Clear Sky Science · en
Surface decoration of bacteria generates robust living therapeutics for improved ulcerative colitis therapy
Turning Friendly Germs into Tiny Bodyguards
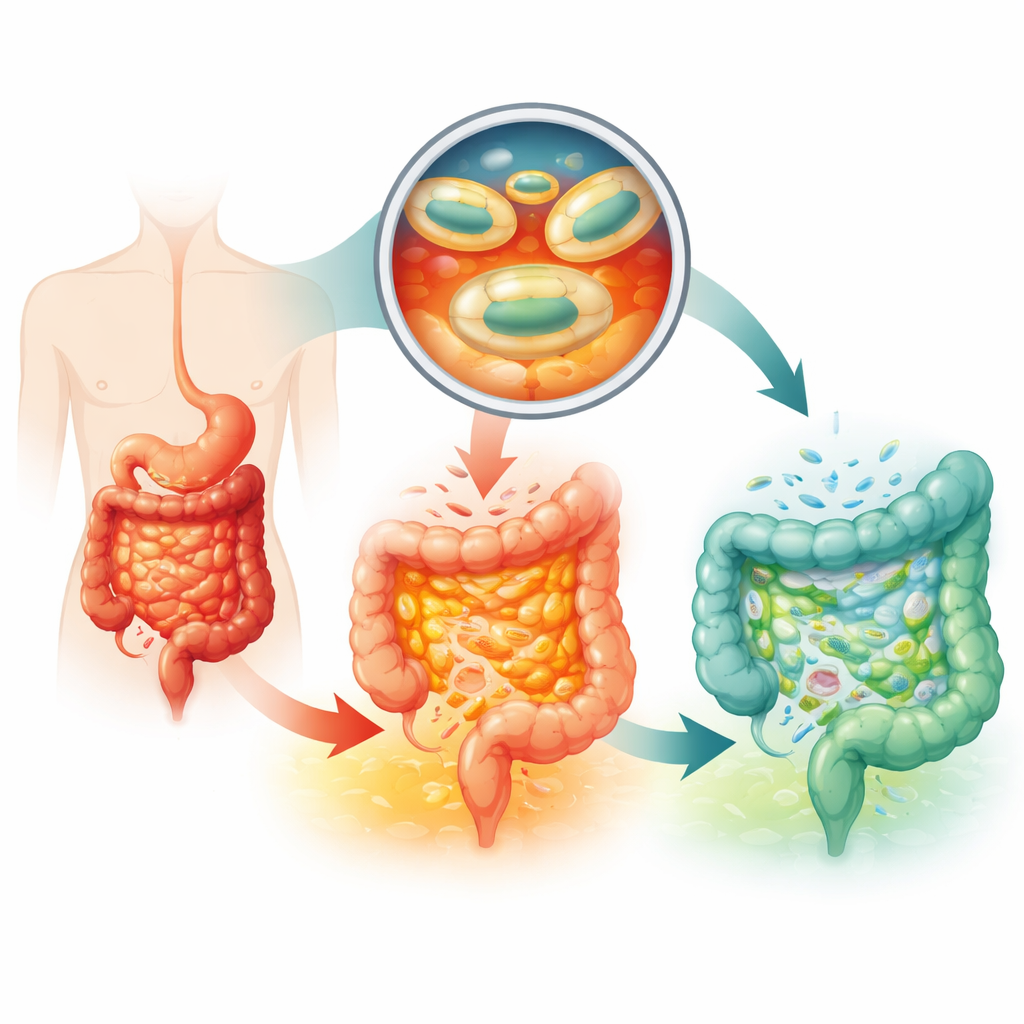
Ulcerative colitis is a painful bowel disease in which the body’s own gut becomes chronically inflamed, leading to urgent diarrhea, bleeding, and a greatly reduced quality of life. Doctors and patients are excited about “good bacteria” pills that might calm this inflammation from within, but most of these microbes die on the harsh journey through the stomach and small intestine before they ever reach the colon, where they are needed. This study describes a new way to dress probiotic bacteria in a protective armor so they can safely pass through the digestive tract, settle in the colon, and help repair the damaged gut.
Why Current Probiotic Pills Often Fall Short
Standard treatments for ulcerative colitis rely on anti-inflammatory and immune-suppressing drugs that can have serious side effects and do not work for everyone. Probiotics offer a gentler approach by helping to rebalance the gut’s microbial community. A special strain of Escherichia coli called Nissle 1917 is already approved as a medicine for this disease, but patients must swallow extremely large doses every day, and even then the benefits are modest. The key problem is survival and delivery: stomach acid, digestive enzymes, and rapid transit through the upper intestines kill or wash away most of the bacteria before they can attach and grow in the colon, where more than 99% of gut microbes normally reside.
Building a Protective Shell Around Helpful Bacteria
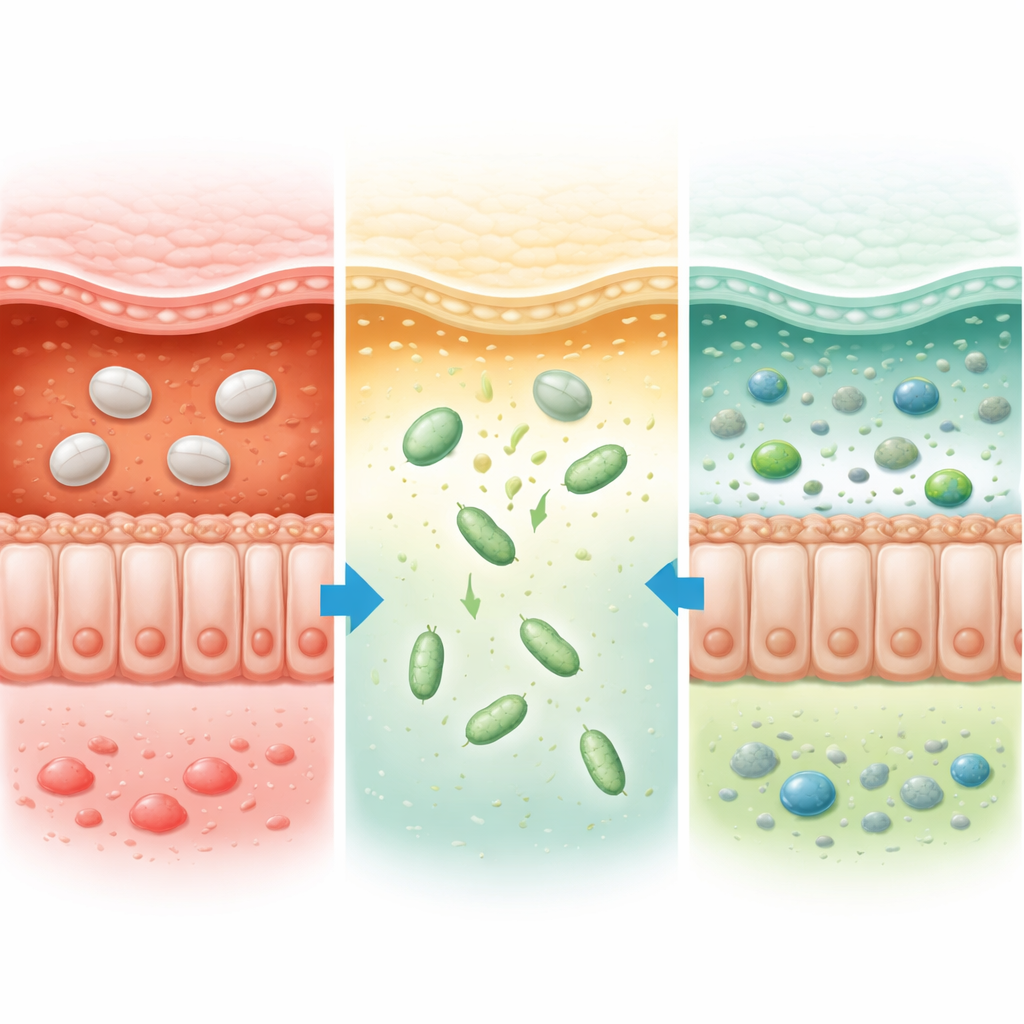
To solve this, the researchers created a thin, smart coating around each probiotic cell using a plant-derived material called lignin. They chemically modified lignin to make it more soluble and sensitive to changes in acidity, producing a substance that stays stable in strong acid but falls apart in the more neutral environment of the colon. Using a gentle layer-by-layer process, they first added calcium ions to the probiotic surface and then wrapped it with the modified lignin, forming a smooth “armor” about one ten-thousandth of a millimeter thick. Microscopy and particle-size measurements showed that this shielded the bacteria without killing them, and the same method worked on several very different bacterial species, suggesting it could serve as a general platform for many probiotic therapies.
Targeting the Colon and Helping It Heal
When tested in simulated digestive fluids, unprotected bacteria were quickly destroyed by stomach acid, while armored bacteria stayed alive for hours and only released their cargo efficiently at colon-like pH levels. In mice with chemically induced ulcerative colitis, the coated bacteria accumulated strongly in the colon, whereas uncoated bacteria or simple mixtures with the coating material largely failed to do so. The team used fluorescent imaging, gene tests, and mathematical modeling to show that the protected bacteria not only arrived in the colon intact but also multiplied there, reaching about one in a thousand of all gut microbes. Treated mice lost less weight, kept longer colons, and had much healthier gut tissue under the microscope, with tighter cell junctions and a restored mucus layer that together form a crucial barrier between the body and the gut contents.
Calming the Immune System and Rebalancing the Microbiome
Beyond physical repair, the armored probiotics reshaped the immune and microbial landscape in the diseased gut. In the blood and colon of sick mice, levels of inflammatory molecules were high and levels of a key anti-inflammatory signal were low; the new treatment reversed these trends, shifting the immune system toward a calmer state. Detailed cell analyses showed changes in the mix of immune cells in both colon and spleen. At the same time, DNA sequencing of gut microbes revealed that the therapy increased overall microbial diversity and boosted the presence of several groups of bacteria linked to gut health, including Akkermansia and Muribaculum. Metabolite profiling further showed broad changes in small molecules tied to vitamin, fatty acid, and amino acid metabolism, suggesting that the rebalanced microbiome was producing a chemical environment more compatible with healing.
From Mouse Study to Future Living Medicines
Altogether, the work demonstrates that giving probiotics a pH-sensitive armor can reliably ferry them through the digestive gauntlet and deposit them where ulcerative colitis does the most damage. In mice, this strategy allowed much lower doses of bacteria to colonize the colon, repair the gut barrier, reduce harmful inflammation, and restore a healthier microbial community and metabolic profile. Because the plant-based coating itself largely passes through the body unchanged and the method can be applied to many bacterial types, the authors argue that their approach offers a broadly useful blueprint for designing next-generation “living” medicines for ulcerative colitis and other colon diseases.
Citation: Liu, J., Fang, Z., Li, X. et al. Surface decoration of bacteria generates robust living therapeutics for improved ulcerative colitis therapy. Commun Biol 9, 428 (2026). https://doi.org/10.1038/s42003-026-09732-6
Keywords: ulcerative colitis, probiotics, gut microbiome, drug delivery, intestinal barrier