Clear Sky Science · en
LukS-PV targeting C5aR inhibits EMT in hepatocellular carcinoma via the BCL6/HDAC6/HSPD1 axis
Turning a Bacterial Weapon into a Cancer Fighter
Hepatocellular carcinoma, the most common form of liver cancer, is often deadly because it spreads from the liver to other organs. This paper explores an unexpected ally against that spread: a protein, LukS-PV, originally part of a toxin made by the bacterium Staphylococcus aureus. The researchers show how this protein can latch onto a specific receptor on liver cancer cells and, instead of harming normal tissue, shut down key steps that allow cancer cells to detach, travel, and form new tumors.
Why Stopping Shape-Shifting Cancer Cells Matters
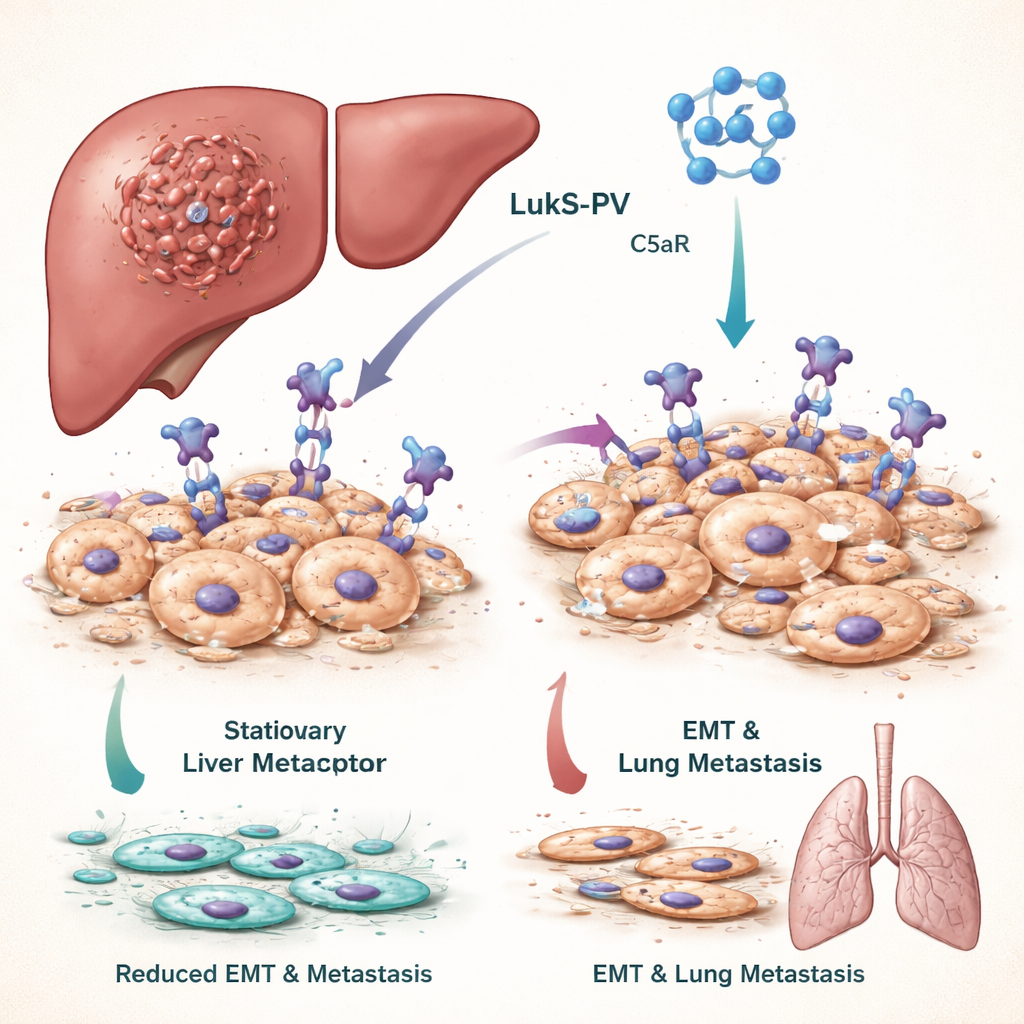
For liver cancer to spread, tumor cells must undergo a dramatic shape and behavior change called epithelial–mesenchymal transition, or EMT. In EMT, tightly packed cells that normally stay put become more flexible, mobile, and invasive, enabling them to slip into blood vessels and seed distant organs such as the lungs. Patients whose tumors show strong EMT activity tend to have worse outcomes. The team focused on a signaling pathway driven by a molecule called C5a and its receptor C5aR, which is unusually abundant on many tumor cells and is known to fuel EMT and metastasis.
A Toxin Component That Targets a Cancer Signal
Panton–Valentine leukocidin, a toxin from Staphylococcus aureus, contains a component called LukS-PV that naturally recognizes and binds C5aR. Previous work by the authors showed that a recombinant, non–pore-forming version of LukS-PV can curb the growth of several cancers without obvious toxicity in animals. In this study, they treated liver cancer cell lines with LukS-PV and observed that the cells became less migratory and invasive. Molecular tests revealed that markers of stationary, well-behaved cells increased, while markers of aggressive, mobile cells declined. In mice injected with liver cancer cells, LukS-PV treatment led to far fewer tumor nodules in the lungs, indicating reduced metastasis.
Blocking a Chain Reaction Inside Tumor Cells
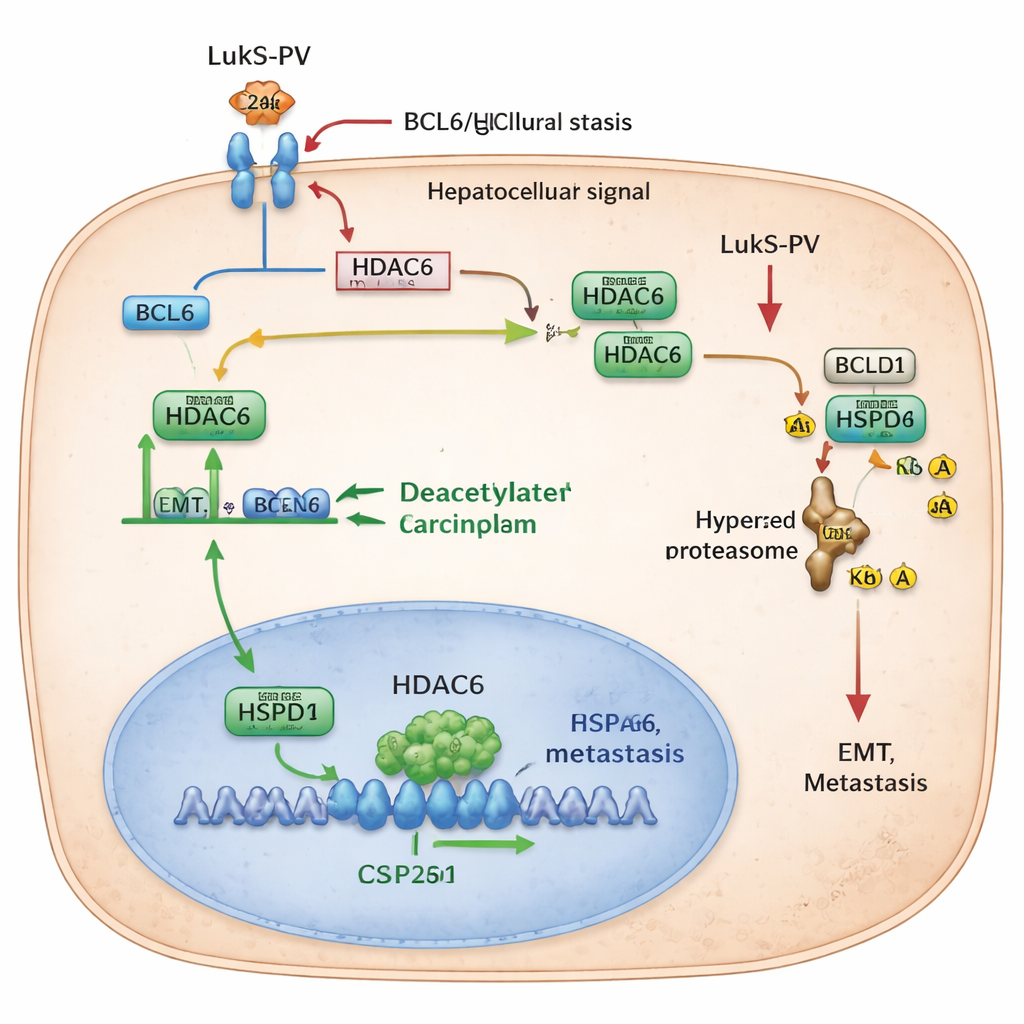
Digging deeper, the researchers uncovered a chain of control proteins inside the cancer cells that LukS-PV disrupts. First, LukS-PV binds to C5aR on the cell surface and effectively blocks the pro-metastatic signal from C5a. This dampening of C5aR signaling lowers levels of a protein called BCL6, a transcription factor that normally switches on certain genes in the nucleus. One of BCL6’s targets is HDAC6, an enzyme that removes small chemical tags known as acetyl groups from other proteins. When BCL6 is abundant, HDAC6 levels rise, and the cell becomes more prone to EMT and movement. LukS-PV’s interference with C5aR reduces BCL6, which in turn cuts HDAC6 production and slows this pro-metastatic program.
Tagging a Chaperone for Destruction
The team then identified a key partner of HDAC6 called HSPD1, also known as HSP60, a “chaperone” protein that helps other proteins fold and stay stable. In liver cancer, HSPD1 levels are higher than in nearby normal tissue and are linked to poorer patient survival. The researchers discovered that HDAC6 physically interacts with HSPD1 and strips acetyl groups from two specific positions on it. When these acetyl tags are removed, HSPD1 becomes more stable and supports EMT and metastasis. When HDAC6 is reduced—either by genetic tricks or by LukS-PV treatment—HSPD1 becomes more acetylated. This hyperacetylated form is recognized by the cell’s waste-disposal machinery, tagged with ubiquitin, and broken down. As HSPD1 levels fall, cancer cells lose some of their ability to migrate, invade, and colonize the lungs.
From Complex Pathways to a Simple Takeaway
To a non-specialist, the message is that the authors have mapped a domino effect inside liver cancer cells: a surface receptor (C5aR) boosts a controller protein (BCL6), which raises an enzyme (HDAC6) that protects a chaperone (HSPD1) from being destroyed. Together, these players help cancer cells change shape, move, and spread. LukS-PV, a bacterial protein repurposed in the lab, interrupts this sequence right at the cell surface, leading ultimately to the breakdown of HSPD1 and a brake on metastasis. While still at the experimental stage, this work suggests that drugs modeled on LukS-PV—or aimed at BCL6, HDAC6, or HSPD1—could offer new ways to keep liver tumors from spreading and improve the outlook for patients.
Citation: Ding, P., Shi, L., Xu, X. et al. LukS-PV targeting C5aR inhibits EMT in hepatocellular carcinoma via the BCL6/HDAC6/HSPD1 axis. Commun Biol 9, 314 (2026). https://doi.org/10.1038/s42003-026-09640-9
Keywords: hepatocellular carcinoma, metastasis, epithelial mesenchymal transition, HDAC6, bacterial toxin therapy