Clear Sky Science · en
Pathological characteristics of SARS-CoV-2 variants and immune responses induced in a COVID-19 macaque model
Why this study matters to everyday life
The coronavirus that causes COVID-19 has kept changing, giving rise to waves driven by Alpha, Delta, Omicron, and other variants. Many people have caught COVID-19 more than once, even after vaccination or previous infection. This study used monkeys with COVID-19–like illness to ask two questions that matter directly to humans: how do different variants behave inside the body, and how does an earlier encounter with the virus shape the immune response to later variants?
Following the variants through the body
Researchers infected cynomolgus macaques, a type of monkey whose COVID-19 symptoms resemble mild human disease, with the original Wuhan strain and six major variants: Alpha, Beta, Gamma, Delta, and Omicron BA.1 and BA.2. All animals received the same dose directly into the windpipe so the infections could be compared fairly. The monkeys showed only mild, temporary signs of illness such as short fevers and changes in appetite, and their weight remained stable. A blood marker of inflammation, C-reactive protein, rose after infection but tended to be lower with Omicron, hinting that these newer variants trigger less whole-body inflammation than earlier ones.
How fast the virus grows and how hard it hits the lungs
The team repeatedly swabbed the monkeys’ noses and throats to track how much virus was being shed and for how long. Delta stood out: it reached the highest viral levels and lingered longest, especially in throat samples that reflect virus growing in the lower airways. Omicron BA.1 and BA.2, in contrast, were barely detectable in nasal swabs and showed much lower overall replication, supporting the idea that Omicron is less suited to infect deep lung tissue. CT scans of the chest confirmed this pattern. Earlier variants, particularly Gamma and Delta, often produced clear patches of pneumonia, while Omicron infections caused fainter, more limited lung changes, consistent with the milder disease typically seen in people.
Second infections and surprising pneumonia
To mimic real-world reinfections, some monkeys were exposed again to either the same or a different variant nearly three months after their first infection. When Delta was used the second time, all animals briefly tested positive again by PCR, showing that reinfection could occur, but none developed pneumonia; prior immunity appeared to restrain the damage even if it did not fully block the virus. Omicron behaved differently. Only about half the monkeys became PCR-positive when re-exposed to Omicron, yet every animal that developed pneumonia after the second round had received an Omicron strain for that second exposure. In these cases, little or no live virus could be found in tissues, suggesting that the lung inflammation was shaped not just by the amount of virus present, but also by how the immune system had been primed.
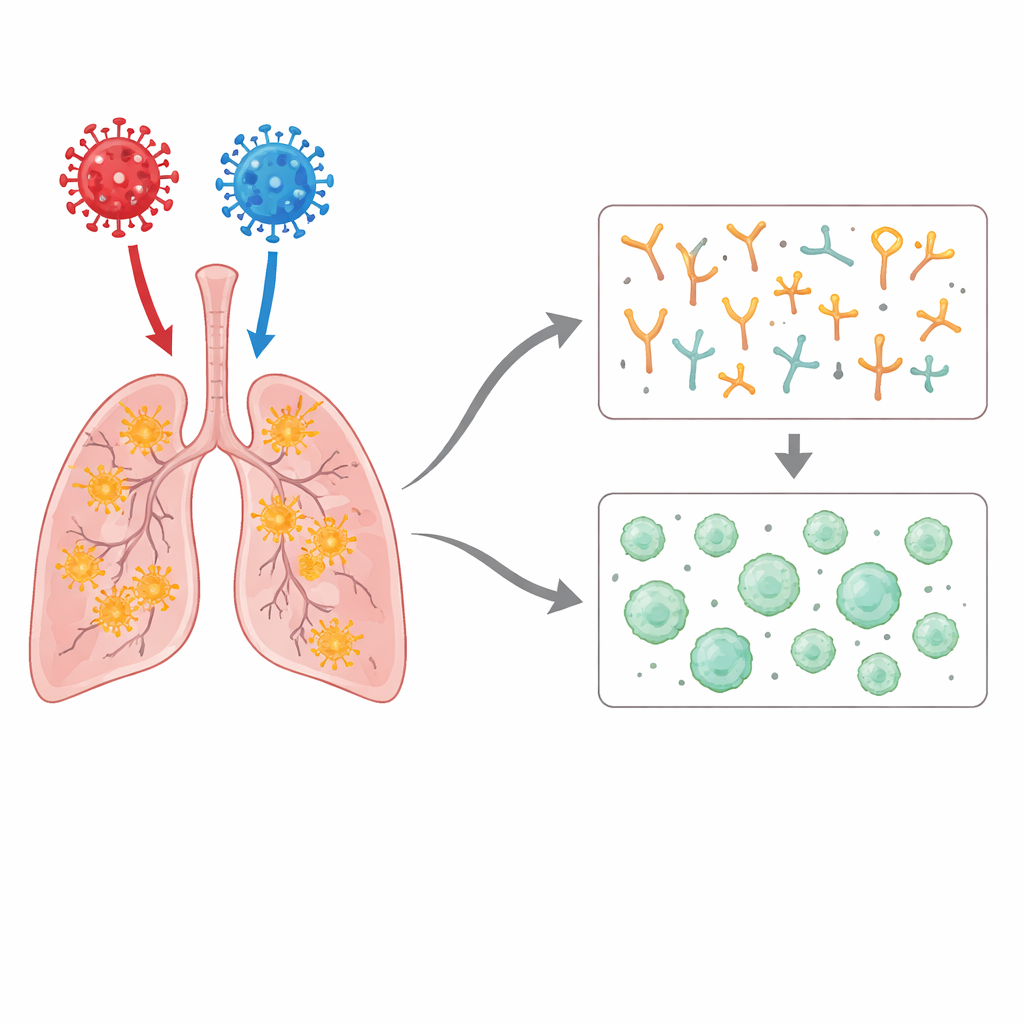
Immune memory and the pull of the first strain
The scientists closely examined antibodies and T cells, the two main arms of adaptive immunity. After infection with Wuhan or Delta, the monkeys produced strong antibodies that recognized the original strain and several early variants. However, even when Omicron itself caused the infection, antibodies that bound tightly to Omicron’s key surface region were weak and slow to appear. Instead, the immune system repeatedly favored responses against the original Wuhan-like spike, a pattern known as “original antigenic sin,” where the first version of a virus that the body encounters leaves a lasting imprint on later responses. T cell reactions to Omicron were also generally weaker than those to earlier variants, especially after only one Omicron infection, reinforcing the picture of a variant that spreads well despite being less inflammatory and less visible to the immune system.
What this means for future vaccines and protection
Put simply, the study shows that not all COVID-19 variants are equal inside the body. Delta grows vigorously and can cause strong lung disease but also provokes robust, cross-reactive immunity. Omicron grows poorly in the lungs and is milder, yet it is also a poor teacher for the immune system, especially when earlier exposure to Wuhan-like viruses keeps pulling antibody responses back toward the original strain. In the long term, this imprinting and Omicron’s low immunogenicity may help explain why Omicron waves and their subvariants have persisted. These findings, drawn from a carefully controlled monkey model, support vaccine strategies that focus on the most current epidemic strain while recognizing that much of the world’s immune memory still points back to the first version of the virus.
Citation: Urano, E., Okamura, T., Higuchi, M. et al. Pathological characteristics of SARS-CoV-2 variants and immune responses induced in a COVID-19 macaque model. Commun Biol 9, 426 (2026). https://doi.org/10.1038/s42003-026-09684-x
Keywords: SARS-CoV-2 variants, Omicron immunity, COVID-19 reinfection, original antigenic sin, macaque model