Clear Sky Science · en
Addictive drug abuse and depression-a focus on epigenetics
Why this research matters to everyday life
Addictive drugs and depression are often treated as separate problems, yet they frequently appear together in the same person. This review explains how long-term use of drugs such as methamphetamine, cocaine, opioids, and cannabis can leave lasting "molecular scars" in the brain that increase the risk of depression. By uncovering these hidden changes, the article points toward future tests that could warn who is most vulnerable—and more precise treatments that might reverse the damage rather than only masking symptoms.
How addiction and low mood become intertwined
The authors begin by outlining the close clinical link between drug abuse and depression. People who repeatedly use addictive substances are far more likely to report persistent sadness, loss of pleasure, sleep problems, and suicidal thoughts. Brain regions that control reward, motivation, memory, and decision-making—the ventral tegmental area, nucleus accumbens, prefrontal cortex, and hippocampus—are central to both conditions. Chronic drug exposure disrupts brain chemicals like dopamine and serotonin, stress hormones controlled by the hypothalamic–pituitary–adrenal axis, immune signals, and the energy supply of neurons. Together these changes make the brain less resilient to stress and more prone to depressive states, even long after the last dose.
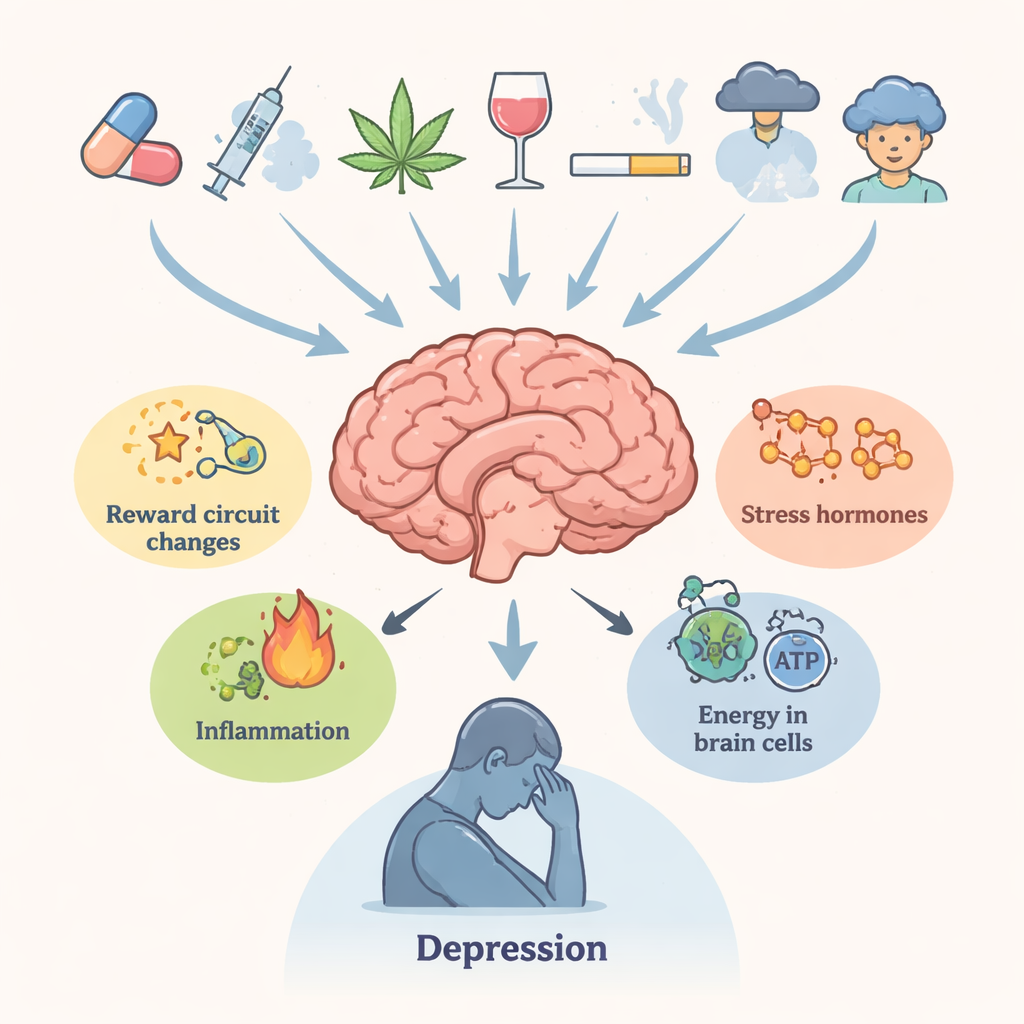
Drugs that reshape the brain’s chemistry
The review surveys several major drug classes. Amphetamine-type stimulants and their relatives, including synthetic "bath salts," cause powerful surges of dopamine and other transmitters that, over time, damage nerve endings, disturb glutamate balance, strain the endoplasmic reticulum, and push cells toward harmful self-digestion (autophagy). Cannabis use in adolescence is linked to later depression and suicide risk, likely through long-lasting changes in cannabinoid receptors, white matter structure, and sensitivity of dopamine circuits. Opioids, while relieving pain, can trigger inflammation, mitochondrial failure, and reduced growth-support signals like brain-derived neurotrophic factor (BDNF). Cocaine repeatedly activates the stress system and alters key regulators such as FKBP5, contributing to anxiety and low mood during withdrawal. In each case, the biological disruptions tend to persist, helping explain why emotional symptoms can outlast obvious intoxication.
Epigenetics: the memory of exposure
To understand why drug effects endure, the authors focus on epigenetics—chemical tags and molecular switches that adjust gene activity without altering DNA letters. Addictive drugs modify DNA methylation (a mark on cytosine bases), change chemical groups on histone proteins that package DNA, and alter noncoding RNAs that fine-tune which proteins are produced. For example, methamphetamine and cocaine shift methylation patterns in reward-related brain regions, changing the levels of enzymes that add or remove these marks. Opioids and stimulants remodel histone acetylation and methylation on genes controlling glutamate receptors, stress signaling, and synaptic structure. Dozens of microRNAs, long noncoding RNAs, and circular RNAs rise or fall with drug exposure, collectively influencing inflammation, neuron growth, and the strength of synapses. These epigenetic changes act like a molecular “memory” of drug history.
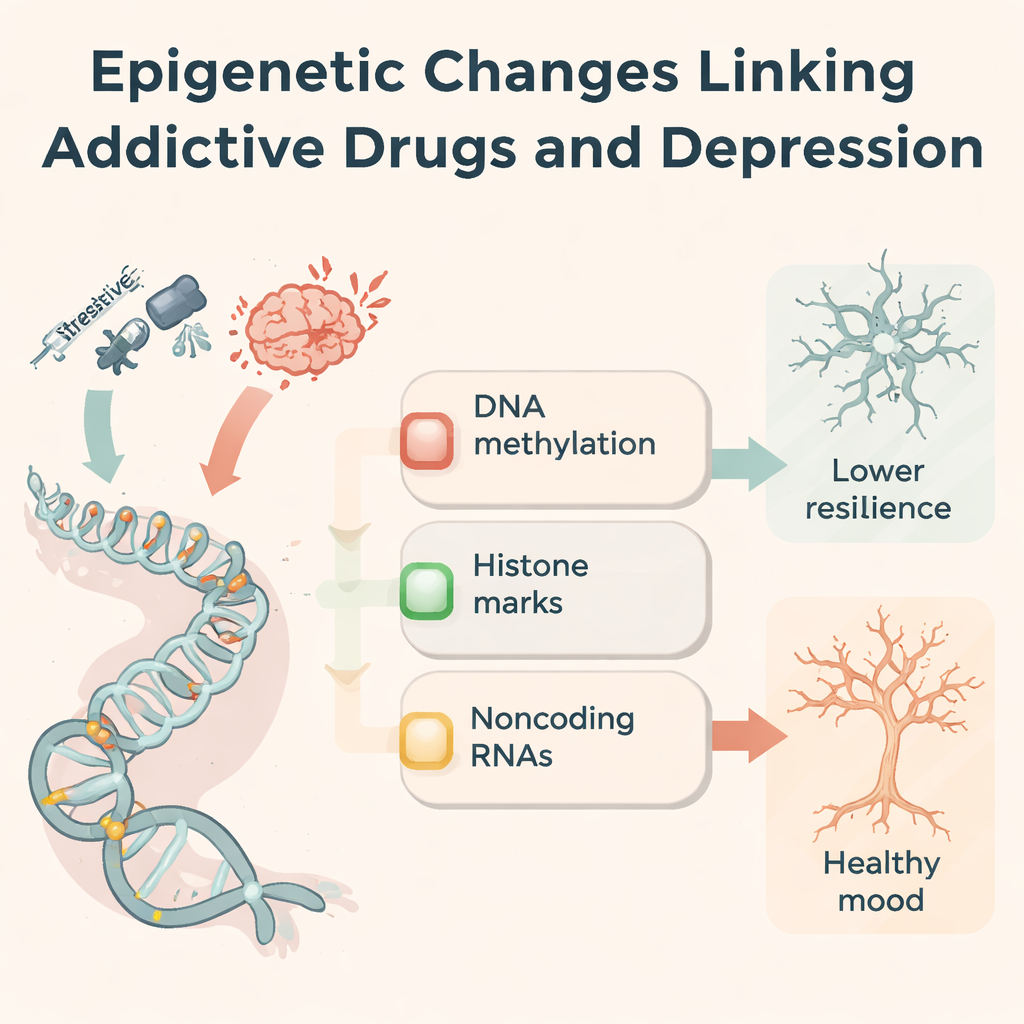
Shared molecular pathways in depression and addiction
The same epigenetic machinery also appears in people and animals with depression who have never taken drugs. Stressful experiences can alter methylation of stress hormone receptors (such as NR3C1 and FKBP5), reduce helpful growth factors like BDNF, and rewire immune and glutamate pathways. Many of the same noncoding RNAs changed by drugs are also misregulated in depression, affecting how new neurons are born, how microglia respond to inflammation, and how strongly synapses connect. The authors propose a three-part framework: stress-response regulation, reward-circuit remodeling, and synaptic plasticity. Across these axes, addiction and depression repeatedly converge on a small set of genes and marks, suggesting a biological reason why the two disorders so often co-occur.
What this means for future prevention and treatment
In closing, the review argues that epigenetic changes could become early-warning biomarkers and targets for next-generation treatments. Broad-acting drugs that wipe epigenetic marks have already shown some antidepressant-like effects in animals, but they are too blunt for routine human use. New tools—such as CRISPR-based epigenome editors and therapies that modulate specific microRNAs or long RNAs—may one day adjust problem genes in particular brain cells while sparing others. The authors caution that most current data come from rodents and bulk brain tissue, and that human biology is more complex. Still, by revealing how addictive drugs and depression write on the same molecular "notebook," this work offers a hopeful path toward more personalized and enduring therapies.
Citation: Zhang, W., Xu, M., Wang, C. et al. Addictive drug abuse and depression-a focus on epigenetics. Commun Biol 9, 297 (2026). https://doi.org/10.1038/s42003-026-09705-9
Keywords: addiction and depression, epigenetics, drug abuse, DNA methylation, noncoding RNA