Clear Sky Science · en
Lysine malonylation regulates human sperm motility
Why the Motion of Sperm Matters
For many couples facing unexplained infertility, a key hidden problem is that sperm simply do not swim well enough to reach and fertilize the egg. This study explores a subtle chemical "tag" on sperm proteins—called lysine malonylation—that seems to act like an invisible brake on their movement. By uncovering how this tag alters the sperm’s energy supply and internal signals, the work points to new ways to diagnose and potentially treat a common form of male infertility linked to poor sperm motility.
A New Chemical Tag on Sperm Proteins
Proteins in our cells are often modified after they are made, gaining small chemical groups that can switch their activity on or off. One such modification, lysine malonylation, was only discovered in 2011 and has been tied to energy metabolism in many cell types. The authors previously showed that human sperm carry many malonylated proteins, but it was unclear what this meant for fertility. In the new study, they mapped where this tag appears in sperm and found it is concentrated in the tail—the long whip-like structure whose rhythmic beating powers forward movement. Using both biochemical tests and high-resolution microscopy, they showed that malonylated proteins are especially abundant in the tail’s mitochondria and surrounding fluid, key sites for energy production and motion control.
Who Adds and Removes the Brake
The team next asked which molecules install and remove this malonyl tag in human sperm. They found evidence that an enzyme known as P300 acts as a "writer," helping transfer malonyl groups onto lysine residues, while another enzyme, SIRT5, serves as an "eraser" that strips them off. When they blocked SIRT5 with a chemical inhibitor, overall malonylation increased; when they inhibited P300, malonylation decreased. They also showed that sodium malonate, a small molecule taken up by sperm and converted to malonyl-CoA, boosted malonylation without disturbing other similar chemical marks. Together, these results outline a regulatory system in which malonyl-CoA supplies the tag, P300 attaches it, and SIRT5 removes it—fine-tuning sperm tail proteins that control movement.
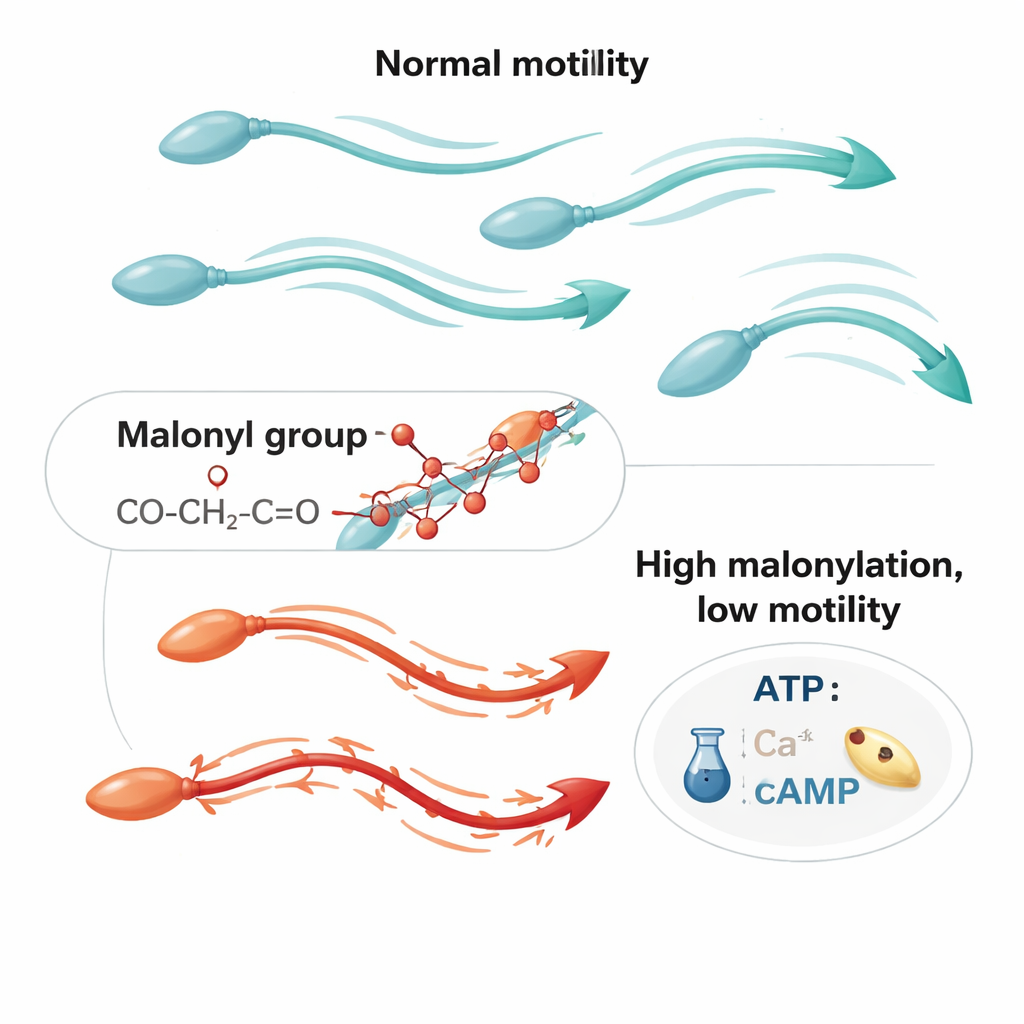
Higher Malonylation in Poorly Moving Sperm
To see whether this chemistry relates to real-world infertility, the researchers compared sperm from men with normal semen profiles to those diagnosed with asthenozoospermia, a condition defined by weak progressive motility. Sperm from the asthenozoospermic group had significantly higher levels of lysine malonylation and lower levels of SIRT5. Across all samples, more malonylation strongly correlated with worse forward swimming and with reduced cellular ATP, the main energy currency. In a subset of men with particularly high malonylation, sperm showed markedly weaker glycolysis—the sugar-burning pathway that provides much of the fuel for human sperm movement. These patterns suggest that excessive malonylation is linked to both impaired energy production and poorer swimming performance.
Experimentally Turning Up the Brake
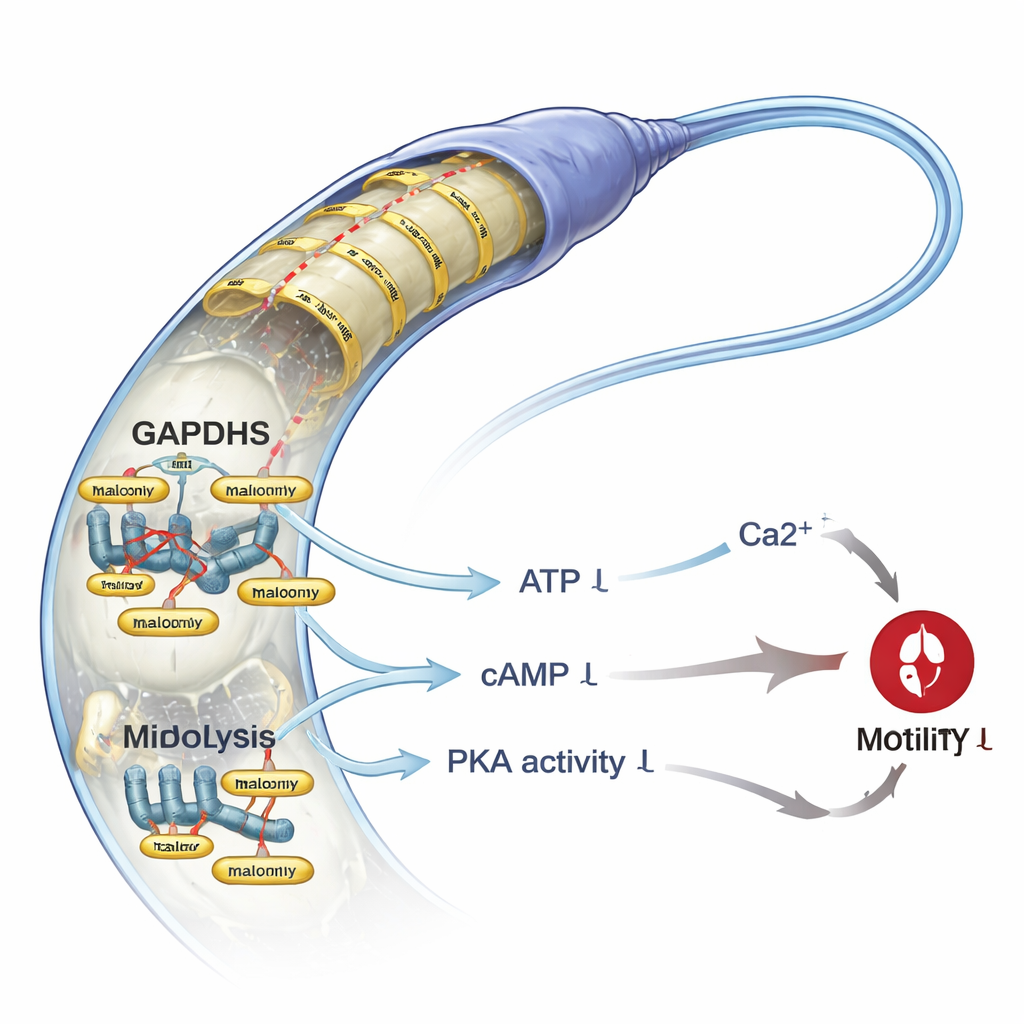
The scientists then asked what happens if they deliberately raise malonylation in otherwise healthy sperm. Treating samples from men with normal semen parameters with sodium malonate increased malonylation levels but did not kill the cells. However, it did significantly reduce total and progressive motility and made it harder for sperm to push through a viscous medium that mimics the female reproductive tract. Mechanistic tests revealed why: sodium malonate-treated sperm had lower glycolytic output, less ATP, and reduced amounts of cAMP, a messenger that activates the key enzyme PKA. PKA activity dropped, as did the phosphorylation of downstream proteins known to support motility. At the same time, the sperm’s internal calcium levels fell by about half, even though the main sperm calcium channel, CatSper, was not directly affected. This combination—less energy, weaker signaling, and diminished calcium—offers a coherent explanation for the observed loss of motility.
From Molecular Tags to Male Fertility
Putting all the findings together, the study proposes that lysine malonylation acts as a negative regulator of human sperm motility. When malonylation levels climb—because SIRT5 is low, malonyl-CoA is high, or related pathways are disrupted—key proteins that drive glycolysis and calcium handling, such as GAPDHS and VDAC3, become excessively tagged. This dampens energy production and critical signaling cascades in the sperm tail, leading to sluggish movement and poorer ability to penetrate thick fluids. For a lay reader, the message is that sperm do not just need enough numbers; they also need finely tuned chemical control systems to power their swim. Disruptions in a small, reversible tag like malonylation may contribute to otherwise unexplained male infertility and could eventually offer new biomarkers or targets for therapies aimed at restoring sperm vigor.
Citation: Cheng, Y., Tian, Y., Chen, H. et al. Lysine malonylation regulates human sperm motility. Commun Biol 9, 178 (2026). https://doi.org/10.1038/s42003-026-09683-y
Keywords: sperm motility, male infertility, post-translational modification, lysine malonylation, energy metabolism