Clear Sky Science · en
Structural mechanism of anti-MHC-I antibody blocking of inhibitory NK cell receptors in tumor immunity
Unlocking the Body’s Silent Cancer Killers
Our immune system has a set of powerful sentinels called natural killer (NK) cells that can detect and destroy cancer and virus-infected cells. Yet tumors often learn to silence these sentinels. This paper reveals, at atomic detail, how a specially designed antibody can remove that “mute button,” reawakening NK cells so they attack tumors more effectively. The work combines cutting-edge structural biology with mouse experiments to show how one antibody could complement existing cancer immunotherapies.
The Immune ID Tags on Our Cells
Every cell in our body displays molecular “ID tags” on its surface called MHC class I molecules. These tags help immune cells distinguish normal tissue from dangerous cells that have turned cancerous or been infected by viruses. NK cells constantly scan these tags. When they sense normal patterns, inhibitory receptors on NK cells send a calming signal that prevents attack. Tumors often keep these tags visible, using them as a protective shield: as long as the inhibitory signal flows, NK cells hold their fire and the cancer can grow.
An Antibody That Grabs the Shield
The researchers studied a monoclonal antibody named B1.23.2 that binds strongly to many human MHC class I variants on tumor cells. Using cryo-electron microscopy and X-ray crystallography, they mapped exactly how this antibody sits on the MHC surface. B1.23.2 latches onto a conserved ridge of the MHC molecule—an α2 helix—without touching its light chain partner and only lightly sensing the presented peptide. This means the antibody can recognize a broad array of human MHC-A, -B, and -C molecules with very high affinity, while being relatively insensitive to the specific small protein fragments the MHC is displaying.
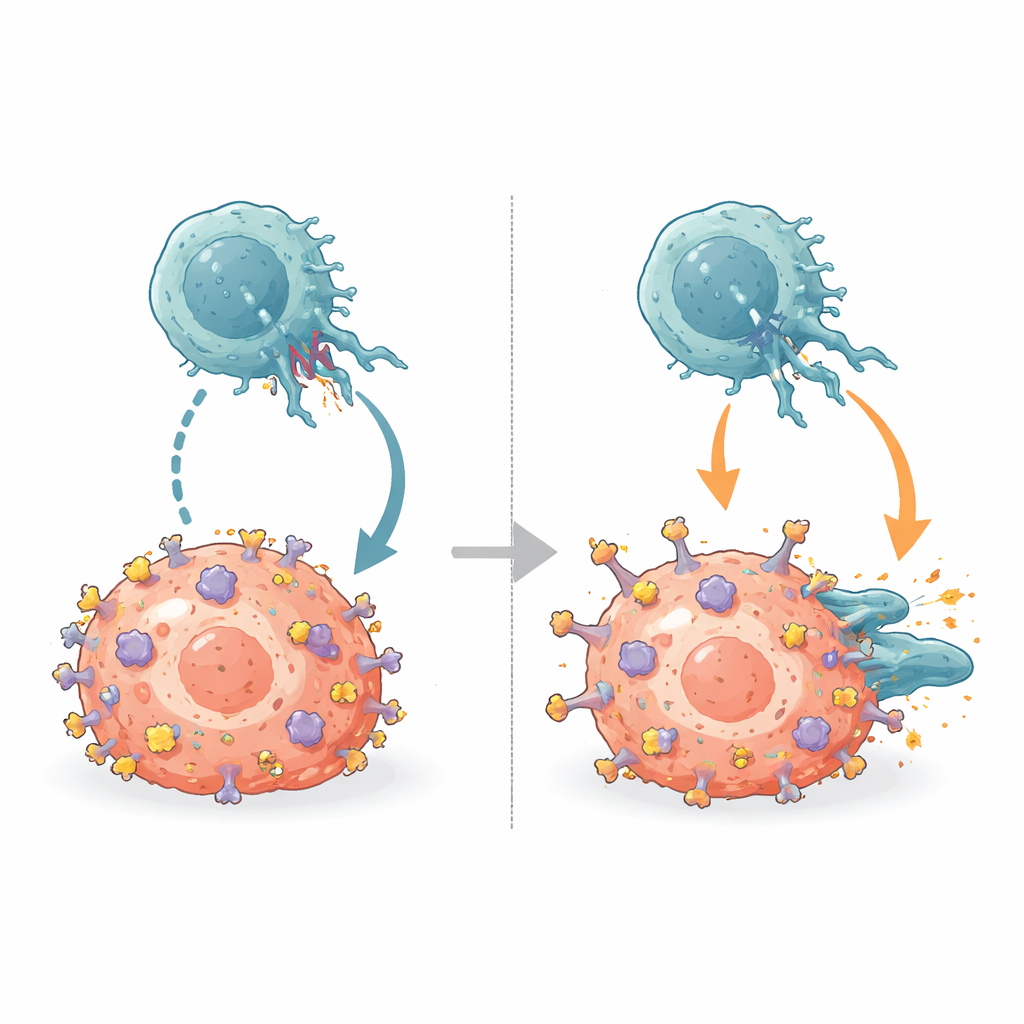
Outcompeting the Immune Brakes
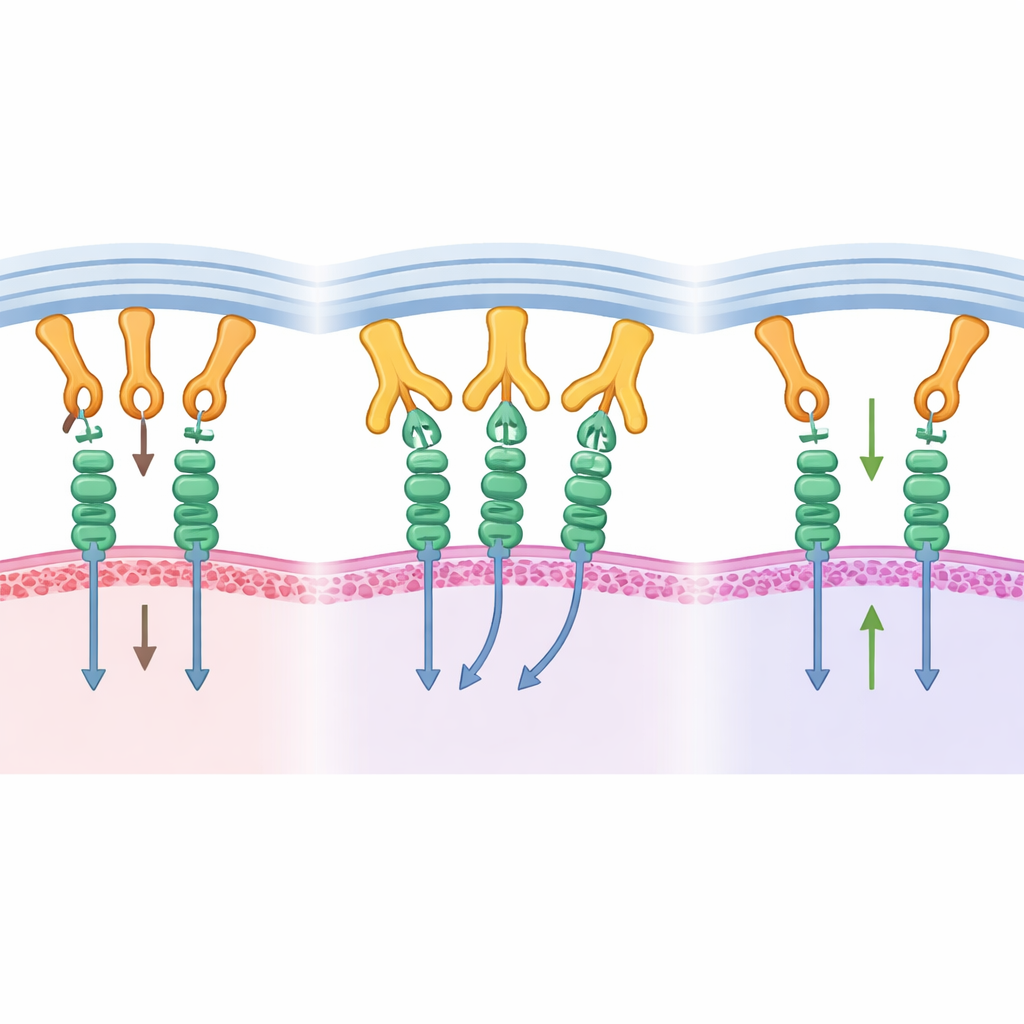
NK cells carry inhibitory receptors called KIRs that normally dock on the same α2 ridge of MHC. By aligning the new antibody–MHC structures with known KIR–MHC structures, the team showed that B1.23.2 physically occupies the very patch of MHC that KIRs need to grip. The antibody’s binding site and the KIR footprints overlap so extensively that both simply cannot bind at once. B1.23.2 also binds more tightly to MHC than KIRs do, meaning it wins this molecular tug-of-war. As a result, when the antibody coats tumor cell MHC molecules, KIRs on NK cells can no longer deliver their inhibitory signals, shifting the balance toward NK cell activation.
Fine-Tuning the Fit and Broad Reach
Although B1.23.2 mainly targets the conserved MHC surface, the authors found that small differences in the MHC-bound peptide can subtly tune antibody strength. By systematically swapping a single amino acid position in the peptide, they showed modest changes in binding—generally within about tenfold—without losing overall recognition. They also probed why certain common MHC types, such as HLA-A*02:01, escape binding. By transplanting key contact residues from a responsive MHC variant into HLA-A*02:01, the team engineered versions that the antibody could now recognize, confirming precisely which positions form the critical docking surface. These insights hint that future antibody variants could be designed to cover even more MHC types or selectively avoid others.
Turning Structural Insight into Tumor Control
Does this molecular blocking act actually help fight tumors? To test this, the researchers used a humanized mouse model carrying a human pancreatic cancer. They engineered a form of B1.23.2 that cannot signal through its own Fc tail, so any effects would mainly come from blocking the MHC–KIR interaction. When mice received the antibody along with human NK cells, tumors grew much more slowly. NK cells showed increased proliferation, higher activity of growth and signaling pathways, and greater production of inflammatory molecules associated with cancer cell killing. Within the tumors, NK cells displayed more activating receptors, consistent with being freed from constant inhibitory input.
What This Could Mean for Future Cancer Therapies
In plain terms, this study shows how a single antibody can slip between tumor cells and the immune system’s brakes, stopping the “do not attack” signal that normally keeps NK cells quiet. By mapping the interaction at atomic resolution and confirming its effects in animals, the work offers a blueprint for a new class of treatments: antibodies that bind the tumor’s ID tags rather than the immune receptors themselves. Such agents could be combined with existing checkpoint inhibitors or cell therapies to mobilize both NK cells and T cells, giving the immune system another angle of attack against hard-to-treat cancers.
Citation: Jiang, J., Panda, A.K., Natarajan, K. et al. Structural mechanism of anti-MHC-I antibody blocking of inhibitory NK cell receptors in tumor immunity. Commun Biol 9, 350 (2026). https://doi.org/10.1038/s42003-026-09641-8
Keywords: natural killer cells, MHC class I, cancer immunotherapy, monoclonal antibodies, inhibitory receptors