes beat constant dosing, even when they use less drug overall. While these results come from lab-grown cells and need more work before guiding patient care, the approach offers a practical framework: by measuring how fast and in which direction cancer cells adapt, doctors may eventually design treatment schedules that not only attack tumors but also steer their phenotypic "learning" into dead ends rather than escape routes.
Citation: Hamis, S., Browning, A.P., Jenner, A.L. et al. Growth rate-driven modelling suggests that phenotypic adaptation drives drug resistance in BRAFV600E-mutant melanoma. Commun Biol 9, 385 (2026). https://doi.org/10.1038/s42003-026-09760-2
Keywords: melanoma, drug resistance, cell plasticity, intermittent therapy, mathematical modeling