Clear Sky Science · en
Generative models of cell dynamics: from Neural ODEs to flow matching
Watching Cells Change Over Time
Every organ in our bodies is built from cells that once started out looking almost identical, then gradually took on different roles. Biologists would love to watch each of these cells change in real time, but current measurement methods destroy the cells they read. This review article explores how a new kind of artificial intelligence tool, called Neural Ordinary Differential Equations, can help reconstruct these hidden stories of change from static snapshots—potentially improving our understanding of development, disease, and how treatments work.

From Snapshots to Living Movies
Modern single-cell sequencing technologies can measure the activity of thousands of genes in individual cells, producing huge tables of numbers. Each row corresponds to a cell, each column to a gene. But each cell is measured only once, so scientists end up with a collection of still photos rather than a movie of how any one cell changes over time. Early approaches tried to put these snapshots into a plausible order using “pseudotime,” arranging cells along developmental paths based on how similar their gene activity patterns looked. This helped reveal broad routes of cell differentiation but struggled when cells branched into multiple fates or followed complex, looping paths.
Describing Cell Change as Motion in a Landscape
To move beyond simple orderings, researchers describe cells as points moving through a landscape: position encodes the current gene activity state, and “momentum” reflects how that state is changing. Classic tools such as RNA velocity estimate where a cell is headed next by comparing different forms of RNA inside it. Others reconstruct vector fields—maps of arrows showing, at each point in this space, the likely direction of motion. These methods began to reveal how gene networks drive cells toward specific fates, and how unstable regions in the landscape may signal upcoming decisions. However, many of these models make strong simplifications about how genes interact and often cannot generate fully continuous paths that match population data at multiple time points.
Neural ODEs: Learning the Rules of Change
Neural ODEs offer a flexible way to learn the underlying rules that govern cellular motion in this landscape. Instead of prescribing formulas in advance, a neural network learns a function that tells you, for any cell state, how that state will change next. An ODE solver then traces full trajectories through time. This framework has been adapted to several single-cell tasks: inferring developmental paths, highlighting branching events, identifying unstable or “primed” states, and even recovering gene regulatory networks—the cause-and-effect relationships between genes that turn each other on or off. By encouraging the learned rules to be simple and sparse, some methods can propose compact regulatory diagrams that are easier to interpret and test experimentally.
Generative Flows: Matching Populations Across Time
Another strand of work treats the problem at the population level rather than tracking individual cells. Here, the goal is to learn how the whole distribution of cell states at one time transforms into the distribution at a later time. Techniques from optimal transport, which were originally developed to find the cheapest way to move mass from one shape to another, have been combined with Neural ODEs and related “flow” models. Continuous normalizing flows and the newer, more efficient “flow matching” methods directly learn vector fields that move one cell population into another, without rerunning expensive simulations during training. These approaches can handle irregular sampling, noisy measurements, and even stochastic (partly random) dynamics, offering generative models that can simulate realistic future populations or reconstruct likely past states.
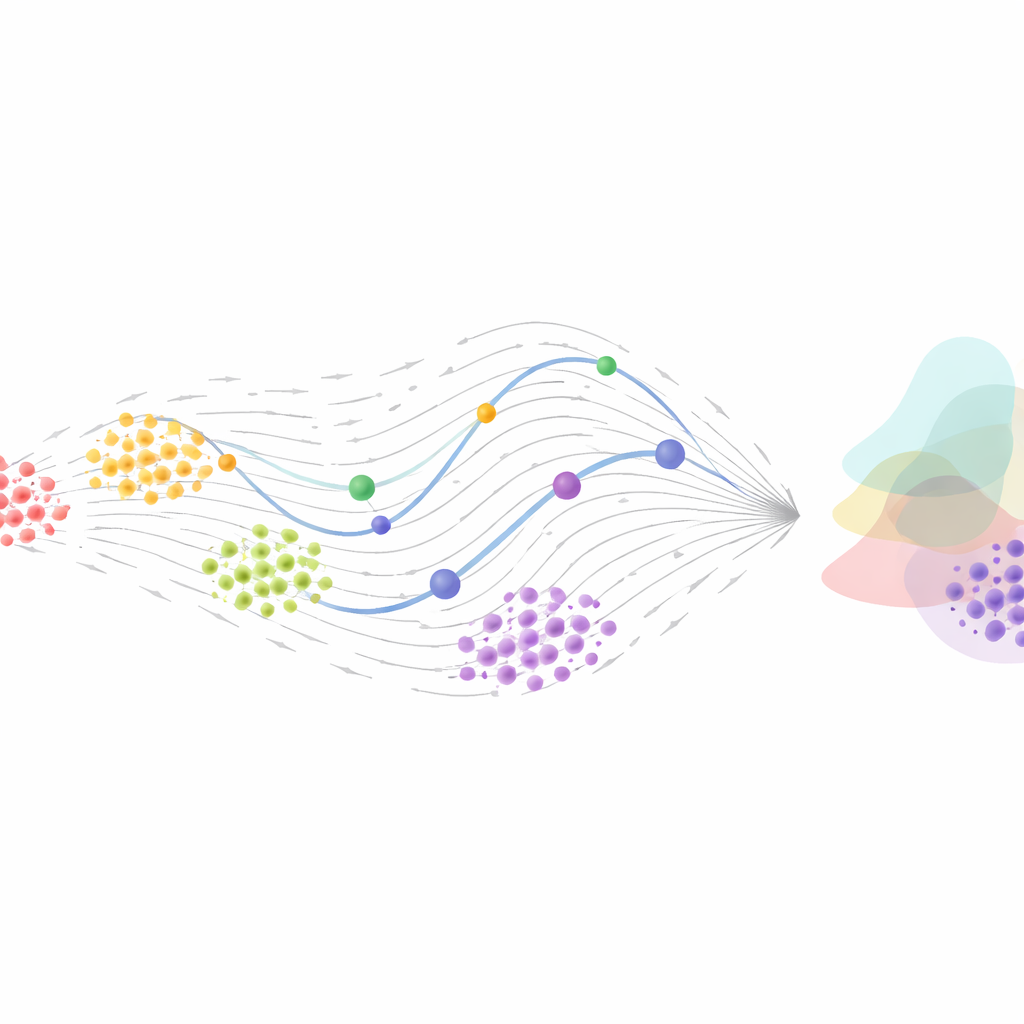
Opportunities and Open Questions
Despite their promise, these models face important challenges. Distinguishing genuine cause-and-effect from mere correlation in gene networks is difficult, especially when some influential factors are unobserved. Researchers are also pushing Neural ODEs to go beyond fitting the data they see toward truly learning general laws that can predict responses to new conditions, such as gene knockouts or drug treatments. Handling noise, randomness, and many overlapping data types—like combining gene activity with spatial location or other molecular layers—remains an active area of research. Still, the same ideas are already being applied beyond genomics, for example to heart dynamics, drug behavior in the body, and medical time series data.
Why This Matters for Health and Disease
For a non-specialist, the key message is that Neural ODEs and flow-matching models let scientists turn static cell measurements into dynamic stories. Instead of just knowing what cells look like at a few isolated stages, these tools help infer how cells are likely to move through their developmental landscape, which switches in their gene circuits drive those moves, and how interventions might redirect them. In the long run, such models could enable in-silico experiments that predict how a stem cell might be nudged into a desired type, how a tumor’s cell population will evolve under treatment, or how complex therapies affect entire cellular ecosystems—bringing us closer to a mechanistic, predictive understanding of life at the single-cell level.
Citation: Richter, T., Wang, W., Palma, A. et al. Generative models of cell dynamics: from Neural ODEs to flow matching. Commun Biol 9, 352 (2026). https://doi.org/10.1038/s42003-026-09758-w
Keywords: single-cell dynamics, neural ODEs, cell differentiation, generative models, optimal transport