Clear Sky Science · en
Activation of cytomegalovirus-encoded G protein-coupled receptor UL33 by an innate N-terminal peptide
How a Common Virus Rewires Our Cells
Human cytomegalovirus (HCMV) quietly infects most people on Earth and usually stays unnoticed. Yet in newborns, transplant patients, and others with weakened immune systems, it can cause serious disease. This study reveals, in molecular detail, how one viral protein called UL33 acts like a built‑in switch that constantly pushes infected cells to favor the virus. Understanding this hidden switch helps explain how HCMV persists for life and points to new ways to disarm it without harming normal cell signaling.
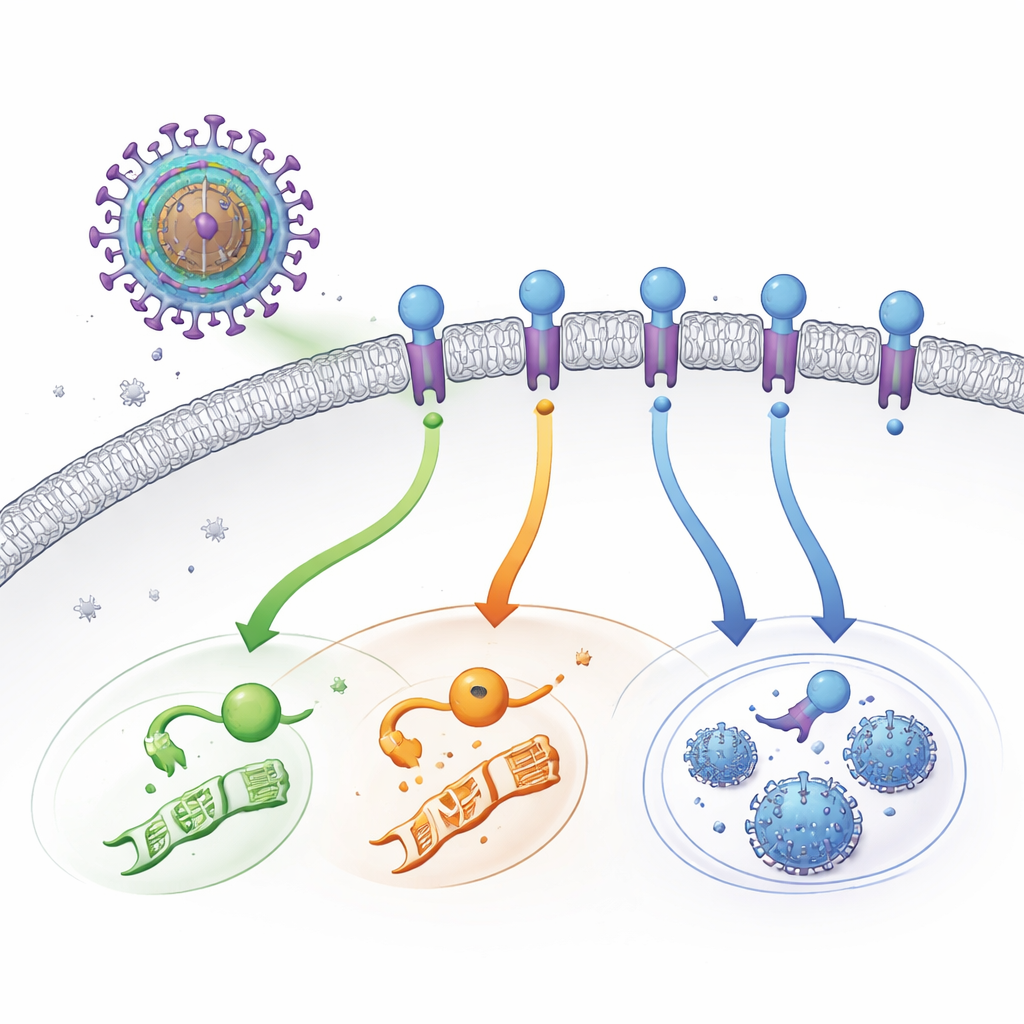
A Stealthy Viral Switch on the Cell Surface
HCMV carries its own versions of cell surface receptors—proteins that normally sense hormones or immune signals. UL33 is one of these viral receptors. Unlike typical receptors, which wait for an external signal, UL33 is “always on.” Once placed in the outer membrane of an infected cell, it can tap into several of the cell’s main communication routes at once. These routes control processes such as gene activity, metabolism, and the decision to lie dormant or to produce more virus. For years, scientists knew UL33 was active but did not know what turned it on, especially because no external activating molecule (ligand) had ever been found.
A Self-Starter: The Receptor That Brings Its Own Key
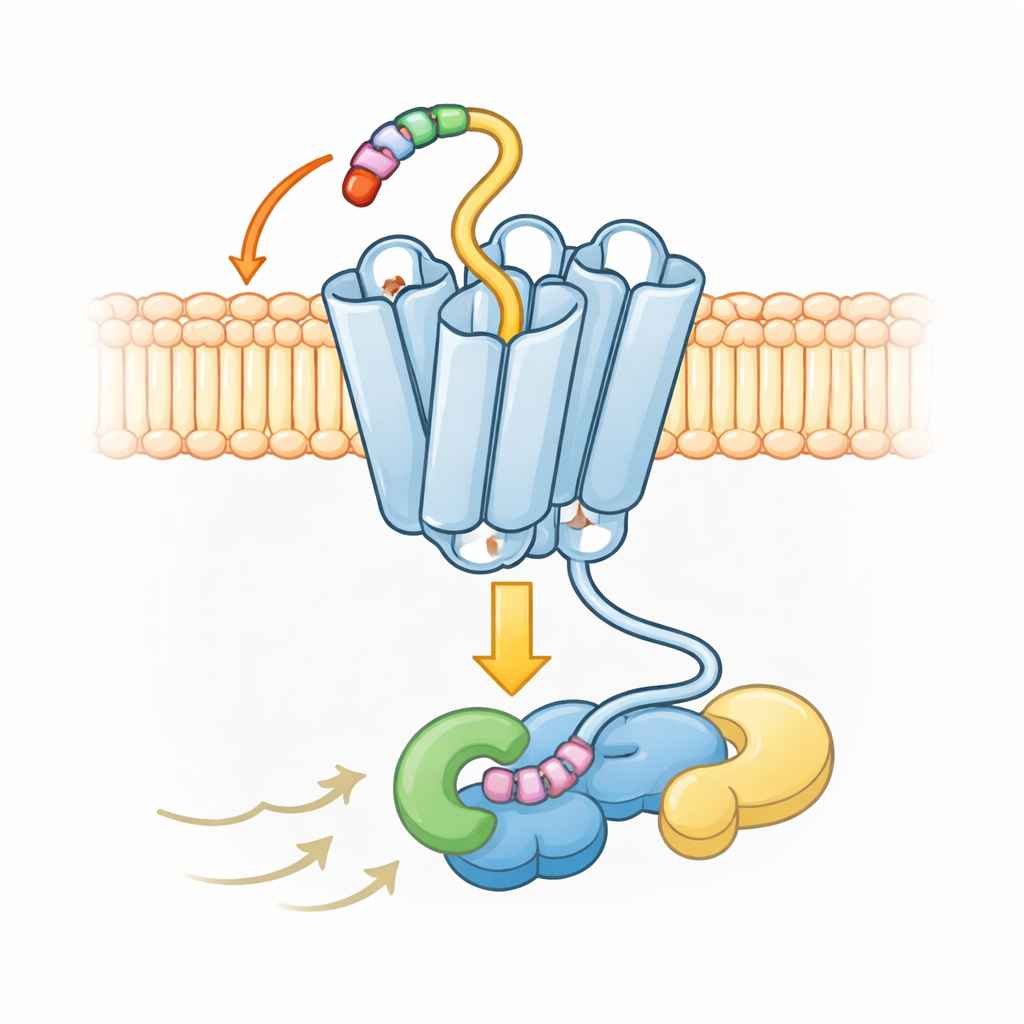
Using advanced cryo‑electron microscopy, the authors captured three‑dimensional snapshots of UL33 bound to one of the cell’s signaling partners, a G protein called Gs. The images show that the very beginning of the UL33 protein—its short N‑terminal tail—folds back into a pocket on the receptor itself, much like a key left permanently in a lock. This “tethered” tail nestles into a small side pocket that, in related human receptors, would normally receive part of an immune signaling protein called a chemokine. By occupying this pocket from the start, UL33 blocks outside chemokines from binding at all, explaining why it behaves as an “orphan” receptor that does not respond to normal host messengers.
Crucial Atoms in the Viral On-Switch
To confirm that this built‑in tail really is the trigger, the team systematically altered individual building blocks of UL33 in human cells. They focused on the first few amino acids of the tail and matching points inside the pocket that grip it. When they swapped these key residues for more neutral ones or removed one of them entirely, UL33’s ability to signal dropped to near zero—even though the altered receptors still reached the cell surface in normal amounts. In other words, the protein was present but mute. This shows that the tail is not just structural decoration; it is the essential self-activating piece that keeps UL33 turned on.
An Unusual Shape for Broad Yet Controlled Signaling
The structural maps also reveal that UL33 adopts a slightly different active shape compared with typical human receptors. In most receptors of this family, one of the seven helices near the inside of the cell swings outward dramatically when the receptor is activated, opening a large cavity for G proteins to dock. In UL33, that helix stays much closer to the center, creating a tighter fit. Despite that, UL33 can still engage several kinds of G proteins—Gs, Gq, and Gi—by making precise contacts with the tip of the G protein that slots into the receptor. These contacts favor some G proteins and exclude others, such as the G12/13 group, allowing the virus to bias the cell’s responses without unleashing uncontrolled signaling.
New Vulnerabilities for an Old Virus
By combining structural imaging with functional tests, the study paints a clear picture: UL33 is a self‑starting viral receptor whose own tail acts as a permanently attached activator. This design lets HCMV gently but persistently tune host cell signaling to support viral replication and reactivation, especially by boosting pathways that turn on viral genes. At the same time, the work exposes weak points that drugs could exploit, such as a narrow tunnel leading into the pocket where the tail binds. Molecules designed to wedge into this region or dislodge the tail might quiet UL33’s activity and reduce viral damage, while sparing the body’s normal receptors. For patients at risk from HCMV, targeting this viral “master switch” could one day offer a more precise way to keep the virus in check.
Citation: Drzazga, A.K., Suzuki, S., Wouters, C. et al. Activation of cytomegalovirus-encoded G protein-coupled receptor UL33 by an innate N-terminal peptide. Commun Biol 9, 415 (2026). https://doi.org/10.1038/s42003-026-09660-5
Keywords: cytomegalovirus, viral GPCR, cell signaling, cryo-EM structure, drug targeting