Clear Sky Science · en
Single-nucleus RNA sequencing and functional studies of acute methamphetamine-induced cognitive impairment
Why this matters for brain health
Methamphetamine is often portrayed as a drug that “burns out” the brain, but what does that damage actually look like inside our cells? This study peers into the memory center of the mouse brain—the hippocampus—cell by cell to see how a short burst of heavy meth use scrambles the machinery that supports learning and memory. By mapping these changes in unprecedented detail, the work points to new biological weak spots that could one day be targeted to protect or restore brain function in people exposed to the drug.
How a binge dose of meth affects memory
The researchers first asked a simple question: does an intense, short-term meth dose harm memory in mice? Animals received four injections of meth within a day, mimicking a binge pattern. When tested, these mice struggled to recognize new objects and took longer to find a hidden platform in a water maze, classic signs of problems with learning and spatial memory. Even after the platform was removed, they searched less in the right place, suggesting that something in the hippocampus—the brain’s navigation and memory hub—was no longer working properly.
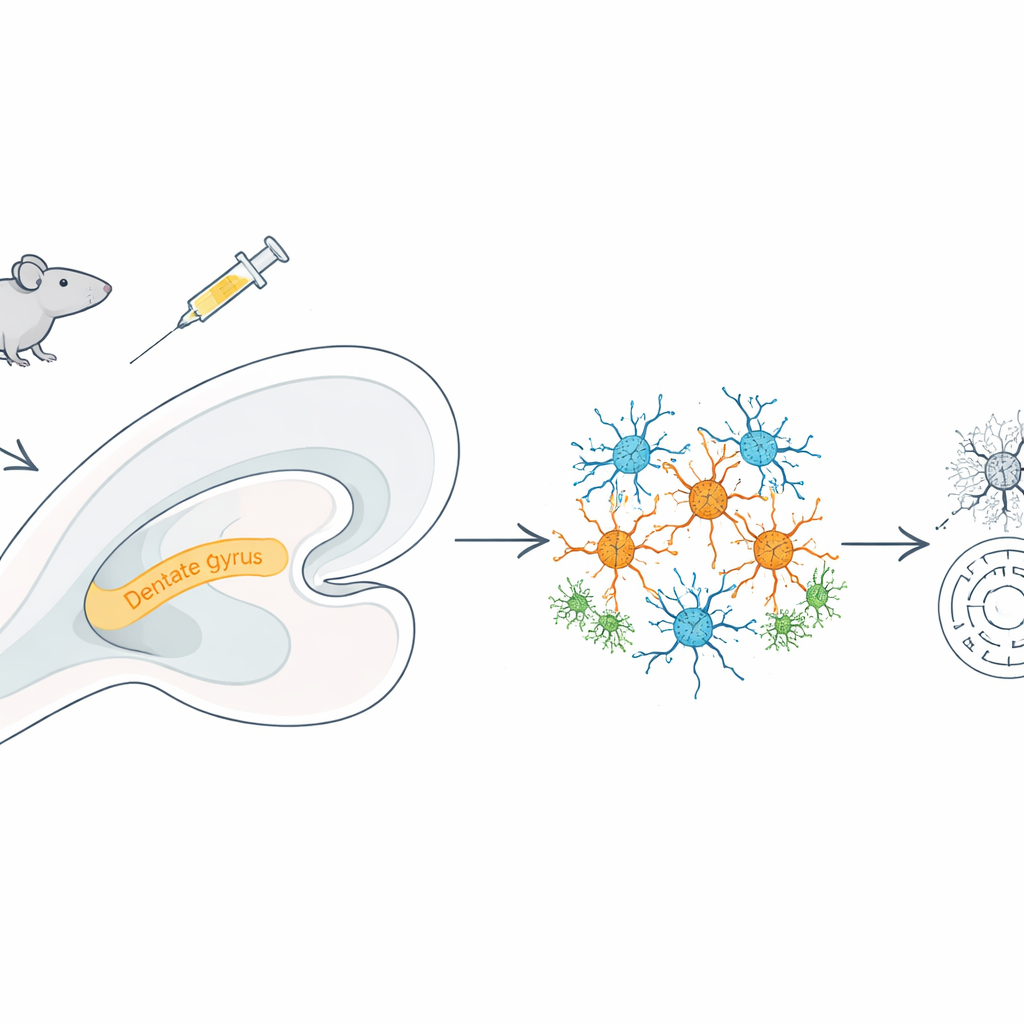
Looking at every nucleus, one by one
To understand what was going wrong, the team used single-nucleus RNA sequencing, a technique that reads which genes are turned on inside thousands of individual cell nuclei at once. From over 36,000 hippocampal nuclei, they identified ten major cell types, including excitatory and inhibitory neurons, astrocytes, microglia, oligodendrocytes, and blood-vessel cells. Meth exposure shifted this cellular landscape: there were more excitatory neurons, microglia, oligodendrocytes, and endothelial cells, but fewer inhibitory neurons. This tipped the balance toward excitation, a state that can make neural circuits more vulnerable to stress and damage.
Energy factories under stress and defenses running low
The most dramatic gene changes appeared in excitatory neurons, especially in a hippocampal region called the dentate gyrus, which is crucial for forming distinct memories. Here, genes tied to oxidative phosphorylation—the process by which mitochondria generate cellular energy—were strongly altered, and electron microscope images showed mitochondria that were fragmented, with broken inner structures. At the same time, pathways linked to reactive oxygen species and inflammatory signals were activated, and a class of organelles called peroxisomes, which help control damaging by-products of metabolism, showed signs of failure. Key peroxisome-related genes, including PEX5, dropped in activity, while genes that drive inflammation and oxidative stress rose, suggesting that meth pushes neurons into an energy crisis while weakening their natural detox systems.
Vulnerable cell groups and scrambled cell-to-cell conversations
Diving deeper, the researchers found that excitatory neurons could be divided into five regional subtypes, with dentate gyrus cells showing the strongest stress signals: heightened inflammation, greater oxidative burden, and a form of fiery cell death known as pyroptosis. Within this region they identified a subgroup of excitatory neurons heavily enriched in meth-treated animals that carried the strongest damage signatures, marking them as the likely casualties of exposure. Across the hippocampus, communication between excitatory neurons and other cell types intensified, while links from inhibitory neurons weakened. Microglia, the brain’s immune sentinels, shifted into a highly inflammatory mode. At the same time, the team traced clusters of genes whose activity was closely tied to memory performance, highlighting networks in excitatory neurons, inhibitory neurons, astrocytes, and oligodendrocytes that may either worsen or buffer cognitive decline.
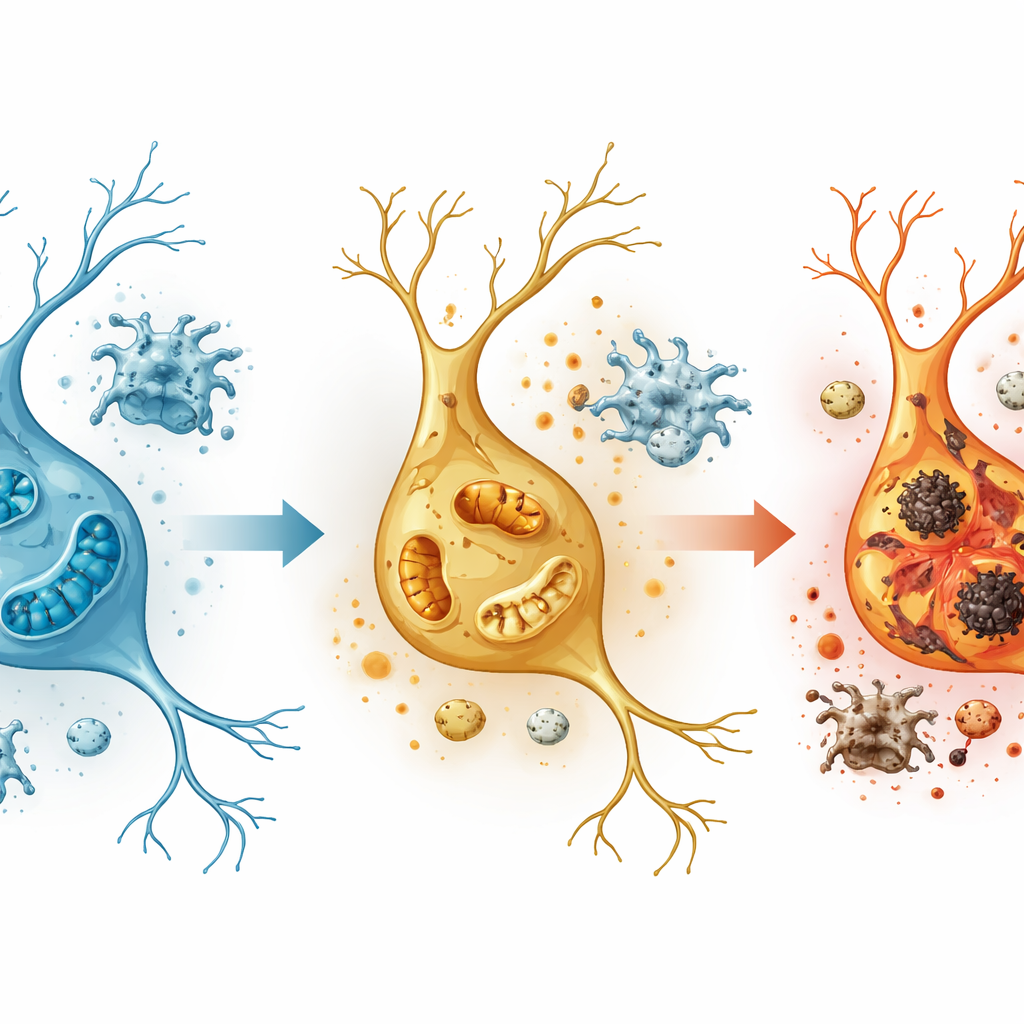
New molecular clues and possible brain-protective routes
The study also spotlighted specific molecules that may be central nodes in meth-induced injury. One, an RNA-processing protein called Ddx5, was strongly increased across many cell types, particularly in the dentate gyrus, hinting at a broad stress response whose protective or harmful role remains to be clarified. In contrast, PEX5 and the lipid-regulating switch PPARα, both important for keeping peroxisomes and mitochondria in balance, were dialed down in excitatory neurons. Taken together, these shifts suggest that restoring energy metabolism, shoring up peroxisome function, and calming overactive immune responses could be promising strategies to limit brain damage after acute meth exposure.
What this means in plain terms
In everyday language, this work shows that even a short-lived meth binge can leave a deep mark on the brain’s memory center. It throws key neurons in the dentate gyrus into an energy shortfall, overloads them with toxic by-products, weakens their clean-up systems, and stirs up nearby immune cells that further inflame the tissue. By charting exactly which cell types, regions, and gene networks are most affected, the study moves beyond the vague idea of “brain damage” and identifies concrete biological processes—like faulty energy production and broken detox pathways—that could be targeted to protect or repair the brain in people harmed by meth.
Citation: An, D., Lu, F., Wang, Y. et al. Single-nucleus RNA sequencing and functional studies of acute methamphetamine-induced cognitive impairment. Commun Biol 9, 440 (2026). https://doi.org/10.1038/s42003-026-09728-2
Keywords: methamphetamine, hippocampus, single-nucleus RNA sequencing, mitochondrial dysfunction, neuroinflammation