Clear Sky Science · en
Structural analysis reveals that water molecules mediate self-activation of GPR99
Why this receptor matters for inflammation
Our airways and other tissues are constantly sensing chemical cues that signal infection or irritation. One such sensor, a protein called GPR99 that sits on cell surfaces, is emerging as an important switch in inflammatory diseases such as asthma and nasal polyps. This study explains, in atomic detail, how GPR99 can switch itself on even without an external chemical trigger—and how tiny clusters of water help it do so—offering clues for designing new anti‑inflammatory drugs.
A built‑in on switch inside a cell sensor
GPR99 belongs to a large family of cell‑surface proteins that relay messages from outside the cell to the inside, often by changing shape and recruiting partner proteins called G proteins. Unlike most such receptors, GPR99 is unusually active even when no signaling molecule is bound. The authors show that a flexible loop on the outer side of GPR99, known as the second extracellular loop, behaves like an internal key: it folds into the usual binding pocket, mimicking an activating signal and turning the receptor on by itself.
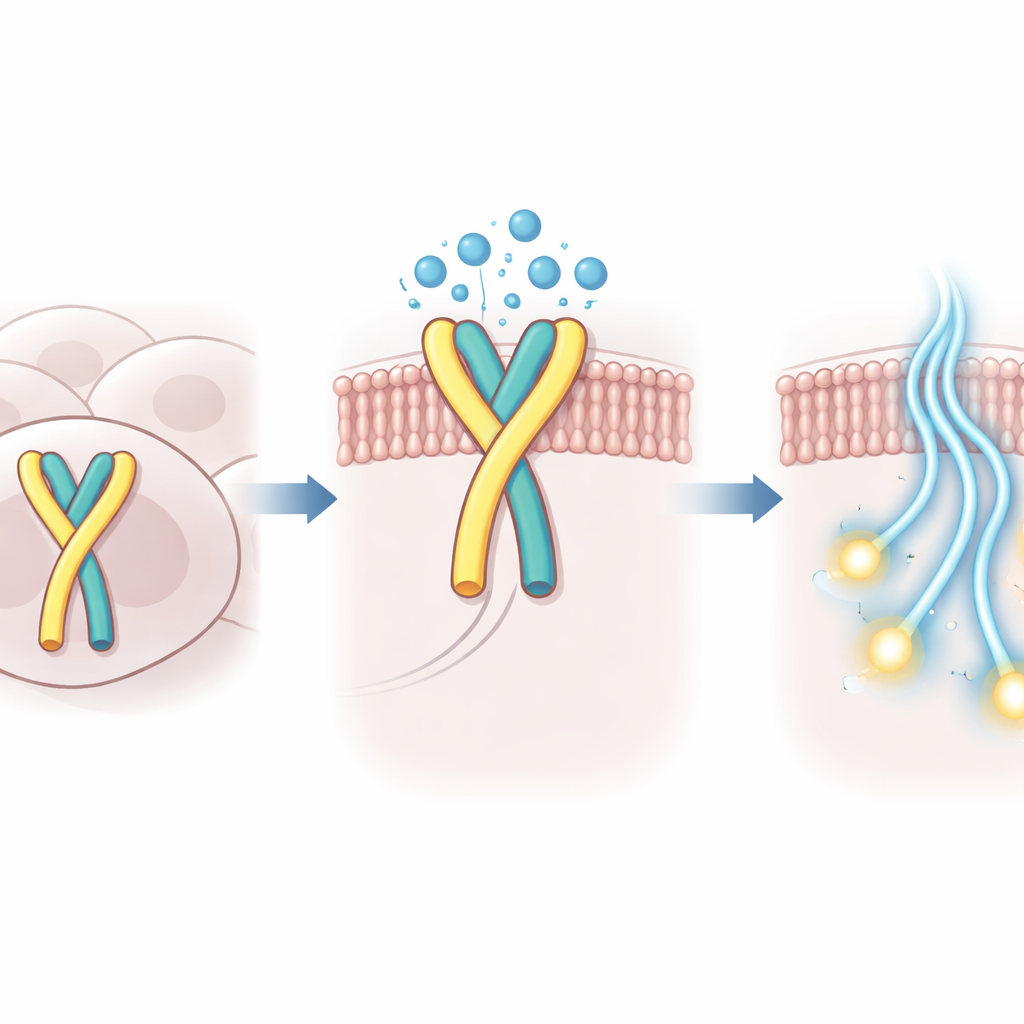
How water helps hold the switch in place
Using high‑resolution cryo‑electron microscopy, the researchers captured the three‑dimensional structure of human GPR99 bound to its G protein partner in this self‑activated state. In the binding pocket where external molecules would normally sit, they observed not only the tucked‑in loop but also a small cluster of ordered water molecules. These water molecules form a polar bridge between the loop and the surrounding protein, compensating for the shallower insertion of the loop and stabilizing the active shape of the receptor. When key amino acids in the loop or in the neighboring pocket were altered so that these water‑mediated contacts were weakened, GPR99’s built‑in activity dropped sharply in cell‑based tests.
Rearranging the inner workings for constant signaling
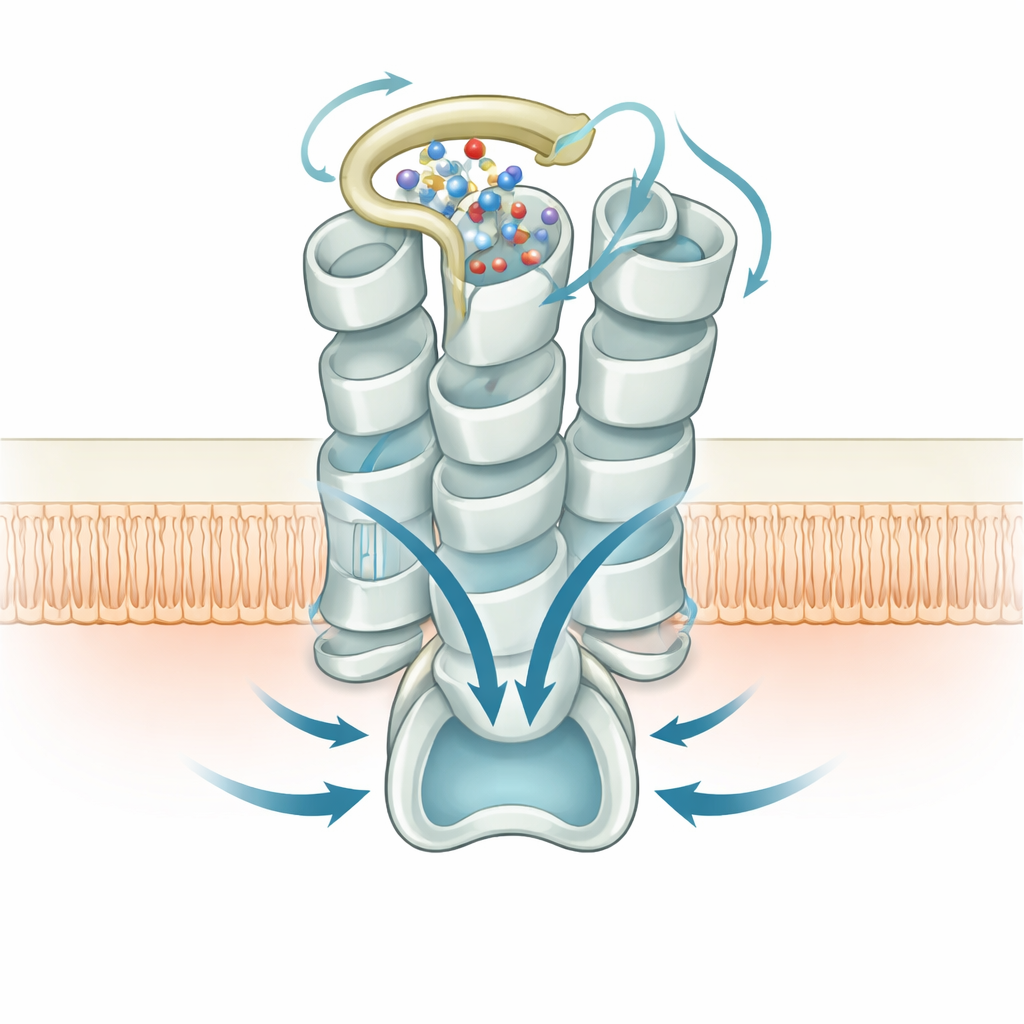
The team compared their self‑activated structure to other family members, including a closely related receptor that binds the metabolic by‑product succinate and an active form of GPR99 bound to the metabolite 2‑oxoglutarate. They found that GPR99 in the absence of any external ligand is already fully poised in an active conformation: several conserved “microswitches” within its helical core adopt shapes normally seen only when these receptors are stimulated. Critical positions that usually help hold receptors in an off state are replaced or reoriented in GPR99, loosening internal restraints and favoring the open configuration that welcomes the G protein partner.
Passing the message to inside the cell
On the inner side of the membrane, the structure reveals how GPR99 grips the Gq‑type G protein that transmits its signal. A helical tail of the G protein nestles into a cavity opened by the outward swing of one of GPR99’s helices, making a network of polar and hydrophobic contacts with the receptor’s inner loops and tail. Mutations that disturb this interface also reduce signaling, confirming that the self‑activation created by the outer loop and water cluster is efficiently coupled to intracellular partners. Together, these structural snapshots connect changes at the outer surface of the receptor to the waves of calcium signaling it triggers inside cells.
Implications for breathing and beyond
The authors suggest that GPR99’s built‑in activity, reinforced by structural water molecules, may keep airway tissues in a “standby” mode—primed to release mucus and mount an immune response when inflammatory lipids or metabolic changes appear. Because GPR99 is highly active even without external signals, and because its activation relies on a specific loop‑and‑water arrangement, these structural insights offer concrete targets for drug designers. Small molecules that disrupt this internal switch or its water network could dial down excessive inflammation, while compounds that fine‑tune the interaction might harness GPR99’s sensitivity for therapeutic benefit.
Citation: Xiao, M., Bao, X., Guo, Y. et al. Structural analysis reveals that water molecules mediate self-activation of GPR99. Commun Biol 9, 342 (2026). https://doi.org/10.1038/s42003-026-09644-5
Keywords: GPR99, GPCR activation, structural biology, inflammation, water-mediated signaling