Clear Sky Science · en
Wnt11 mediates fibroblast–smooth muscle cell interaction to promote neurogenic bladder fibrosis in rats
When Nerves Fail, the Bladder Scars
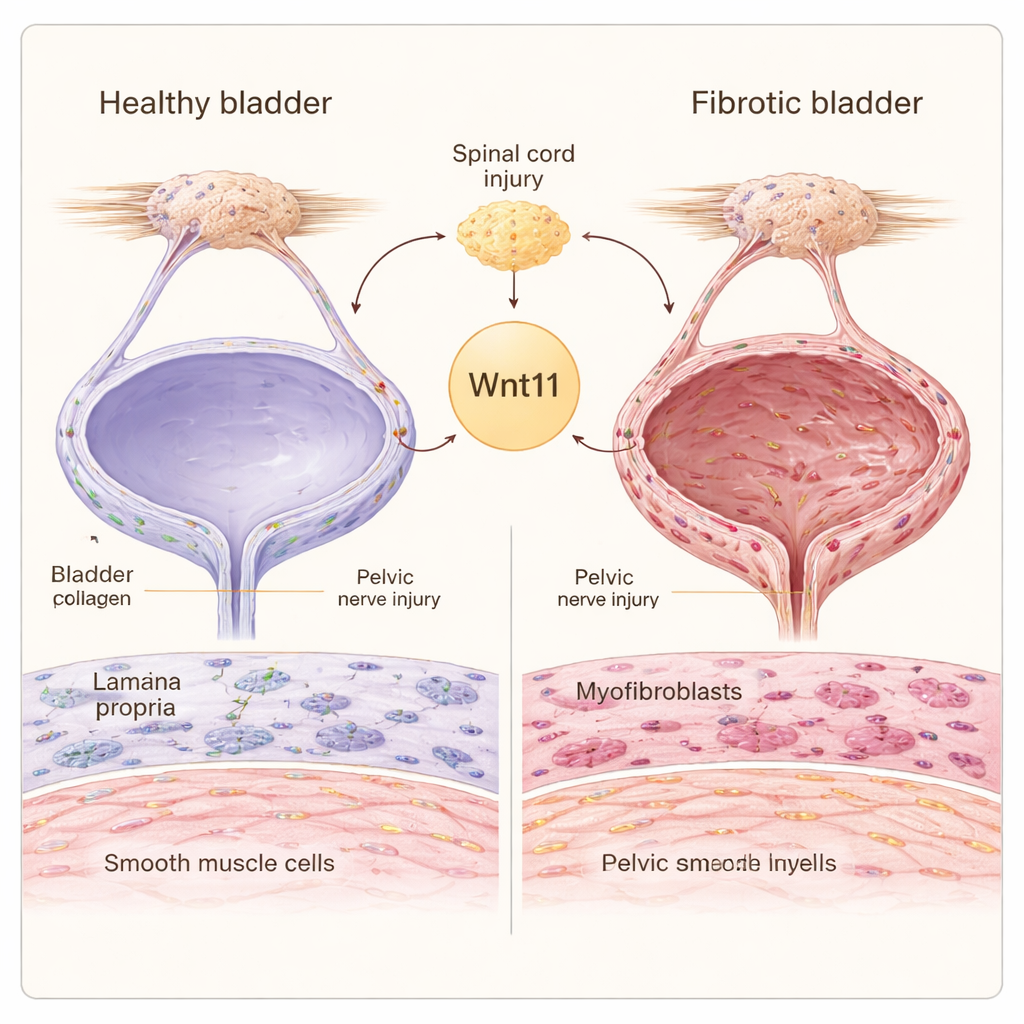
Many people with spinal cord injuries, multiple sclerosis, or Parkinson’s disease develop a “neurogenic bladder,” where damaged nerves can no longer properly control urine storage and release. Over time, the bladder wall can become thick and stiff with scar-like tissue, threatening kidney health and quality of life. This study in rats uncovers a key molecular culprit behind that scarring and suggests new ways to slow or even prevent the damage.
A Hidden Cost of Nerve Damage
When nerves that serve the bladder are injured, either in the spinal cord or near the pelvis, the organ must work harder to handle urine. The bladder wall contains two major supporting cell types: fibroblasts, which make the scaffolding material around cells, and smooth muscle cells, which squeeze to push urine out. In neurogenic bladder, fibroblasts can shift into highly active “myofibroblasts” that churn out excess collagen, while smooth muscle cells switch from a lean, contractile state to a bulkier, more synthetic one. Together, these changes lay down stiff scar tissue that reduces the bladder’s ability to stretch and empty.
A Signaling Molecule Steps Into the Spotlight
The researchers focused on a family of cell–cell communication proteins called Wnt ligands, which are known to regulate tissue growth and scarring in other organs. Screening bladders from rats with nerve damage, they found that one member, Wnt11, was consistently and strongly increased in fibrotic bladders caused by either spinal cord injury or pelvic nerve crush. Wnt11 levels rose in both fibroblasts and smooth muscle cells, and the amount of Wnt11 tracked with the severity of bladder dysfunction. When normal bladder cells in a dish were exposed to another pro-scarring factor, TGF-β1, they also ramped up Wnt11 production, hinting at a reinforcing loop between these two signals.
From Chemical Message to Lasting Scar
To test whether Wnt11 is merely a bystander or an active driver, the team added extra Wnt11 protein to rats with nerve injury and also blocked Wnt secretion with a drug called LGK974. Extra Wnt11 enlarged the bladders, worsened pressure patterns during filling and emptying, thickened the muscle layer, and boosted collagen deposition. Blocking Wnt secretion had the opposite effect, easing urinary problems and reducing scar tissue. In isolated cells, Wnt11 pushed fibroblasts toward myofibroblast status and nudged smooth muscle cells toward the synthetic, fibrosis-promoting phenotype. Silencing the Wnt11 gene itself blunted these changes, making it clear that Wnt11 is a central accelerator of bladder remodeling.
How Cells Decode the Fibrosis Signal
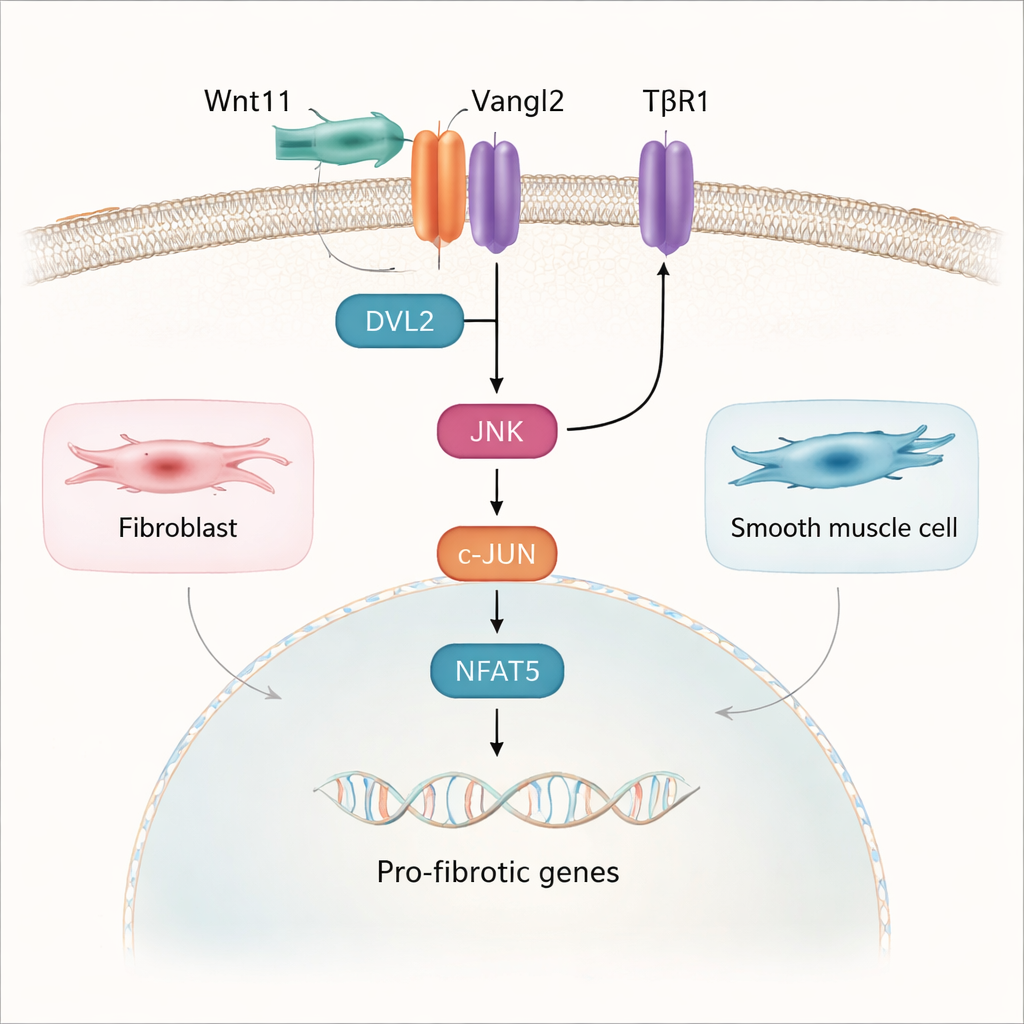
Delving deeper, the study mapped the pathway from Wnt11 at the cell surface to gene changes in the nucleus. Wnt11 was found to bind a receptor called Vangl2 on bladder fibroblasts and smooth muscle cells. This partnership did not use the classic Wnt route involving the protein β-catenin; instead, it activated a different chain called the planar cell polarity, or PCP, pathway. Here, Wnt11 and Vangl2 recruit an adaptor protein called DVL2, which then switches on the enzyme JNK. JNK in turn activates the transcription factors c-JUN and NFAT5, which move into the nucleus and help turn on pro-fibrotic genes. The TGF-β1 pathway, a well-known driver of scarring, physically and functionally converged with this Wnt11–Vangl2–JNK route, so that the two signals reinforce each other at both the cell membrane and in the nucleus.
Toward Softer, Healthier Bladders
Because Wnt11 and TGF-β1 cooperate to harden the bladder, the team tested drugs that block key steps in both pathways. In rats with neurogenic bladder, combined inhibition of the TGF-β receptor TβR1 and the JNK enzyme yielded the best results: smaller bladders, thinner muscle walls, less collagen, and improved urine storage and release compared with single-drug treatment. For a layperson, the take-home message is that a specific nerve injury–induced signal, Wnt11, helps convert normal support and muscle cells into scar-forming cells. Interrupting this signal, especially together with classic TGF-β blockers, could form the basis of future treatments aimed not just at managing symptoms, but at preserving the bladder’s structure and function in people with neurogenic bladder.
Citation: Ge, Q., Zhang, J., Fan, Z. et al. Wnt11 mediates fibroblast–smooth muscle cell interaction to promote neurogenic bladder fibrosis in rats. Commun Biol 9, 194 (2026). https://doi.org/10.1038/s42003-026-09647-2
Keywords: neurogenic bladder, fibrosis, Wnt11, smooth muscle cells, TGF-β signaling