Clear Sky Science · en
Multilayered regulation of GluK3 kainate receptors is mediated by Neto subunits and zinc
Why tiny brain switches matter
Every thought, memory, and mood in the brain depends on microscopic switches that let charged particles flow in and out of nerve cells. This study focuses on one such switch, a little‑known receptor called GluK3. By uncovering how partner proteins and zinc ions fine‑tune GluK3’s behavior, the work offers clues to how brain circuits stay balanced—and how that balance may tip in disorders such as epilepsy, depression, and schizophrenia.
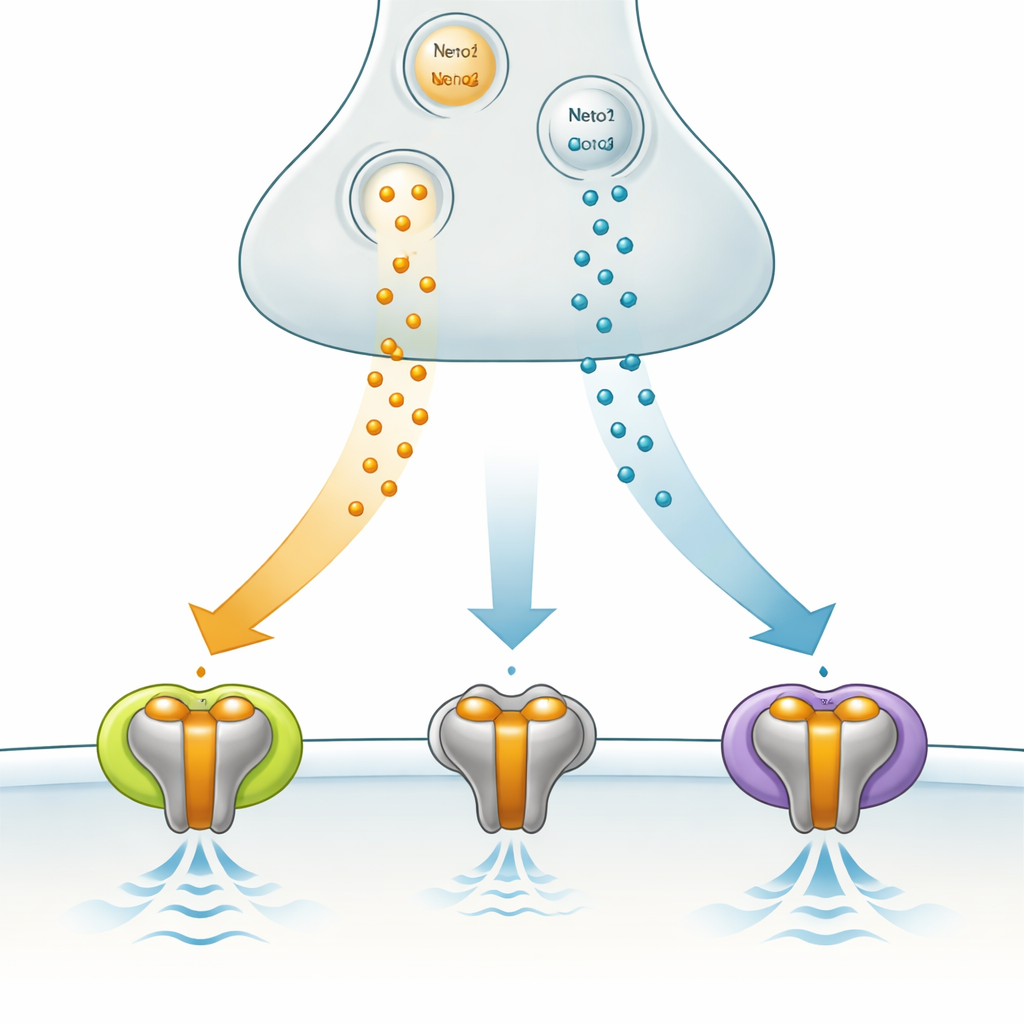
A specialized signal gate in the brain
GluK3 belongs to the kainate receptor family, part of the larger group of glutamate receptors that mediate fast communication between neurons. Unlike some of its cousins, GluK3 does not simply turn signals on or off; it acts more like a filter that responds best to brief, strong bursts of the chemical messenger glutamate. It is enriched in brain regions such as the hippocampus, which is crucial for memory and patterning network rhythms, and changes in GluK3 function have been linked to anxiety‑related behaviors. These features make GluK3 a promising, but still poorly understood, target for shaping brain activity in health and disease.
Helper proteins that pull the strings
The team examined how two helper proteins, Neto1 and Neto2, alter GluK3 behavior in human cells engineered to produce these components. Both helpers slowed how quickly GluK3 switches shut off after being activated, and both reduced a natural form of internal blockage that normally limits current flow. However, they had opposite effects on how fast GluK3 can be reused. With Neto1, receptors bounced back quickly, ready to respond to rapid, repeated signals. With Neto2, recovery became much slower, favoring integration of signals over longer intervals. In essence, by choosing different helper proteins, a synapse can decide whether GluK3 behaves like a rapid‑fire detector or a slower, averaging sensor.
Zinc as a second layer of control
Many glutamate‑releasing nerve terminals also release zinc, which can bind to receptors and change how they work. Earlier work showed that zinc unusually boosts GluK3 activity, while tending to dampen other receptor types. Here, the authors found that this boost depends strongly on which Neto protein is present. When GluK3 acts alone, zinc roughly doubles the time the receptor stays active and modestly increases current. Adding Neto1 blunts this effect. By contrast, when GluK3 partners with Neto2, zinc and Neto2 act together to greatly enhance the current, far more than either could alone. This suggests that synapses rich in Neto2 and zinc can strongly amplify GluK3‑mediated signals during intense activity, whereas Neto1‑rich synapses remain more restrained.
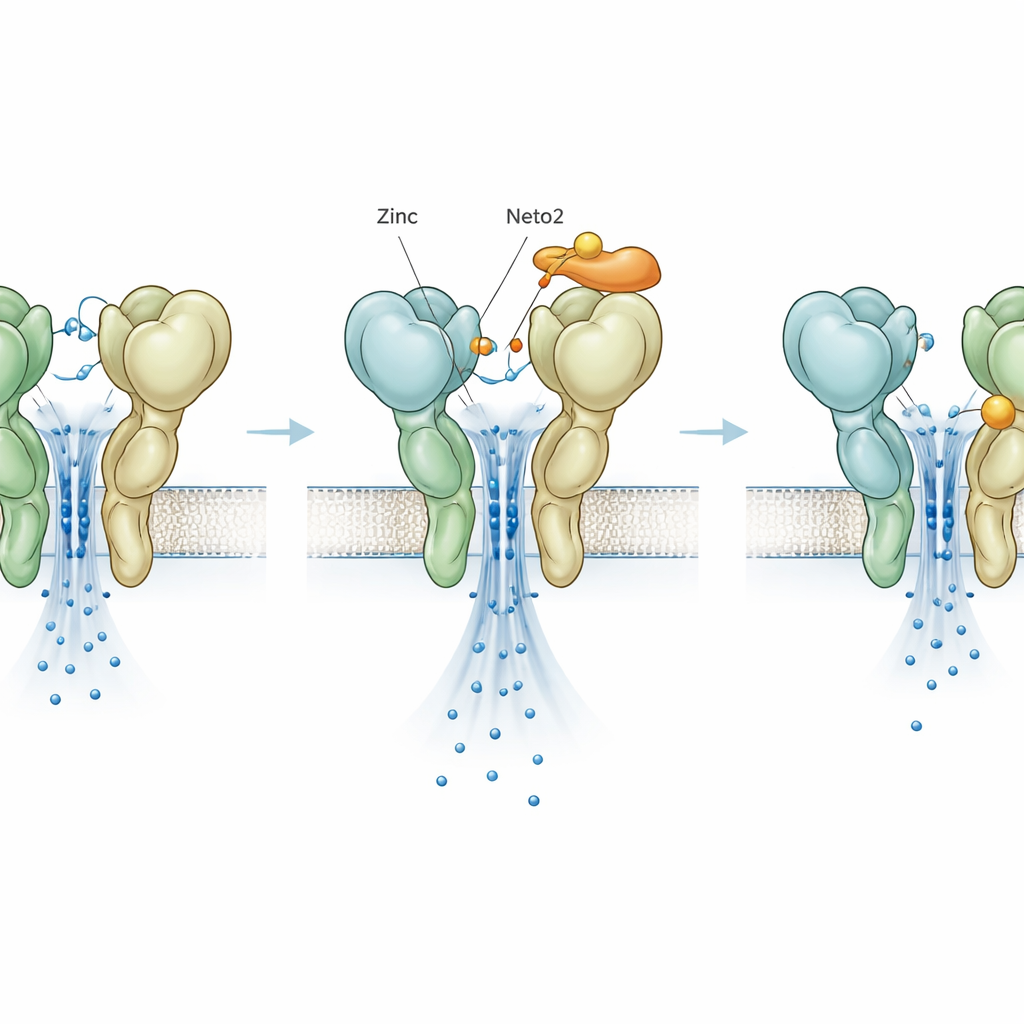
A hidden brake revealed by mutation
To separate zinc’s actions from those of the helper proteins, the researchers engineered a single‑letter change in GluK3, called D759G, that removes a known zinc‑binding site. As expected, this mutation itself made the receptor more stable and slower to switch off, mimicking the usual effect of zinc. Surprisingly, when zinc was added to this mutant, it no longer boosted activity; instead, it sped up shutoff and reduced current, revealing a second, previously hidden zinc site that acts as a brake. Neto1 and Neto2 still exerted their characteristic effects on the mutant, showing that their core influence does not depend on the original zinc site. Yet even this newly uncovered inhibitory zinc action was tuned differently by the two helpers, adding another adjustable dial to the system.
Seeing structure behind behavior
To connect function to form, the team used cryo‑electron microscopy to visualize the mutant GluK3 receptor frozen in a non‑active state. The images revealed that the region which binds glutamate forms a more compact, tightly packed unit in the D759G mutant compared with normal GluK3. This structural tightening likely makes it harder for the receptor to swing into a shut‑off shape, explaining why the mutant, like zinc‑bound GluK3, stays active longer. At the same time, the images showed that not all parts of the receptor were locked into one arrangement, pointing to an inherently flexible architecture that is especially sensitive to small chemical nudges.
What this means for brain health
Taken together, the study portrays GluK3 not as a simple on–off switch but as a finely adjustable hub where glutamate, helper proteins, and zinc converge. Neto1 and Neto2 set how quickly signals fade and how fast receptors can respond again, while zinc can either further boost or, under some conditions, restrain activity through multiple binding sites. Because GluK3, Neto proteins, and zinc all co‑exist at synapses involved in memory and are implicated in epilepsy and psychiatric conditions, understanding this multilayered control may guide future therapies that gently retune synaptic signaling rather than shutting it down outright.
Citation: Vinnakota, R., Dawath, B.K., Assaiya, A. et al. Multilayered regulation of GluK3 kainate receptors is mediated by Neto subunits and zinc. Commun Biol 9, 420 (2026). https://doi.org/10.1038/s42003-026-09707-7
Keywords: kainate receptors, GluK3, Neto proteins, synaptic zinc, synaptic plasticity