Clear Sky Science · en
African swine fever virus–encoded protein MGF 505–3R impairs innate immunity via ubiquitin–mediated degradation of MyD88
Why this matters for animal health and beyond
African swine fever has devastated pig farms around the world, threatening food supplies and causing huge economic losses. The virus behind it is deadly in part because it slips past the body’s early warning systems. This study uncovers how one viral protein, called MGF 505–3R, quietly sabotages a key alarm switch inside cells and shows that a tiny fragment of this protein can also be turned into a powerful anti-inflammatory tool in mice.
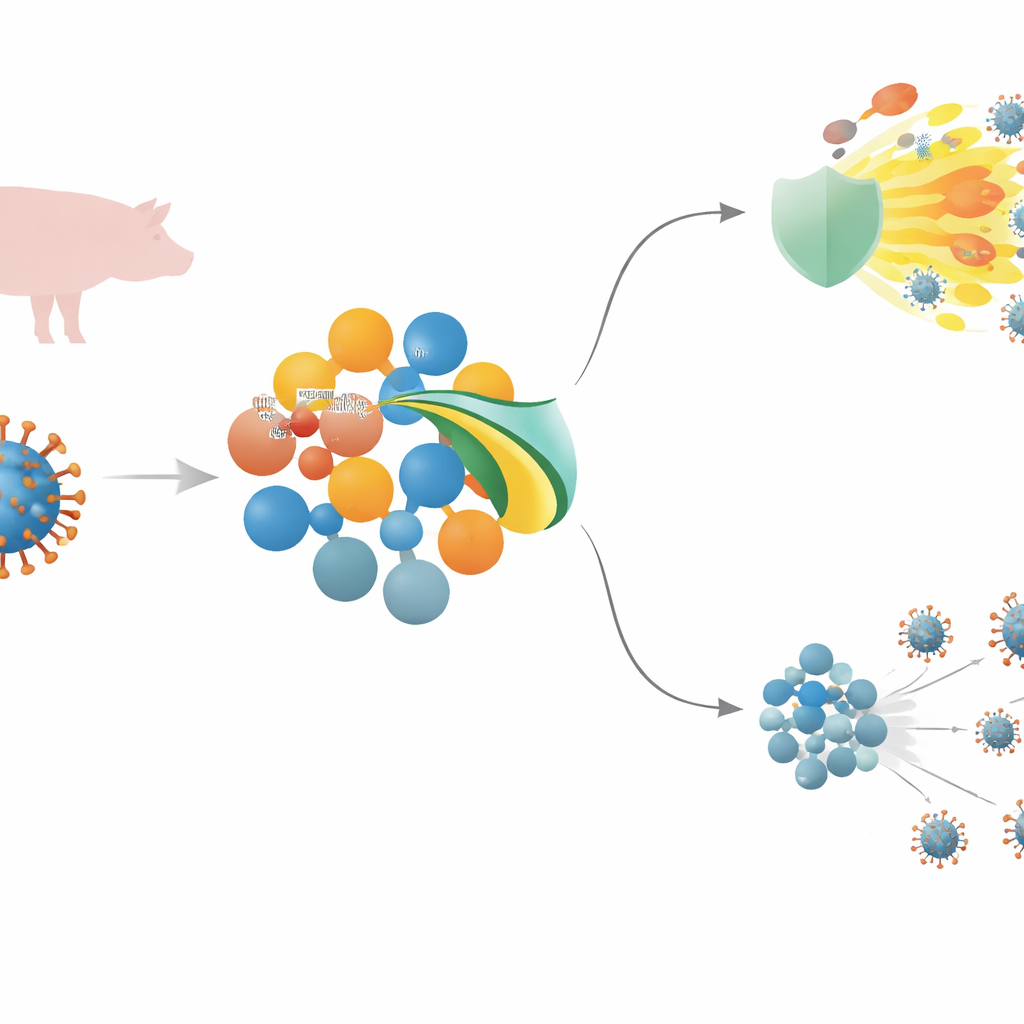
A deadly pig virus and the body’s first line of defense
African swine fever virus (ASFV) is a large DNA virus that can kill nearly every infected pig. To gain a foothold, it must outsmart innate immunity—the rapid, frontline defense that detects invaders and sparks inflammation and antiviral molecules. Central to this response is a signaling chain that includes sensors on the cell surface, a relay protein called MyD88, and a master switch known as NF–κB, which together drive the production of inflammatory messengers and antiviral interferons. ASFV carries many genes believed to interfere with these signals, but for several of them, including MGF 505–3R, their exact tricks were not well understood.
How the virus cuts the wires of the immune alarm
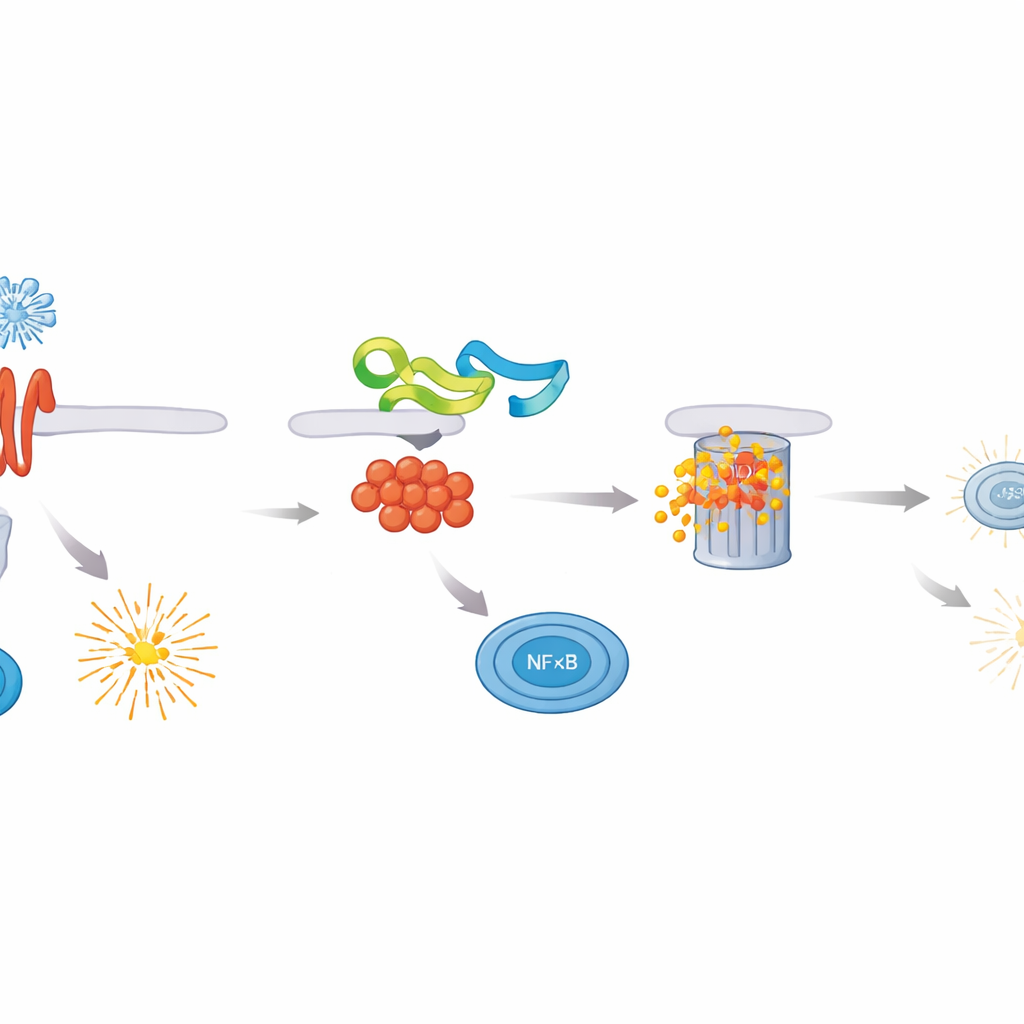
The researchers screened ASFV proteins for their ability to dampen NF–κB activity and found that MGF 505–3R was a particularly strong inhibitor. When this viral protein was present, cells exposed to different immune triggers produced far fewer inflammatory cytokines and much lower levels of type I and type III interferons. Closer inspection showed that MGF 505–3R acts directly on MyD88, the central adaptor that connects many immune sensors to NF–κB. MGF 505–3R binds MyD88 and marks it for disposal via the cell’s protein-shredding machinery, using a form of tagging called K48-linked ubiquitination. As MyD88 is degraded, NF–κB fails to move into the nucleus and turn on protective genes, leaving cells more permissive to virus growth.
Zooming in on a tiny but powerful protein fragment
To pinpoint the crucial part of MGF 505–3R, the team made shortened versions of the protein and tested their effects. They discovered that amino acids 89–277 were both necessary and sufficient to bind MyD88, promote its ubiquitination, and block NF–κB activation. Guided by computer predictions of protein structure, they carved out two short peptides from this region. One of them, pep3R–1, stood out: it sharply reduced NF–κB activity, blocked phosphorylation and nuclear movement of the NF–κB subunit p65, and lowered the expression of inflammatory cytokines and interferons in immune cells stimulated with multiple danger signals. In cell culture, both full-length MGF 505–3R and pep3R–1 not only dimmed antiviral signaling but also allowed a test virus to replicate more efficiently, underscoring how potent this pathway is for controlling infections.
Turning a viral trick into a potential medicine
The team then asked whether the peptide could be harnessed to calm harmful inflammation rather than to help viruses. In a mouse model of colitis triggered by a chemical called DSS, animals normally lose weight, develop bloody diarrhea, and show severe damage and immune cell invasion in the colon. Mice treated with pep3R–1 fared much better: disease scores were lower, colons were longer and healthier, and microscopic examination revealed preserved tissue structure with far fewer inflammatory cells. Levels of key inflammatory molecules such as TNF–α, IL–1β, IL–6, and an enzyme linked to white blood cell influx were all reduced in colon tissue and blood. In a separate experiment, pep3R–1 also lessened the inflammatory response to a systemic bacterial toxin, indicating that it can dampen both local and whole-body inflammatory storms.
What this means for pigs, people, and future treatments
This work shows that ASFV protein MGF 505–3R helps the virus thrive by dismantling a central immune relay, MyD88, thereby shutting down both inflammatory and interferon defenses. At the same time, a small fragment of this viral protein, pep3R–1, can be repurposed to safely cool excessive inflammation in mice, including in a gut disease model. While much remains to be done—such as improving stability, delivery, and testing in more disease settings—the study reveals both a critical weak point that antiviral strategies could target and a promising blueprint for designing new peptide-based anti-inflammatory drugs inspired by viral immune evasion.
Citation: Liu, H., Sun, L., Wang, F. et al. African swine fever virus–encoded protein MGF 505–3R impairs innate immunity via ubiquitin–mediated degradation of MyD88. Commun Biol 9, 407 (2026). https://doi.org/10.1038/s42003-026-09681-0
Keywords: African swine fever, innate immunity, NF-kappaB, MyD88, anti-inflammatory peptide