Clear Sky Science · en
A dual respiratory and auditory function for the coelacanth lung
Ancient fish with a surprising sense
For decades, the rare coelacanth has been famous as a living fossil—a deep-sea fish that seems frozen in time. This study reveals that its extinct relatives may have hidden a remarkable trick inside their bodies: a lung that not only helped them breathe but also acted as part of their hearing system. By re-examining fossil skeletons and the inner ears of modern coelacanths with powerful X‑ray imaging, the authors propose that early coelacanths used an internal gas-filled organ to pick up sound vibrations, hinting at how hearing in our distant fish ancestors first evolved.

A strange organ in stone
Fossil coelacanths from over 200 million years ago preserve a row of large, thin bony plates inside the body. These plates enclose hollow chambers that were almost certainly filled with gas when the fish were alive. Earlier work suggested that this structure was an unusual type of lung, used for breathing. Yet its exact purpose remained a mystery, because modern coelacanths no longer carry such a large organ, retaining only a tiny shriveled remnant near the gut. The new study focuses on two exceptionally well-preserved Triassic species from France, whose three-dimensional skeletons were scanned in detail. The scans show that their internal chambers formed a multi-part, plate-covered organ positioned in the abdominal cavity, strongly supporting its identity as a lung-like structure.
Clues from delicate bony ridges
In both fossil species, the front chamber of this lung bears a pair of tall, blade-like ridges that extend upward toward the spine. The authors call these features “chamber wings.” In life, the wings would have attached to the tough outer sheath of the notochord, the flexible rod that supports the backbone. While these ridges could simply have helped suspend the lung, their shape and position suggest something more: they sit very close to where pathways from the inner ear would have run toward the back of the skull. The team proposes that the wings acted like receivers, conducting pressure changes from the gas-filled lung into nearby tissues, and then toward the head.
The inner ear map of a living fossil
To test this idea, the researchers turned to the modern coelacanth, Latimeria, whose soft tissues can still be studied. Using synchrotron X‑ray scans and old histological slides, they reconstructed the fish’s inner ear and surrounding spaces in three dimensions. They uncovered a complex, unpaired network of fluid-filled canals—known as the perilymphatic system—that links the brain cavity to the ear. These canals connect to two distinct patches of sound-sensitive tissue, called papillae, which resemble those found in amphibians. In Latimeria today, the lung is tiny and the back portion of this canal system is clogged with connective tissue, hinting that whatever role it once played has largely been lost.
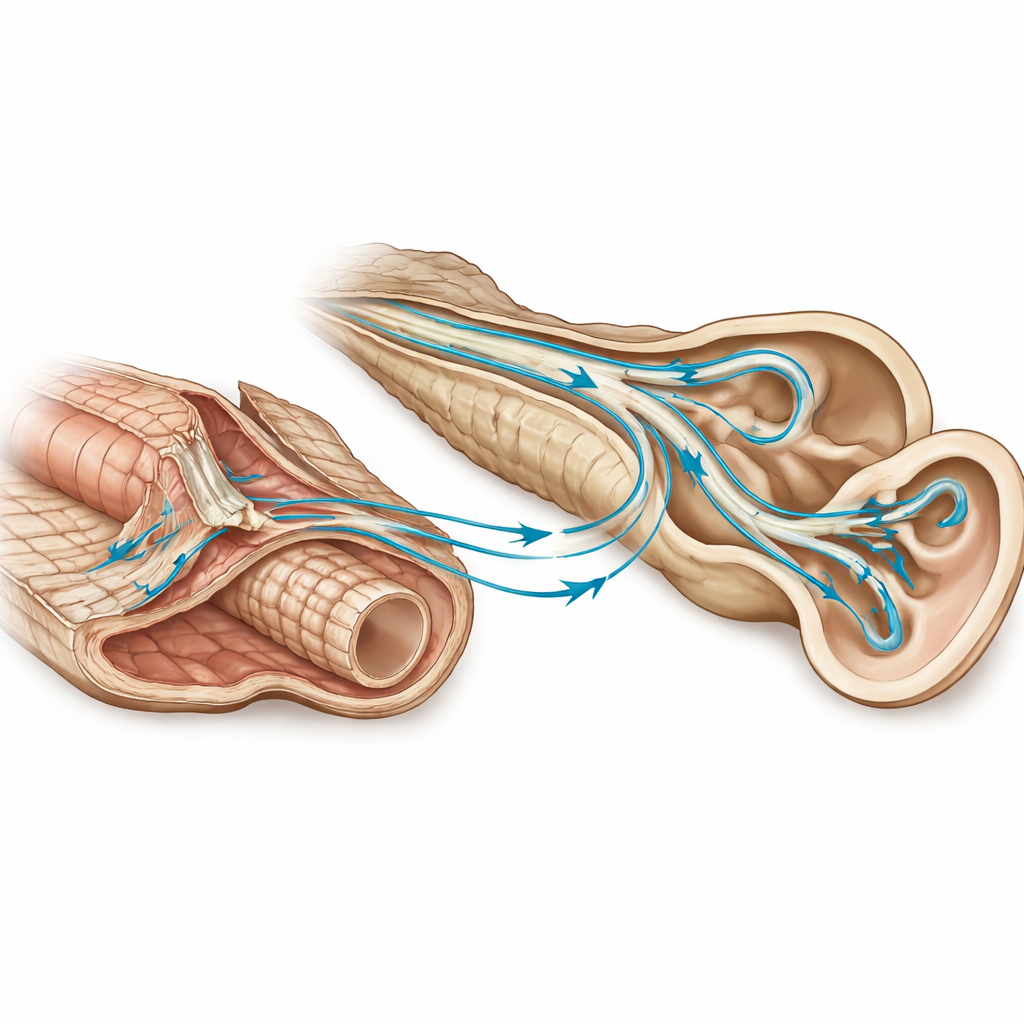
Rebuilding an ancient hearing pathway
By comparing modern Latimeria with a heavily ossified Devonian coelacanth and the new Triassic fossils, the authors argue that this perilymphatic canal layout is an ancestral feature of the group. In extinct species with large gas-filled lungs, sound waves in water would have compressed the gas in the chambers, causing the chamber wings and attached tissues to vibrate. Those vibrations could have travelled along the notochord and into the perilymphatic canals, finally reaching the two papillae in the inner ear. In this scenario, the lung served a dual purpose: supplying oxygen and acting as a built-in sound receiver, much as some modern fishes use their swim bladders to enhance hearing.
What this means for our own story
The study concludes that early coelacanths likely possessed lungs that functioned both in breathing and in detecting sound pressure, long before the evolution of a classic middle ear and eardrum in land vertebrates. This suggests that inner ear organs capable of sensing airborne or gas-borne vibrations may have existed in our distant fish relatives, setting the stage for later adaptations as vertebrates moved onto land. In other words, part of the machinery that lets humans hear may trace its roots back to ancient fishes whose lungs doubled as listening devices.
Citation: Manuelli, L., Clément, G., Herbin, M. et al. A dual respiratory and auditory function for the coelacanth lung. Commun Biol 9, 400 (2026). https://doi.org/10.1038/s42003-026-09708-6
Keywords: coelacanth, fossil lung, evolution of hearing, inner ear, sarcopterygian fishes