Clear Sky Science · en
A two-step actin-mediated strategy enables Campylobacter jejuni to promote mitochondrial aggregation and iron homeostasis, for intracellular survival and persistence
Why this tiny battle inside cells matters
Food poisoning from undercooked chicken is often blamed on a bacterium called Campylobacter jejuni. Most people recover, but this microbe is surprisingly good at surviving in many environments, which helps it spread from farm to fork. This study looks at what happens when the bacterium hides inside a free‑living amoeba—a common freshwater microbe—and uncovers a surprisingly sophisticated survival trick that revolves around reshaping the cell’s internal skeleton, clustering energy‑making “power packs” called mitochondria, and controlling iron and harmful oxygen chemistry. Understanding this hidden phase of its life cycle could point to new ways to curb contamination before the bacterium ever reaches people.
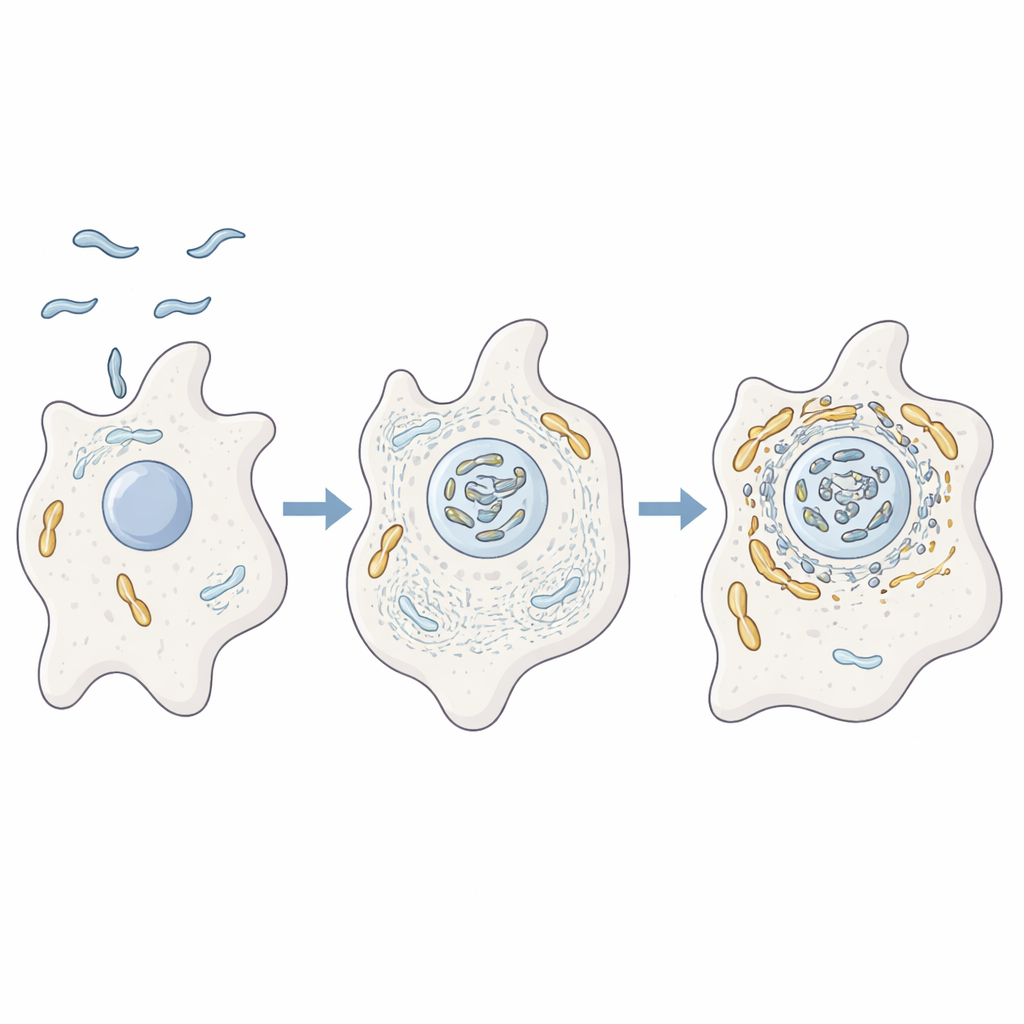
A microbe that trains inside amoebae
The authors studied how C. jejuni behaves inside the amoeba Acanthamoeba castellanii, a single‑celled organism that lives in soil and water. These amoebae act like training grounds for many disease‑causing bacteria, offering protection from harsh conditions and from disinfectants. Using advanced protein analysis, microscopy, and genetic mutants, the researchers showed that when C. jejuni infects amoebae it ends up closely associated with the host’s mitochondria. At the same time, the amoeba’s internal scaffolding made of actin, along with proteins that regulate its shape and movement, are strongly enriched near these mitochondria, suggesting the bacterium is actively rearranging the host cell’s inner architecture.
Bending the cell’s skeleton to move power packs
To test whether actin really drives this interaction, the team blocked actin from forming filaments or from branching into networks. Under these conditions, mitochondria no longer gathered near the bacteria and instead remained as scattered dots, and the bacterium’s survival dropped. They then focused on two bacterial helper proteins, CiaD and CiaI, which are injected into host cells through the bacterium’s flagellum, a whip‑like tail. CiaD turned out to boost actin growth and promote spiky projections on the amoeba surface, helping the cell scoop up the bacteria. Once inside, mitochondria were seen clustering around the bacterial compartment in an actin‑dependent manner, placing the bacterium in a privileged zone rich in host energy machinery.
A molecular switch that remodels mitochondria
While CiaD is key for entry, CiaI controls what happens afterward. When the researchers deleted the gene for CiaI, C. jejuni no longer triggered strong mitochondrial clustering and survived poorly inside amoebae. Adding purified CiaI attached to tiny beads, without any live bacteria, was enough to cause nearby mitochondria to fuse and cluster and to locally strip away actin. Biochemical tests showed that CiaI binds energy‑carrying molecules in a cooperative way, behaving like a molecular switch that may turn its activity on or off depending on conditions inside the host. The authors propose that after CiaD‑driven actin growth brings mitochondria close, CiaI drives local actin breakdown and reshapes the mitochondrial network into dense aggregates surrounding the bacteria‑containing compartment.
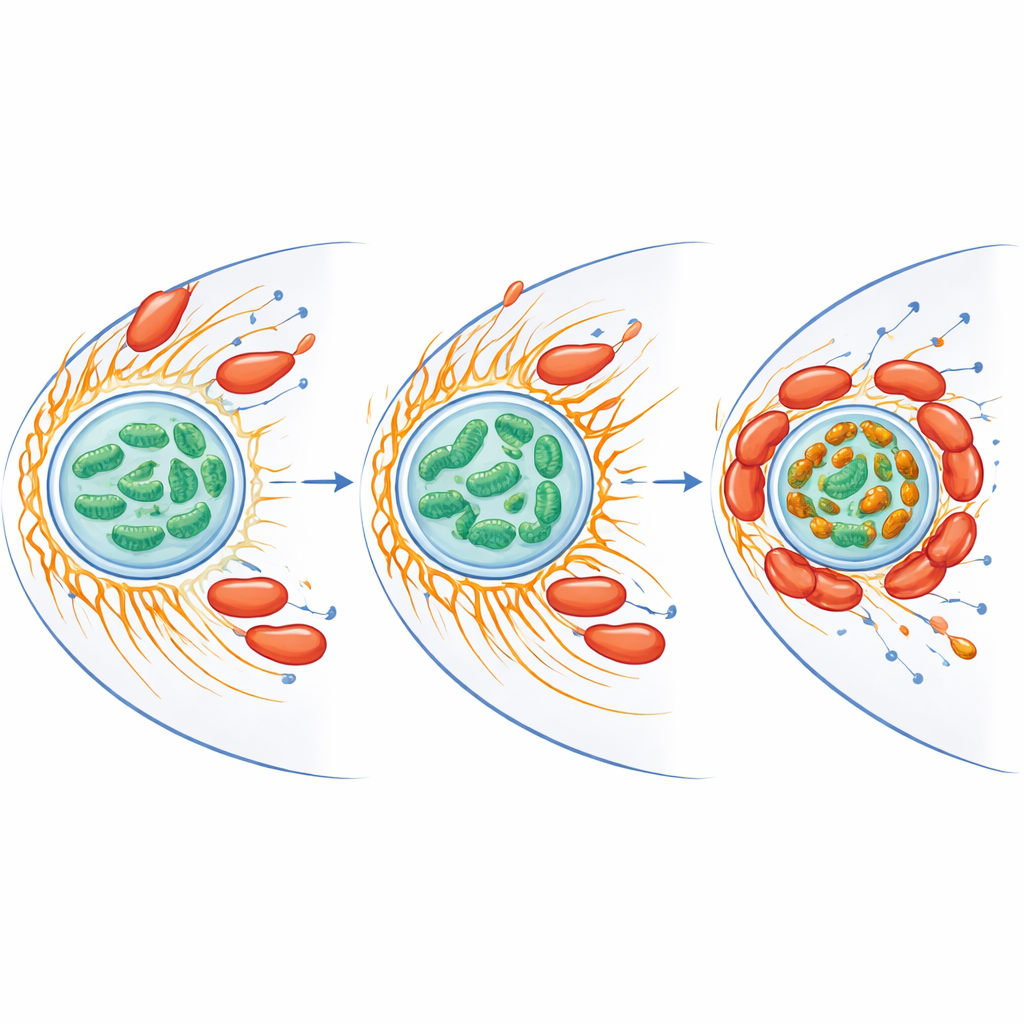
Rewiring iron and stress inside the host cell
Mitochondria do more than make energy—they also handle iron and produce reactive oxygen species, chemically aggressive forms of oxygen that can kill microbes. The researchers found that infection reduced overall free iron inside mitochondria but, at the same time, created bright iron‑rich clusters where mitochondria aggregated around the bacteria. Proteins involved in iron handling and in defending against oxidative stress rose in both host and bacterium. When the team removed free iron using a chelating compound, the amoeba’s burst of reactive oxygen species fell and C. jejuni survival increased dramatically. Mitochondria in infected cells also showed a higher membrane potential, a sign of elevated activity, suggesting the bacterium nudges the host’s power packs into a state that favors its own persistence while limiting damaging oxygen chemistry.
What this means for food safety and infection
Put together, the results support a two‑step strategy: first, CiaD stimulates actin growth to help the bacterium get inside amoebae and to pull mitochondria toward the entry site; second, CiaI locally dismantles actin and remodels mitochondria into iron‑rich clusters wrapped around the bacterial compartment, while blunting the host’s oxidative defenses. This finely tuned control of cell shape, energy use, and metal balance helps C. jejuni survive inside amoebae and likely contributes to its ability to persist in the environment and later infect humans. By revealing the key players in this microscopic tug‑of‑war, the study points to new targets—such as the Cia proteins, actin‑mitochondria contacts, or iron handling pathways—that could be disrupted to reduce contamination and disease.
Citation: Nasher, F., Wren, B.W. A two-step actin-mediated strategy enables Campylobacter jejuni to promote mitochondrial aggregation and iron homeostasis, for intracellular survival and persistence. Commun Biol 9, 431 (2026). https://doi.org/10.1038/s42003-026-09713-9
Keywords: Campylobacter, amoebae, mitochondria, actin cytoskeleton, iron homeostasis