Clear Sky Science · en
Abnormal iron homeostasis mediates cochlear hair cell impairment and hearing loss in Gprasp2-deficient mice
Why this matters for everyday hearing
Hearing loss is often thought of as simple "wear and tear," but for many people it is driven by hidden defects in their genes. This study uncovers how a rarely studied gene, GPRASP2, helps protect the delicate sound-sensing cells of the inner ear from iron-driven damage. By showing exactly how things go wrong when this gene is missing, the work opens doors to more precise diagnoses and future therapies for inherited hearing problems and possibly related mood disorders.
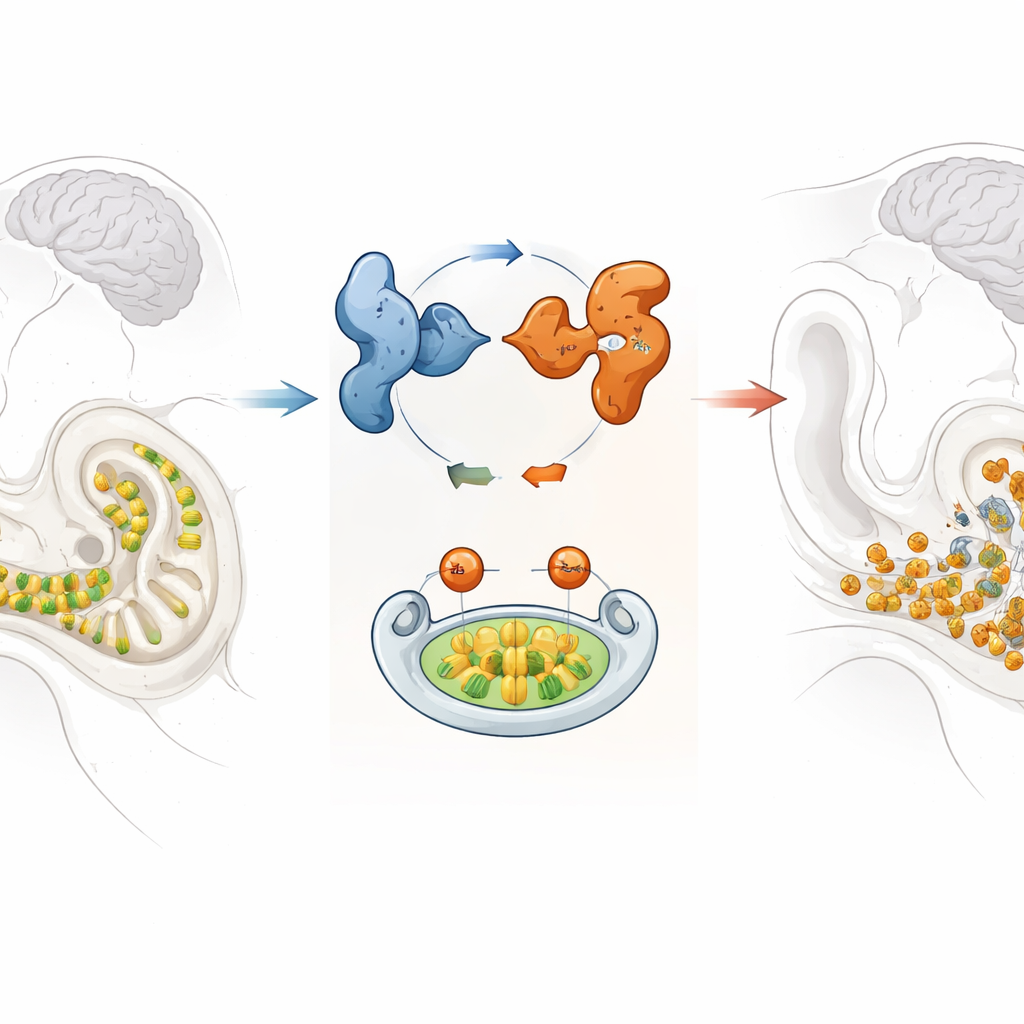
A fragile sound sensor in the inner ear
Inside the coiled cochlea of the inner ear sit rows of hair cells that convert tiny vibrations into electrical signals the brain can understand. Once these hair cells are lost, they do not grow back, so keeping them healthy is critical for lifelong hearing. The researchers focused on GPRASP2, a gene previously found mutated in a family with X-linked syndromic hearing loss, meaning the condition mainly affects males and can come with additional symptoms. Although GPRASP2 was known to be active in the brain and inner ear, its exact role in hearing remained unclear.
What happens when the gene is missing
To mimic the human condition, the team used CRISPR gene editing to create mice that lack a working copy of the Gprasp2 gene. These animals showed clear hearing loss across a wide range of sound frequencies when tested with sensitive electrical recordings from the auditory nerve. Their responses to sudden loud sounds were blunted, yet their balance and motor skills were mostly normal, pointing to a specific problem in hearing rather than a general movement disorder. Interestingly, the mice also showed depression-like behaviors in several standard tests, hinting that this gene links hearing and mood through its actions in both ear and brain.
Damage inside the cochlea
When the scientists examined the inner ears of Gprasp2-deficient mice, they found that many outer hair cells were missing or misplaced, and the remaining cells often had distorted bundles of tiny projections that normally act like tuning forks. They also saw changes in the stria vascularis, a highly active tissue that maintains the special fluid and electrical environment required for hearing. At the microscopic level, markers of cell death and oxidative stress were elevated in the outer hair cells and nearby nerve cells, indicating that these structures were under heavy attack from reactive molecules and were more likely to die.
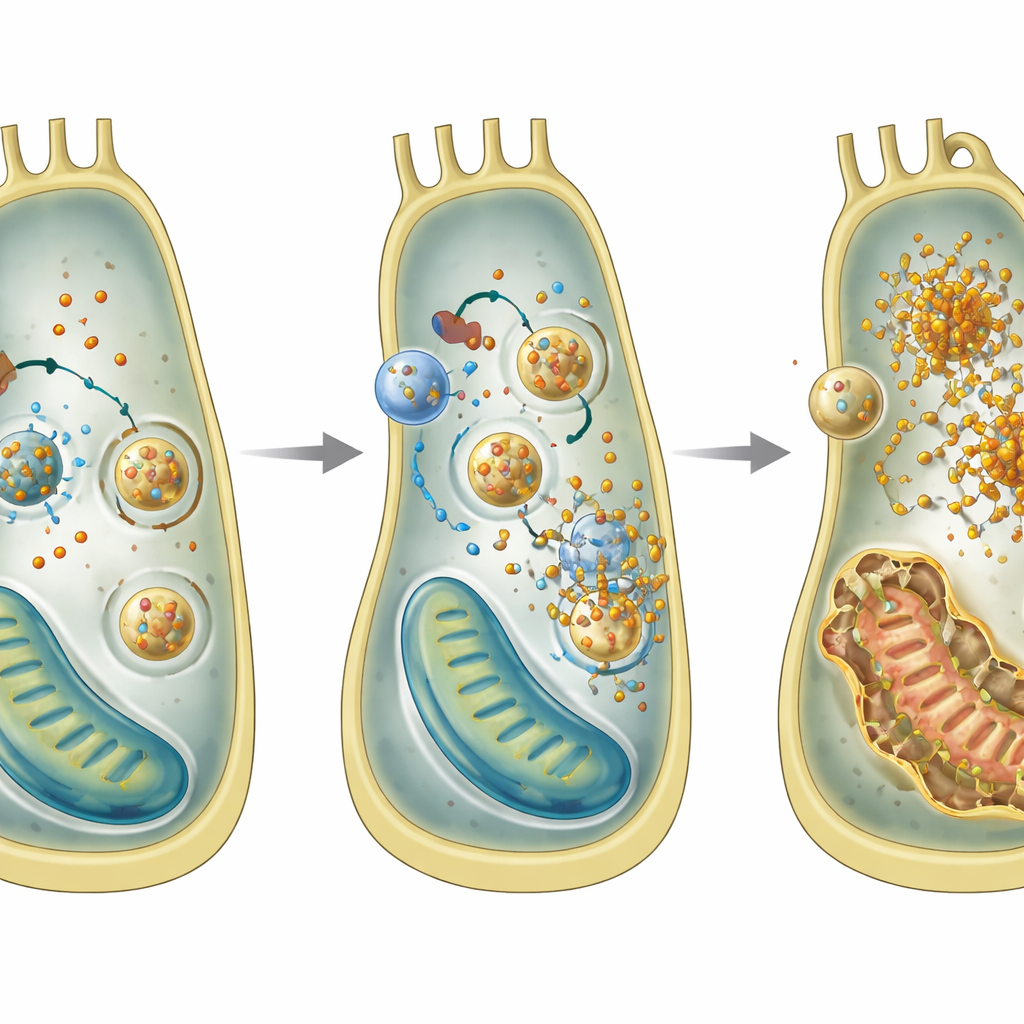
Iron overload and runaway recycling
Diving deeper, the team used an inner-ear cell line to explore what goes wrong inside individual cells without GPRASP2. They discovered a pattern characteristic of a form of cell death called ferroptosis, which is driven by iron and lipid peroxidation. Cells lacking Gprasp2 accumulated more ferrous iron, produced more reactive oxygen species, had lower levels of the antioxidant glutathione, and showed damaged mitochondria. Gene and protein analyses pointed to an increase in ferritinophagy, a process where iron-storage particles are delivered to recycling compartments and broken down, releasing extra iron into the cell. Blocking this recycling step reduced iron buildup, supporting the idea that excessive ferritinophagy is central to the damage.
A key partner protein that keeps iron in check
The researchers then asked how GPRASP2 controls this iron-handling machinery. By mapping proteins that interact with GPRASP2, they identified NCAM1, a cell-adhesion molecule best known for its roles in learning, memory, and mood. They showed that GPRASP2 physically binds NCAM1, and that loss of GPRASP2 causes NCAM1 levels to drop in hair cells and in cultured auditory cells. Lower NCAM1 was linked to increased ferritinophagy and iron overload. Restoring NCAM1 in Gprasp2-deficient cells reduced iron levels and dampened key ferritinophagy regulators, even though general autophagy remained active. This suggests GPRASP2 fine-tunes a specific iron-recycling route through NCAM1, rather than turning the whole recycling system on or off.
What this means for hearing and beyond
In simple terms, this work shows that GPRASP2 acts like a safety manager for iron inside cochlear hair cells. When GPRASP2 is present, it cooperates with NCAM1 to prevent too much stored iron from being dumped back into the cell, keeping oxidative damage in check. When the gene is disrupted, iron handling goes off balance, the cells rust from within, and crucial hair cells die, leading to hearing loss. Because GPRASP2 and NCAM1 also function in the brain, the same pathway may help explain mood changes seen in some patients. Understanding this iron-based failure mode offers a clear target for future drugs or gene therapies aimed at preserving hearing in people with GPRASP2-related or similar genetic disorders.
Citation: Lu, Y., Sheng, F., Yao, J. et al. Abnormal iron homeostasis mediates cochlear hair cell impairment and hearing loss in Gprasp2-deficient mice. Commun Biol 9, 425 (2026). https://doi.org/10.1038/s42003-026-09679-8
Keywords: hereditary hearing loss, cochlear hair cells, iron homeostasis, ferroptosis, GPRASP2