Clear Sky Science · en
Exploring microbial diversity using cell-size fractionated enrichment incubations from subsurface aquifers at Äspö, Sweden
Hidden Life Deep Below Our Feet
Far beneath the forests and coasts of Sweden, in cold, dark fractures of ancient rock, vast communities of microbes quietly endure on a trickle of energy. These tiny organisms help drive global cycles of carbon and sulfur, yet many of them are so small and so dependent on neighbors that they have eluded scientists for decades. This study peers into that underground world, revealing a surprisingly rich “microbial dark matter” made up of ultra-small cells that seem to survive by living in close association with other microbes rather than by growing quickly on fresh food.
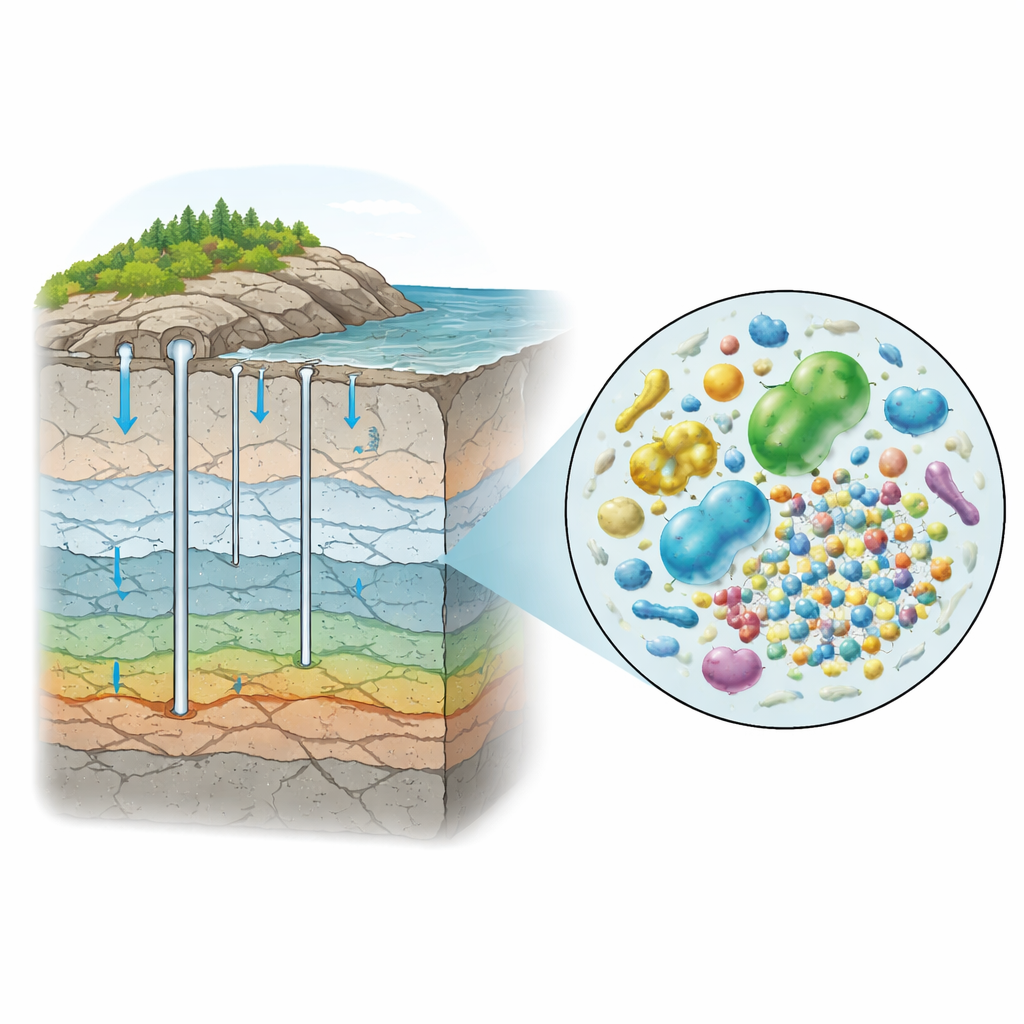
Deep Underground Water as a Hidden Habitat
The researchers worked at the Äspö Hard Rock Laboratory on Sweden’s Baltic coast, where tunnels cut through ancient granite hundreds of meters below the surface. There, groundwater slowly moves through narrow cracks in the rock. Some of this water is relatively young and influenced by rain and soil; some is more like diluted seawater; and some is very old and salty, cut off from the surface for long periods. Earlier work had shown that these waters host diverse microbial communities, including many unusual lineages with extremely small cells and genomes.
Focusing on the Smallest Cells
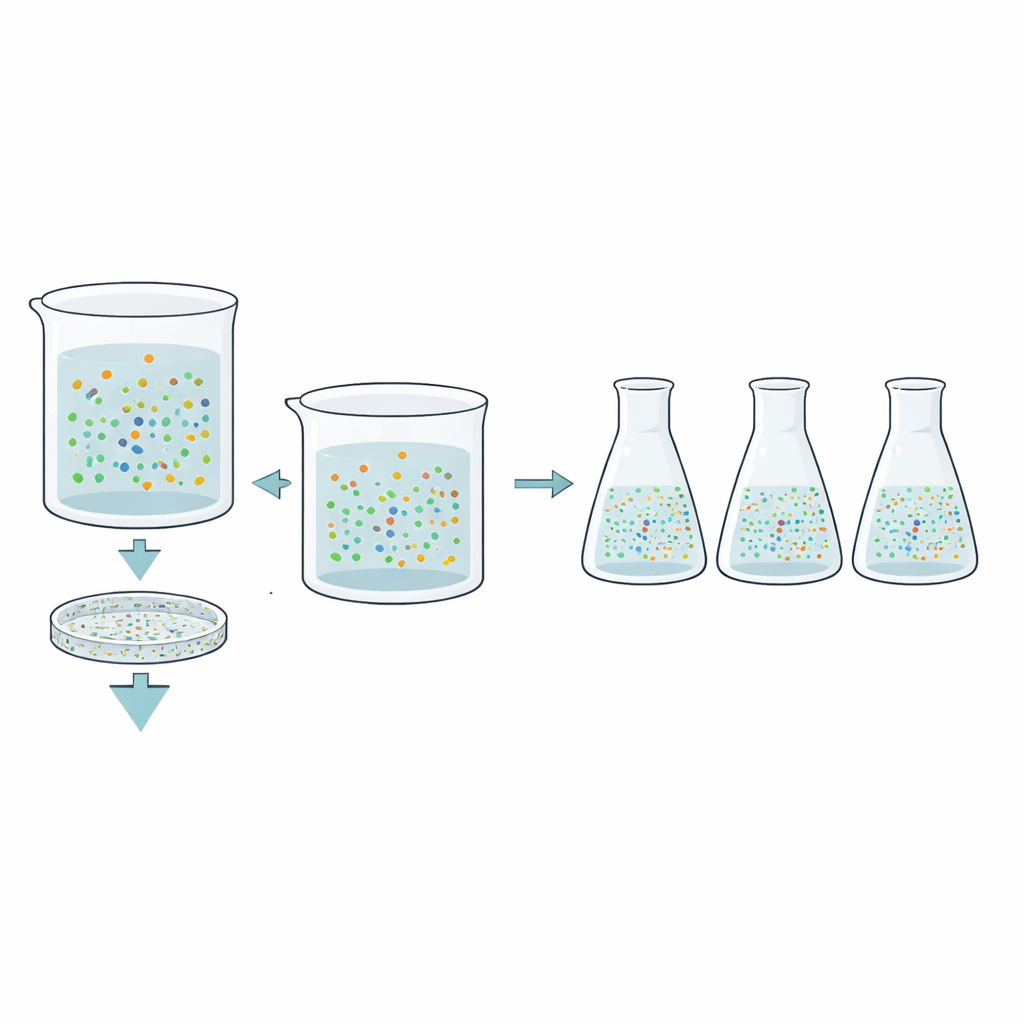
To study these elusive microbes, the team collected anoxic groundwater from three different depths and chemistries—meteoric (soil-influenced), marine, and saline. They then set up anaerobic incubations in the lab, adding either a simple carbon source (acetate) or a complex mixture made from broken bacterial cells. Crucially, for many incubations they first passed the groundwater through a filter that removed cells larger than 0.45 micrometers. What slipped through were ultra-small bacteria and archaea, such as groups known as Patescibacteria, Nanobdellota, and Omnitrophota, which are suspected to have limited metabolisms and to rely on close partners or hosts.
Diversity Without Growth
Over weeks to months, the scientists tracked cell numbers and community composition using DNA sequencing and microscopic counts. In incubations that contained the full, unfiltered groundwater, total cell numbers typically rose by about tenfold, but diversity dropped as just a few fast-growing groups—such as Bacillota, Spirochaetota, or Desulfobacterota—came to dominate. In striking contrast, the size-fractionated incubations, which held only ultra-small cells, showed almost no increase in cell numbers, even when given extra acetate or rich cell lysate. Yet their genetic diversity remained very high, often matching that of the original groundwater. This suggested that a large share of subsurface biodiversity resides in these tiny forms that persist without obvious growth under the provided conditions.
Genetic Clues to a Dependent Lifestyle
To understand how these microbes make a living, the team reconstructed dozens of draft genomes from the incubations. Larger-genome bacteria, such as Desulfobacterota and some Pseudomonadota, carried genes for a wide range of metabolic tricks: breaking down complex organic matter, respiring oxygen or sulfate, and even fixing carbon dioxide. By contrast, genomes from Patescibacteria and Nanobdellota were tiny and stripped-down. They lacked full pathways for central carbon metabolism and showed few capabilities for independent growth, yet they did carry genes for cell division, cell-wall construction, attachment structures, and transport systems. This combination is consistent with a lifestyle in which they cling to or closely associate with other microbes, scavenging key nutrients from their partners rather than fully manufacturing them.
Microbial Partnerships in the Dark
Looking across many groundwater samples and incubations, the researchers also examined which microbial lineages tend to appear together. They found strong patterns of co-occurrence between Patescibacteria and several other groups, especially Desulfobacterota, Chloroflexota, and Omnitrophota. While these patterns do not prove direct host–symbiont relationships, they fit the idea that ultra-small microbes may cluster around metabolically versatile neighbors that can supply missing building blocks and energy. Such partnerships could be a key survival strategy in the deep subsurface, where energy is scarce and resources arrive slowly and unpredictably.
Why This Matters for Life on Earth
Overall, the study shows that a major fraction of microbial diversity in deep, low-energy groundwaters is made up of ultra-small cells that barely grow in standard enrichment experiments and likely depend on tight interactions with other microbes. Rather than booming when extra food is added, these lineages remain numerically stable yet taxonomically rich, hinting at a slow, tightly coupled web of life that operates on timescales and energy budgets very different from surface ecosystems. Understanding these hidden communities not only reshapes estimates of Earth’s biomass and biodiversity, but also offers a window into how life can persist under extreme scarcity—a lesson that may apply to subsurface environments on other planets as well.
Citation: Westmeijer, G., Turner, S., Hevele, P. et al. Exploring microbial diversity using cell-size fractionated enrichment incubations from subsurface aquifers at Äspö, Sweden. Commun Biol 9, 378 (2026). https://doi.org/10.1038/s42003-026-09706-8
Keywords: deep subsurface groundwater, ultra-small microbes, microbial symbiosis, Patescibacteria, low-energy ecosystems