Clear Sky Science · en
Aromatic patch in whiB-like transcription factors facilitates primary sigma factor interaction in mycobacterium tuberculosis
How Bacteria Sense Danger
Mycobacterium tuberculosis, the germ that causes tuberculosis, survives inside the human body by quickly reprogramming which genes are turned on or off when conditions change, such as during exposure to antibiotics or immune attack. This study uncovers a tiny structural feature—an "aromatic patch"—in a family of bacterial proteins that helps them grab onto the cell’s main gene-switching machinery. Understanding this microscopic handshake reveals how tuberculosis and related bacteria adapt, and may point toward new ways to weaken this pathogen.
A Special Family of Bacterial Switch Proteins
The work centers on WhiB-like (Wbl) proteins, a group found only in actinobacteria and their viruses, including Mycobacterium tuberculosis. These proteins carry a small iron–sulfur cluster, a metal-based cofactor that lets them sense changes in oxygen and other stresses. Wbl proteins are known to control key processes such as cell division, response to oxidative and nutrient stress, and resistance to antibiotics. Yet most of them lack the classic shapes used by many regulators to stick directly to DNA, leaving a long-standing mystery: how do they actually control gene activity?
Grabbing the Main Gene-Reading Machine
Earlier work showed that several Wbl proteins activate genes by binding to a conserved region, called region 4, of the primary sigma factor. The sigma factor is the part of RNA polymerase—the enzyme that reads DNA—that recognizes where to start copying genes. In Mycobacterium tuberculosis, this primary sigma factor uses the same region 4 to recruit many different regulators. The authors combined X-ray crystallography, biochemical pull-downs, and calorimetry to show that essentially all tested Wbl proteins from this bacterium (with one special case) latch onto the very same spot on sigma factor region 4 and do so with very tight binding.
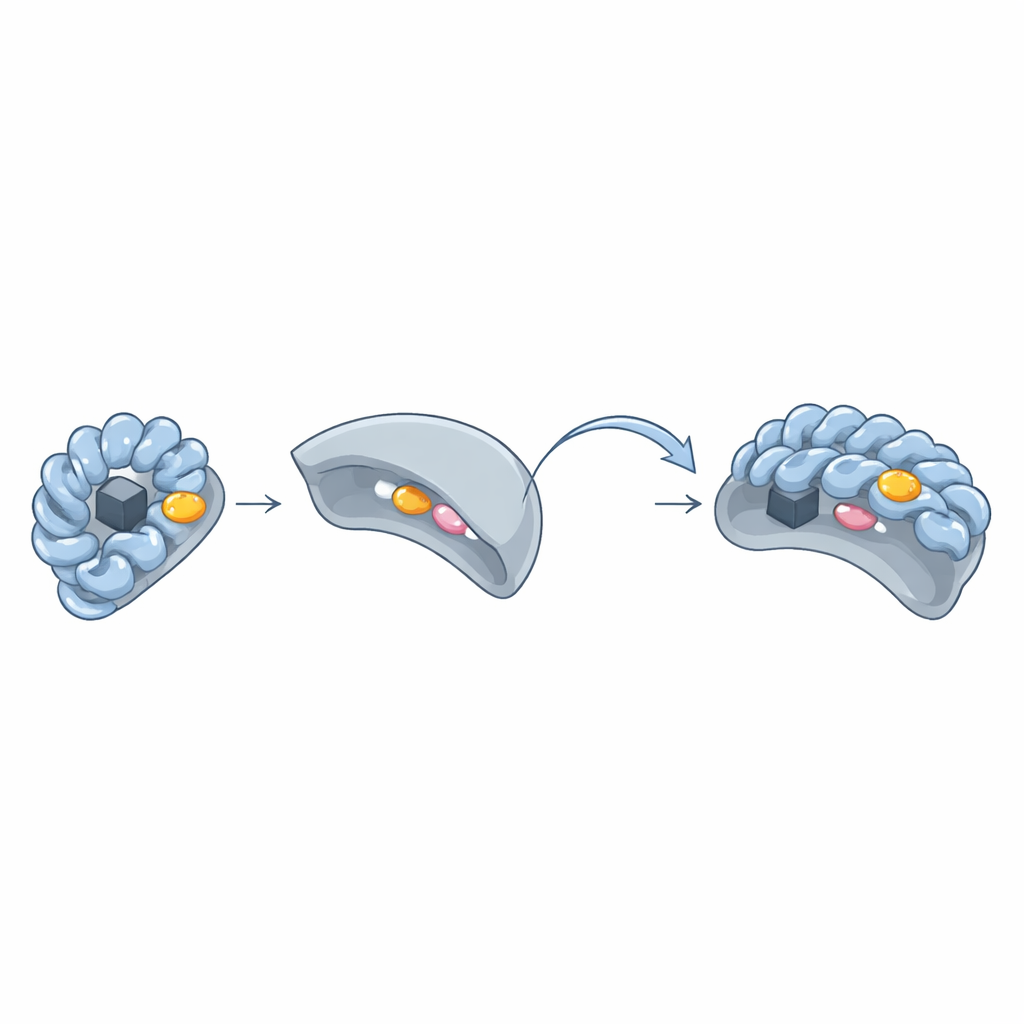
The Hidden "Aromatic Patch" Lock and Key
By comparing three-dimensional structures of Wbl–sigma pairs, the team discovered a recurring cluster of bulky ring-shaped amino acids—tryptophan, phenylalanine, tyrosine, or histidine—forming an "aromatic patch" on the surface of Wbl proteins. This patch sits around the iron–sulfur cluster and presses directly against two key amino acids in sigma region 4. When the researchers changed these aromatic patch residues to simpler ones, the Wbl proteins could no longer form stable complexes with sigma, and their iron–sulfur clusters often became unstable. Even in Wbl variants that appeared unusual—such as WhiB6 or WhiB5—either alternative residues or nearby aromatic side chains compensated to preserve the same style of interaction.
A Shared Design Across Bacteria and Their Viruses
To see how widespread this feature is, the authors analyzed 995 Wbl protein sequences from many actinobacterial species and their infecting viruses (actinobacteriophages). They grouped these proteins into 29 subfamilies and found that five major branches, represented by five Mycobacterium tuberculosis Wbl proteins, account for about 80% of all sequences. Structural modeling with AlphaFold revealed that almost all Wbls—more than 98%—carry at least two aromatic residues in positions matching the aromatic patch, and nearly all have at least one in the most critical central positions. Experiments with several phage-encoded Wbl proteins confirmed that these viral versions also bind to the same sigma region in a patch-dependent manner, indicating that the same molecular design is reused across bacteria and their phages.
An Evolutionary Tug-of-War Over Control
The phylogenetic tree built from these 995 sequences shows phage and bacterial Wbl proteins intermingled, with clear signs of horizontal gene transfer in both directions. Some viral Wbls sit at the base of major bacterial branches, suggesting that phages may have gifted these regulators to early bacteria, which then adapted them for their own needs. Other viral Wbls appear embedded within largely bacterial clusters, indicating later gene transfers back into phages. Because Wbl proteins tightly control stress responses, cell development, and drug resistance through their sigma-binding aromatic patches, these back-and-forth exchanges likely shaped how both bacteria and their viruses manipulate the host transcription machinery.
What This Means for Tuberculosis and Beyond
In simple terms, this study shows that many actinobacterial regulators share a tiny but crucial sticky spot—the aromatic patch—that lets them dock onto the same part of the gene-reading engine and fine-tune which genes are active under stress. In Mycobacterium tuberculosis, this shared docking mechanism helps coordinate responses that support persistence, virulence, and antibiotic resistance. By revealing how this microscopic interface works and how widely it is conserved, the work highlights a potential weak point that might one day be targeted to disrupt the pathogen’s ability to adapt and survive inside its host.
Citation: Guiza Beltran, D., Wan, T., Seravalli, J. et al. Aromatic patch in whiB-like transcription factors facilitates primary sigma factor interaction in mycobacterium tuberculosis. Commun Biol 9, 424 (2026). https://doi.org/10.1038/s42003-026-09698-5
Keywords: Mycobacterium tuberculosis, transcription factors, sigma factor, iron-sulfur proteins, bacteriophage evolution