Clear Sky Science · en
Sleep deprivation disrupts lacrimal gland homeostasis via hypothalamic-pituitary-adrenal axis and gut dysbiosis in mice
Why Lost Sleep Can Make Your Eyes Feel Gritty
Most people know that skimping on sleep can leave them tired and foggy the next day. Fewer realize that chronic sleep loss may also silently damage the glands that keep our eyes comfortable and protected. This study in mice explores how ongoing sleep deprivation dries out the eye’s surface by disturbing stress hormones and gut microbes, and it points to possible ways to guard our vision in a sleepless world. 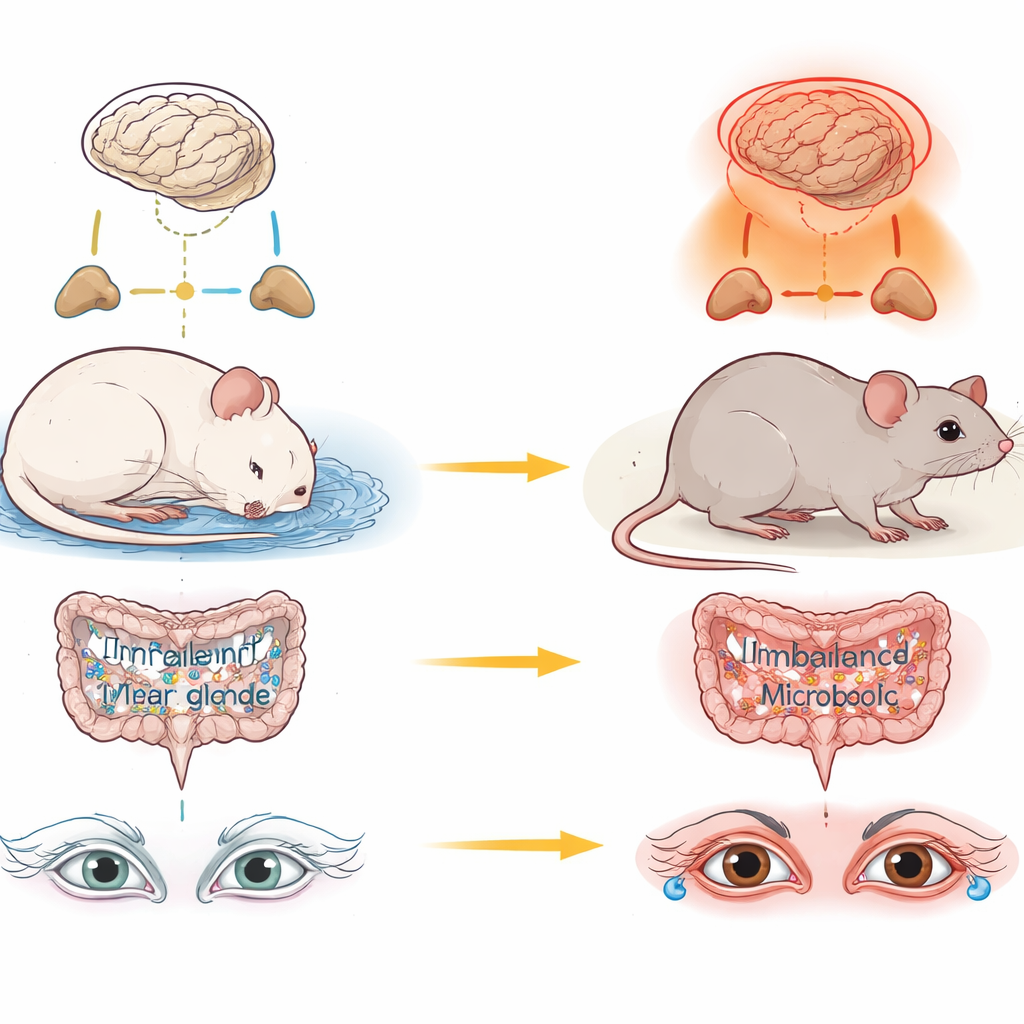
A Hidden Chain from Brain to Gut to Eye
The researchers focused on the lacrimal glands, small structures near the eyes that produce the watery layer of our tears. When the team kept mice awake for weeks using a gentle, automated system, the animals produced fewer tears and their lacrimal glands shrank and lost their normal cellular structure. At the same time, gene activity in these glands shifted strongly toward immune and inflammatory programs, including a powerful pathway driven by a molecule called IL-17. In simple terms, the glands that should calmly bathe the eye in tears instead began to look and act like irritated, inflamed tissue.
Stress Hormones Turn Up the Heat
Sleep loss is a well-known trigger for the body’s main stress circuit, called the hypothalamic–pituitary–adrenal axis, which ends in the release of stress hormones from the adrenal glands. In the sleep-deprived mice, levels of the steroid hormone corticosterone rose, and the adrenal glands themselves showed signs of immune activation. When the scientists gave a drug called metyrapone to block the production of these stress hormones, much of the damage was reduced: lacrimal glands retained more normal size and structure, tear production improved, and inflammatory immune cells were less abundant. This suggested that overactive stress signaling is not just a side effect of lost sleep, but a key driver of eye gland injury.
Gut Microbes Out of Balance
The team then turned to the gut, where trillions of microbes help digest food and train the immune system. Chronic sleep loss made the intestinal wall more leaky and markedly altered the makeup of gut bacteria. Levels of short-chain fatty acids—small molecules produced by friendly microbes that help keep inflammation in check—dropped in both the gut and the bloodstream. When the researchers wiped out gut bacteria with antibiotics, the damage to the lacrimal glands became even worse, hinting that a healthy microbial community normally helps protect the eye. 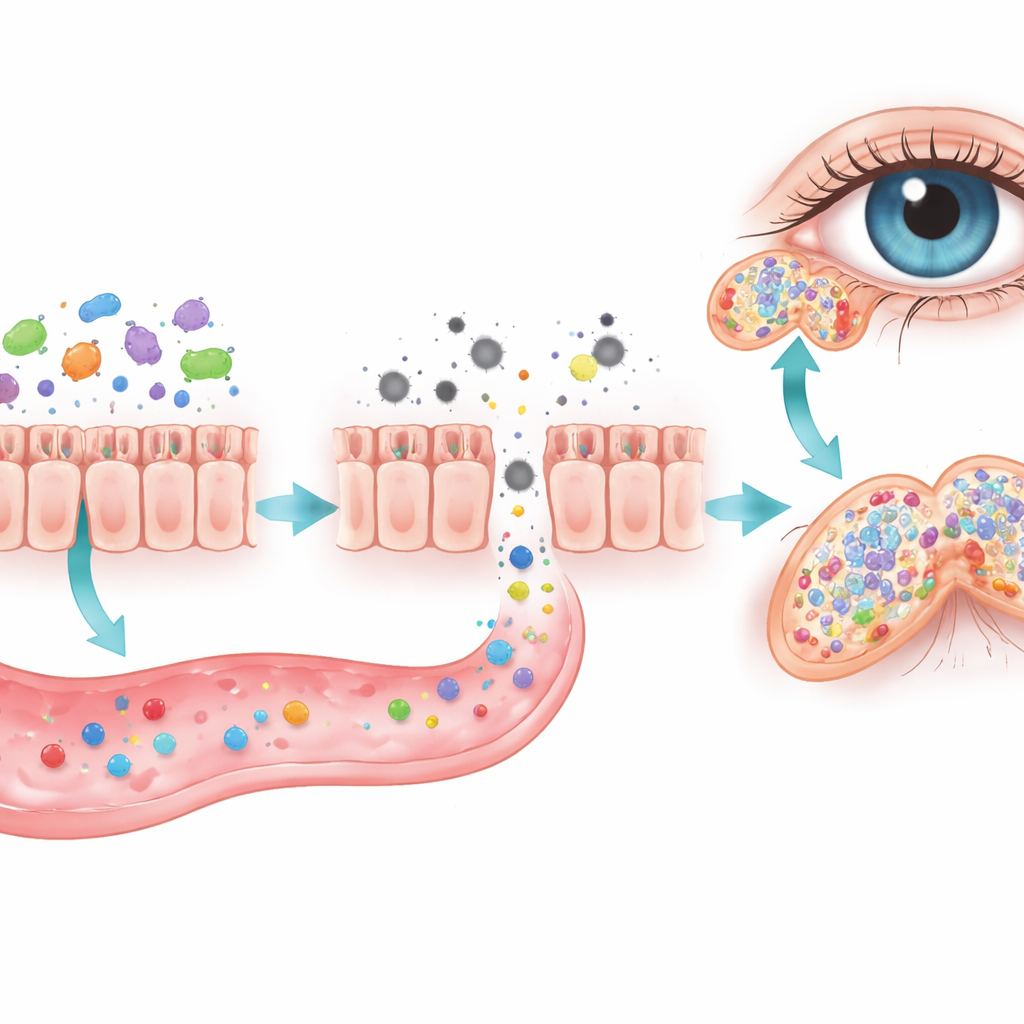
Restoring Microbes and Their Helpful Metabolites
To see whether fixing the gut could rescue the eyes, the scientists tried two strategies. In one, they transplanted gut microbes from healthy mice into sleep-deprived animals. In the other, they added back a cocktail of short-chain fatty acids in the drinking water. Both approaches partially restored normal gut communities, boosted these beneficial metabolites, and eased lacrimal gland injury. Tear production climbed, gland structure improved, and invading immune cells declined. Crucially, all three interventions—stress-hormone blockade, microbe transplantation, and short-chain fatty acid supplementation—converged on lowering IL-17–related inflammation in the glands.
What This Means for Tired Eyes
Taken together, the work outlines an “axis” linking brain-driven stress, gut microbes, and tear glands. In this model, chronic sleep loss overactivates stress hormones, disturbs the gut ecosystem, depletes soothing microbial products, and loosens the intestinal barrier. Signals leaking from the gut then help ignite IL-17–centered inflammation in the lacrimal glands, shrinking them and cutting tear output. While the study was done in male mice, the findings raise the possibility that better sleep, careful control of stress, and future microbiome-based therapies could one day help protect people who suffer from dry eye and other sleep-related eye problems.
Citation: Huang, S., Yu, S., Zhang, W. et al. Sleep deprivation disrupts lacrimal gland homeostasis via hypothalamic-pituitary-adrenal axis and gut dysbiosis in mice. Commun Biol 9, 367 (2026). https://doi.org/10.1038/s42003-026-09657-0
Keywords: sleep deprivation, dry eye, gut microbiome, stress hormones, short-chain fatty acids