Clear Sky Science · en
Investigating Salmonella biofilm responses to antibiotic treatment using optical photothermal infrared spectroscopy
Why Sticky Germ Cities Matter
Many disease-causing bacteria prefer to live in tightly packed communities called biofilms, where cells stick to each other and to surfaces inside the body or on medical devices. Within these slimy “cities,” microbes can shrug off antibiotics that would normally kill them. This study introduces a new way to look inside biofilms in fine detail, revealing which cells are actively growing and how they react when powerful drugs are added. Understanding these invisible activity patterns could help doctors design treatments that finally break biofilms’ stubborn defenses.
Hidden Life Inside Bacterial Cities
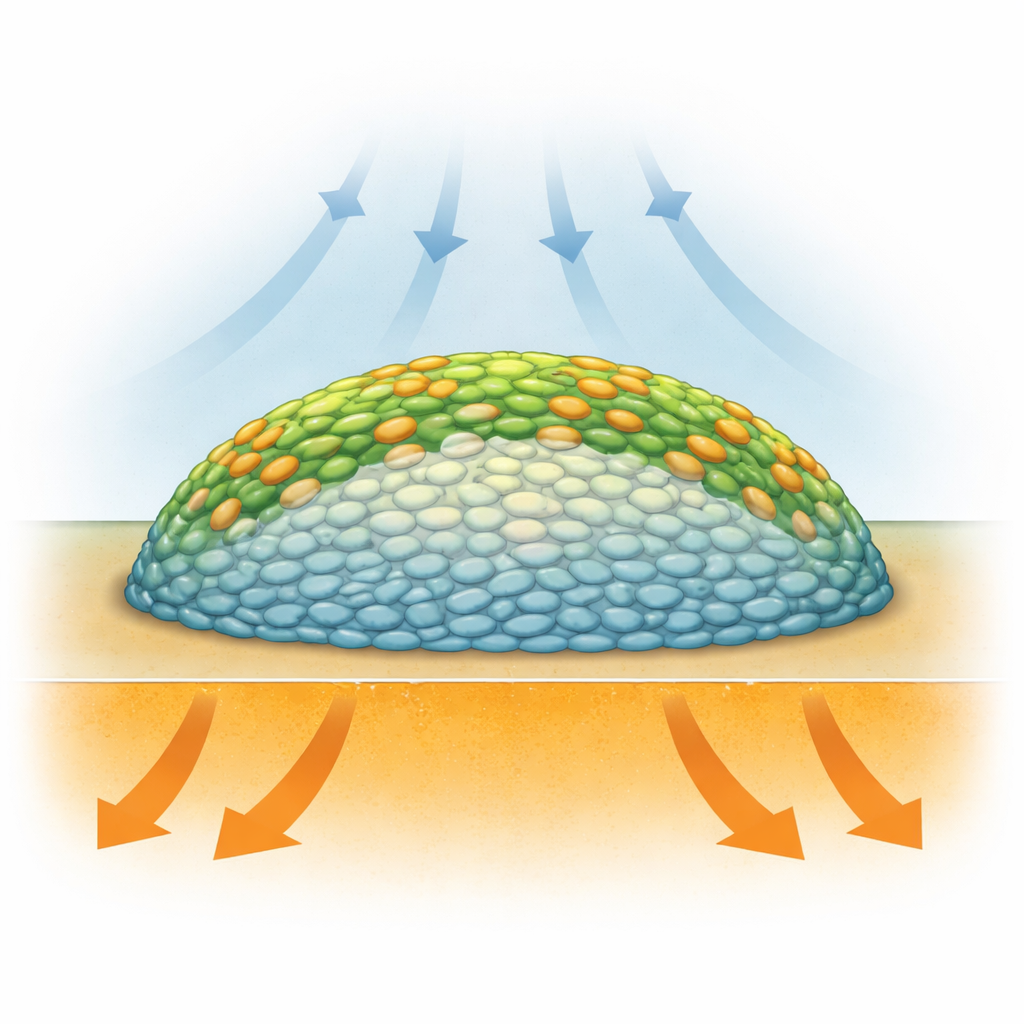
Biofilms are not just random clumps of germs: they are layered structures where bacteria live in very different conditions depending on where they sit. Cells on the outside have more access to oxygen from the air, while those buried in the center are starved of oxygen and may slow down or go dormant. Previous methods like fluorescent microscopy could show structure, but often required added dyes or genetic tags that might disturb the biology, and they did not directly measure what the cells were doing chemically. Researchers needed a way to “see” the chemistry of living biofilms, layer by layer, without tearing them apart or heavily altering them.
Seeing Chemistry With Invisible Light
The team turned to a technique called optical photothermal infrared (O-PTIR) spectroscopy, which uses pulses of invisible infrared light and a visible laser to detect tiny vibrations in molecules. These vibrations act like fingerprints of key cell components, such as proteins, fats, and DNA. To track metabolism—the active use of nutrients—they fed Salmonella Typhimurium biofilms a special form of sugar in which the carbon atoms were slightly heavier than usual (a stable isotope known as carbon-13). When cells are busy growing and making new proteins, they build these heavier atoms into their own structures, which causes subtle but detectable shifts in the infrared signal. By cryosectioning the biofilm and scanning thin cross-sections, the researchers produced chemical maps showing where in the biofilm this heavy carbon had been incorporated.
Activity Rings and Quiet Cores
Infrared imaging of untreated Salmonella biofilms revealed a striking pattern: the outer regions and surface layers showed strong incorporation of the heavy carbon, while a large central core displayed very little. In other words, the biofilm behaved like a ring of active cells surrounding a quiet interior. Statistical analysis of the infrared spectra confirmed that signals from protein-related vibrations shifted more in the outer layers than in the center, indicating higher protein production at the edges. These findings support the idea that oxygen availability, which is greatest near the surface, is the main driver of metabolic “hot zones” in this colony model, even though nutrients flow in from below. The study provided one of the clearest, highest-resolution views yet of how metabolism is stratified inside a biofilm.
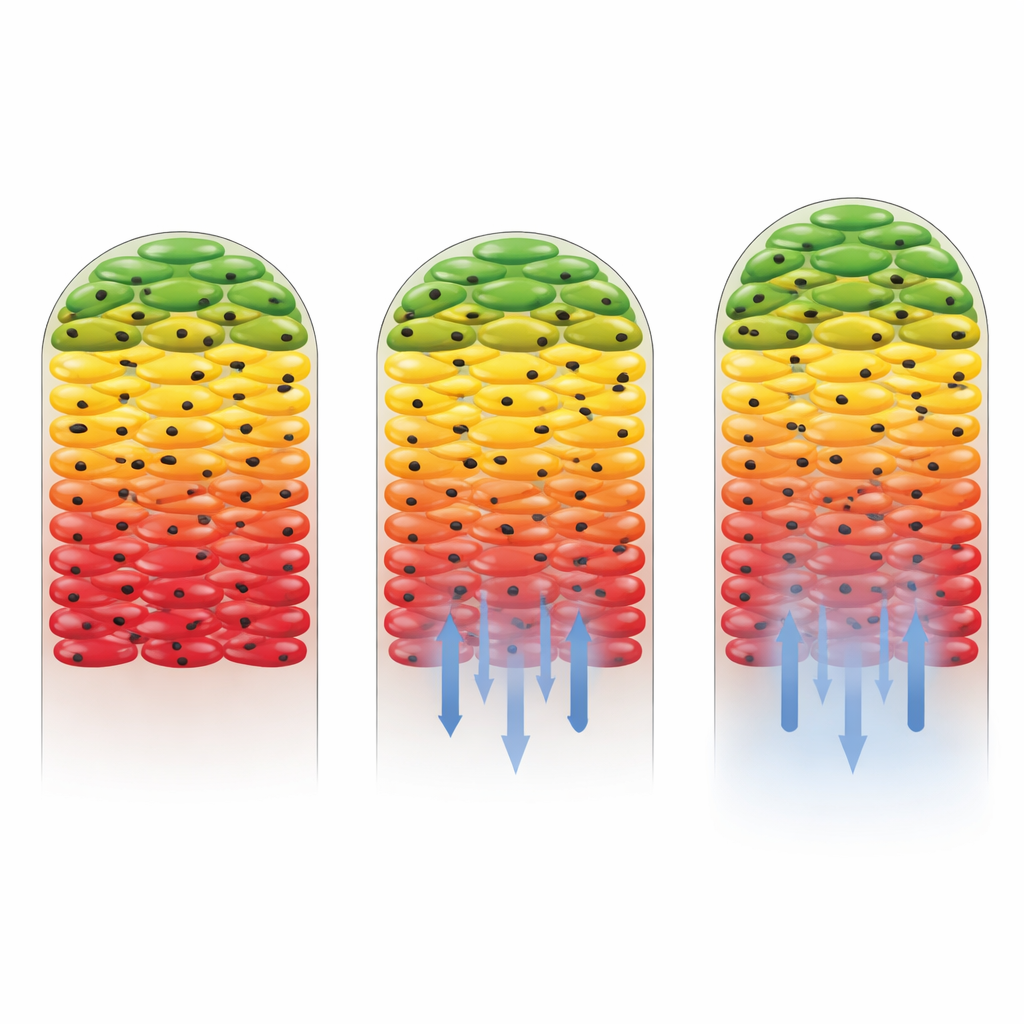
How Different Antibiotics Reshape Biofilm Activity
The researchers then asked what happens when antibiotics are introduced. They used two drugs: kanamycin, which the Salmonella strain is sensitive to, and gentamicin, to which the strain had been engineered to be largely non-susceptible. When gentamicin was present, the overall pattern of active outer layers and a quiet core barely changed, showing that resistant cells continued to grow and incorporate heavy carbon throughout much of the biofilm. In contrast, kanamycin dramatically reduced metabolic activity, especially near the bottom of the biofilm where the drug concentration was highest. Only a thin band of cells farthest from the antibiotic source still showed signs of carbon uptake. This revealed not only that the drug was effective, but also that the biofilm structure itself helped shield some cells by weakening the drug as it diffused inward.
New Windows Into Treating Tough Infections
From a lay perspective, the key message is that this work offers a powerful “chemical microscope” to watch how bacterial communities live and survive treatment in three dimensions. By combining a non-destructive infrared imaging method with a safe heavy form of carbon, the researchers could pinpoint where bacteria were actively growing and how that activity shifted under different antibiotics. Their results confirm that biofilms shelter a mix of busy and nearly dormant cells, and that effective drugs can still be blunted by the physical barrier of the biofilm. In the future, this approach could be used to test new therapies, explore how different species cooperate or share resistance inside mixed biofilms, and ultimately help design smarter strategies to clear persistent infections.
Citation: Smaje, D., Zhu, X., Hinton, J.C.D. et al. Investigating Salmonella biofilm responses to antibiotic treatment using optical photothermal infrared spectroscopy. Commun Biol 9, 405 (2026). https://doi.org/10.1038/s42003-026-09655-2
Keywords: biofilms, Salmonella, antibiotic resistance, infrared imaging, stable isotope probing