Clear Sky Science · en
Single-cell transcriptomics reveal mechanisms of skeletal muscle differentiation across duck embryonic development
Why duck muscle development matters
Skeletal muscle is what lets animals move, fly, and, in the case of farm species, also determines how their meat feels and tastes. This study focuses on how the breast muscle of Pekin ducks forms before hatching, using powerful single-cell tools to watch tens of thousands of individual cells as they grow and specialize. By following each cell’s journey from an early stem-like state to a fully formed muscle fiber, the researchers uncover how different muscle fiber types arise, how they can switch identity, and how these rules are shared across birds and mammals.
Building muscle from the earliest cells
Duck muscle begins as a mix of highly flexible stem-like cells early in the embryo. The team created a detailed “cell atlas” by sequencing the RNA of nearly 77,000 single cells from duck embryos at ten time points, from very early development through hatching. They found two main stem cell pools that dominate the earliest stages and gradually give rise to many supporting and muscle-forming cell types. Among these, a subset of mesenchymal stem cells marked by a molecule called MYL9 appears to be the main source of future muscle progenitor cells. Over time, these progenitors become myoblasts that fuse into larger structures, eventually yielding the long, multinucleated fibers that make up functional muscle.
Two key branches: fibers that work and cells that repair
When the researchers followed muscle-lineage cells over developmental “pseudotime,” they saw that early progenitors split into two major branches. One branch produces the mature muscle fibers needed for contraction. The other forms satellite cells, the long-lived “repair crew” that stays mostly quiet until they are needed for growth or regeneration later in life. Along the satellite-cell branch, specific genes turn on and off in a coordinated way, shifting cells from a resting state into active, dividing cells. The analysis points to a handful of control genes that likely act as switches for this activation process. In the fiber-making branch, the study highlights cellular processes such as membrane traffic and cell–cell adhesion that are crucial for myoblasts to fuse and build robust muscle fibers.
How slow fibers become fast fibers
One of the most striking findings is that muscle fibers do not simply form as either “slow” or “fast” from the outset. Instead, early in duck development, slow-twitch fibers—better at steady, endurance-like work—are common, while fast-twitch fibers that power quick, strong movements are rare. As the embryo matures, the balance flips. By tracking gene activity within individual fibers, the researchers discovered a stepwise “slow-to-fast” transition. Slow fibers move through an intermediate state, including a newly described subtype marked by the factor LEF1, and then acquire fast-fiber features. Along the way, some fibers briefly display a hybrid identity, showing traits of both slow and fast types, hinting at a flexible window when their fate can still change.
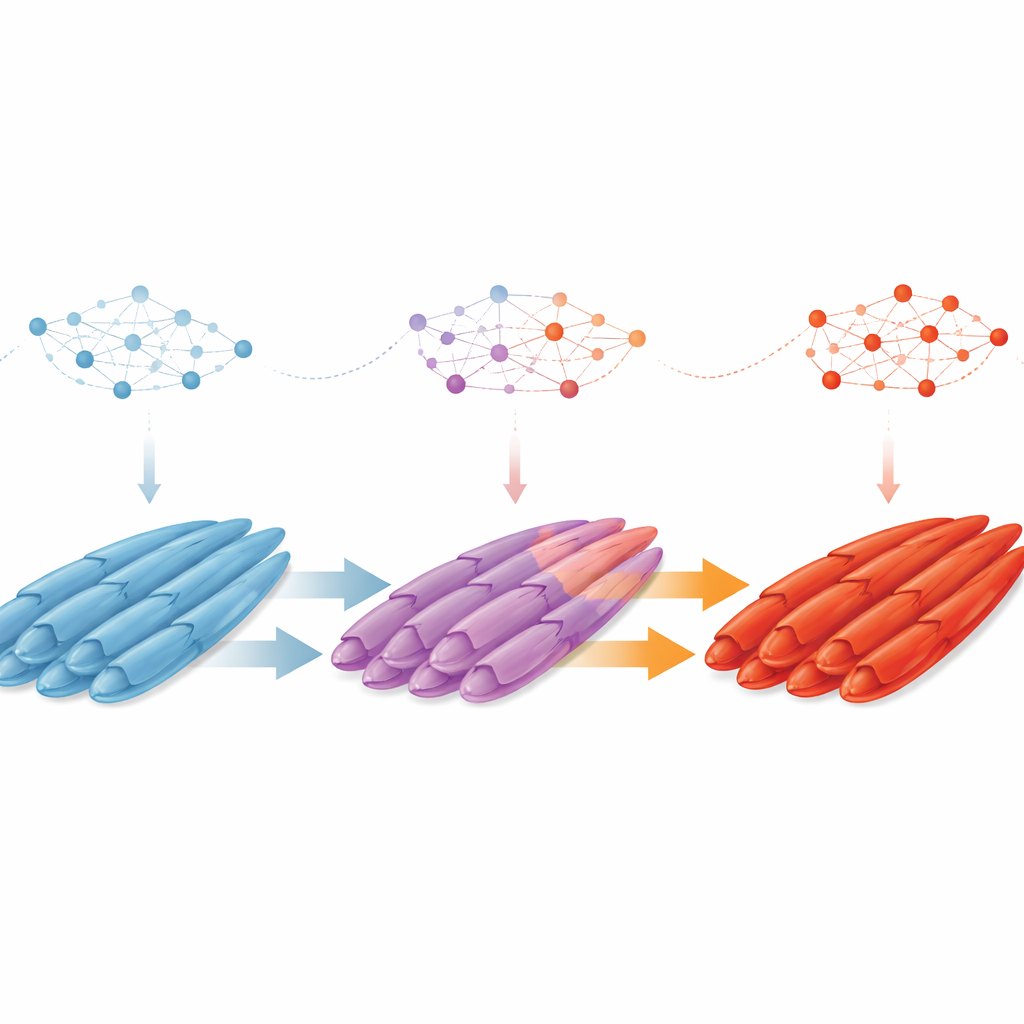
Control genes and shared rules across species
The team then asked what regulates this shift in fiber identity. By reconstructing gene networks, they identified 13 key transcription factors—master regulators that coordinate groups of genes—guiding cells from early progenitors through myoblasts, maturing fibers, and satellite cells. Two factors, TBX15 and PBX3, stand out as prime candidates for steering fibers from slow to fast behavior, working through well-known growth and survival pathways such as PI3K–Akt and receptor tyrosine kinase signaling. Finally, by comparing duck data with single-cell maps from pigs, chickens, and mice, the authors show that many cell types, marker genes, and even the overall pattern of slow-to-fast fiber transition are conserved across birds and mammals. This suggests that deep, shared genetic programs shape how vertebrate muscles specialize.
What this means for biology and beyond
For non-specialists, the main takeaway is that muscle fibers are not fixed from birth: in ducks, and likely in many vertebrates, early slow fibers can transform into fast fibers through a well-organized sequence of intermediate states controlled by specific genes and signaling pathways. Understanding this roadmap helps explain how animals fine-tune their muscles for flight, running, or other tasks, and why fiber makeup influences meat quality. In the long term, the same principles could inform strategies to improve livestock muscle traits or to design regenerative therapies that rebuild or reshape human muscle after injury or disease.
Citation: Sun, Y., Li, Z., Jie, Y. et al. Single-cell transcriptomics reveal mechanisms of skeletal muscle differentiation across duck embryonic development. Commun Biol 9, 404 (2026). https://doi.org/10.1038/s42003-026-09665-0
Keywords: skeletal muscle development, muscle fiber types, single-cell transcriptomics, duck embryogenesis, slow-to-fast fiber transition