Clear Sky Science · en
Heterogeneity between VIP and GRP neurons underlies AVP receptor signaling in the mouse suprachiasmatic nucleus
Why our body clock is so hard to reset
Anyone who has stumbled off a long flight or struggled with night shifts knows that our internal clock does not like sudden schedule changes. This paper asks a very specific question at the heart of that problem: inside the brain’s master clock, why do some nerve cells strongly feel a timing signal while their neighbors largely ignore it? By tracing a single chemical signal through this tiny clock circuit in mice, the authors uncover a hidden subgroup of cells that help determine how stubborn our body clock is when facing “jet lag.” 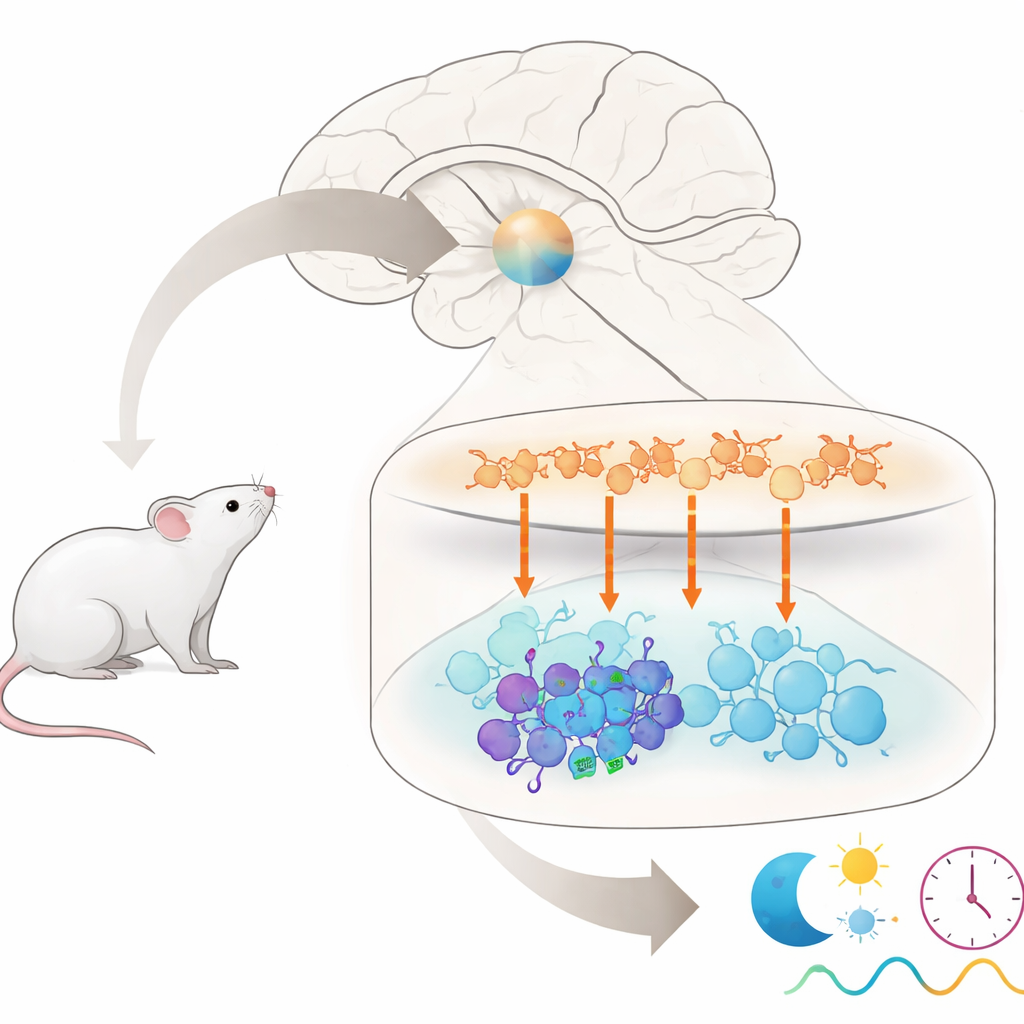
A tiny clock with many different players
The master clock in mammals sits in a small region of the brain called the suprachiasmatic nucleus, or SCN. Though it contains only about 20,000 nerve cells, it is made up of several distinct groups that talk to each other using different chemical messengers. Cells in the upper “shell” region mainly release a messenger called arginine vasopressin (AVP), while cells in the lower “core” region often release other messengers, including vasoactive intestinal peptide (VIP) and gastrin-releasing peptide (GRP). Together, these groups must stay in sync to generate the 24-hour rhythms that govern sleep, hormone release, and many other bodily functions. But which exact cells in the core listen to signals from AVP cells in the shell has been unclear.
Following the wiring from sender to receiver cells
The researchers first mapped how shell AVP cells physically connect to VIP and GRP cells in the core. They used a virus to label the outgoing fibers and possible contact points of AVP neurons in mice, then stained brain slices to see where these fibers ended. They found that AVP fibers formed apparent contact sites on both VIP and GRP cells, but in different proportions: roughly a quarter of VIP cells and more than half of GRP cells showed these appositions. This showed that AVP neurons reach both types of core cells, but did not yet reveal which ones actually respond to the signal. To test this, the team used optogenetics—activating AVP neurons with blue light—and looked for c-Fos, a marker of cellular activation, in VIP and GRP neurons.
A small VIP subgroup that truly “hears” AVP
When AVP neurons were stimulated by light, many AVP cells themselves turned on c-Fos, as expected. Importantly, a small subset of VIP cells in the core also showed c-Fos activation, even though they did not directly express the light-sensitive protein. This indicated they were being driven indirectly by AVP input. In contrast, GRP cells rarely showed c-Fos, despite receiving AVP contacts. The authors then looked for the gene message of the AVP receptor known as V1a in these populations using sensitive RNA imaging methods. They discovered that V1a was present in only a minority of VIP cells and was barely detectable in GRP cells. Thus, among the many VIP neurons, a small V1a-positive subgroup appears specially tuned to receive and respond to AVP signals, while most GRP neurons are not. 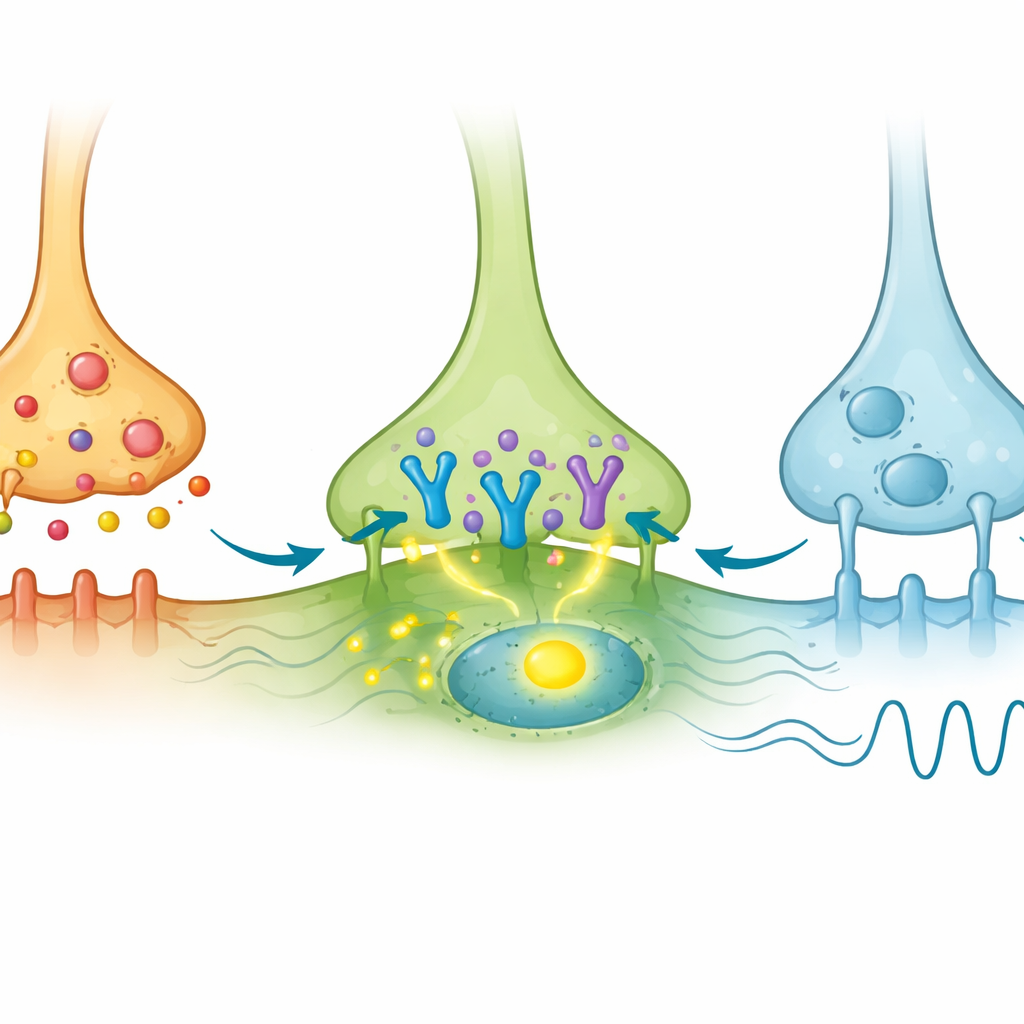
How a tiny cell group shapes jet-lag recovery
To probe the function of these V1a-bearing VIP neurons, the researchers created mice in which the V1a receptor was removed only from VIP cells. These animals kept a normal daily rhythm length in constant darkness, showing that this receptor in VIP neurons is not needed to set the basic pace of the clock. However, when the light–dark schedule was suddenly advanced or delayed by eight hours to mimic jet lag, the modified mice adjusted their activity patterns faster than their normal littermates, especially after advances and in male mice. In other experiments, deleting V1a throughout the brain or specifically in the SCN had produced similar “jet-lag resistant” behavior. The new results therefore suggest that a surprisingly small group of VIP neurons carrying V1a receptors can have an outsized influence on how rigid or flexible the overall clock is.
What this means for our sense of time
In everyday terms, this study shows that within the brain’s central clock, AVP-producing neurons act like timekeepers in an upper tier, sending signals through V1a receptors to a select set of VIP neurons in the lower tier. Those few VIP cells help the whole network resist sudden changes in the light–dark cycle, slowing how quickly the clock resets after a shift. Removing their AVP-sensitive receptor makes the system more willing to shift, easing jet-lag-like adjustments in mice. By charting this hidden diversity among clock neurons, the work offers a more detailed wiring diagram of how our internal sense of time is stabilized—and hints that selectively tweaking similar pathways in humans might one day help manage jet lag or other rhythm disorders.
Citation: Zhou, H., Moriyasu, D., Hsiao, SW. et al. Heterogeneity between VIP and GRP neurons underlies AVP receptor signaling in the mouse suprachiasmatic nucleus. Commun Biol 9, 414 (2026). https://doi.org/10.1038/s42003-026-09694-9
Keywords: circadian clock, suprachiasmatic nucleus, vasopressin signaling, VIP neurons, jet lag