Clear Sky Science · en
Mesenchymal stem cell-derived extracellular matrix for musculoskeletal tissue regeneration
Helping the Body Rebuild Its Own Frame
As people live longer and stay active into old age, injuries and wear-and-tear to bones, joints, muscles, and tendons are becoming a major cause of pain and disability. This article explores a new class of “smart” repair materials grown from our own stem cells—special support structures called mesenchymal stem cell–derived extracellular matrix (mECM). These natural scaffolds could one day help the body rebuild damaged musculoskeletal tissues more safely and effectively than many current treatments.
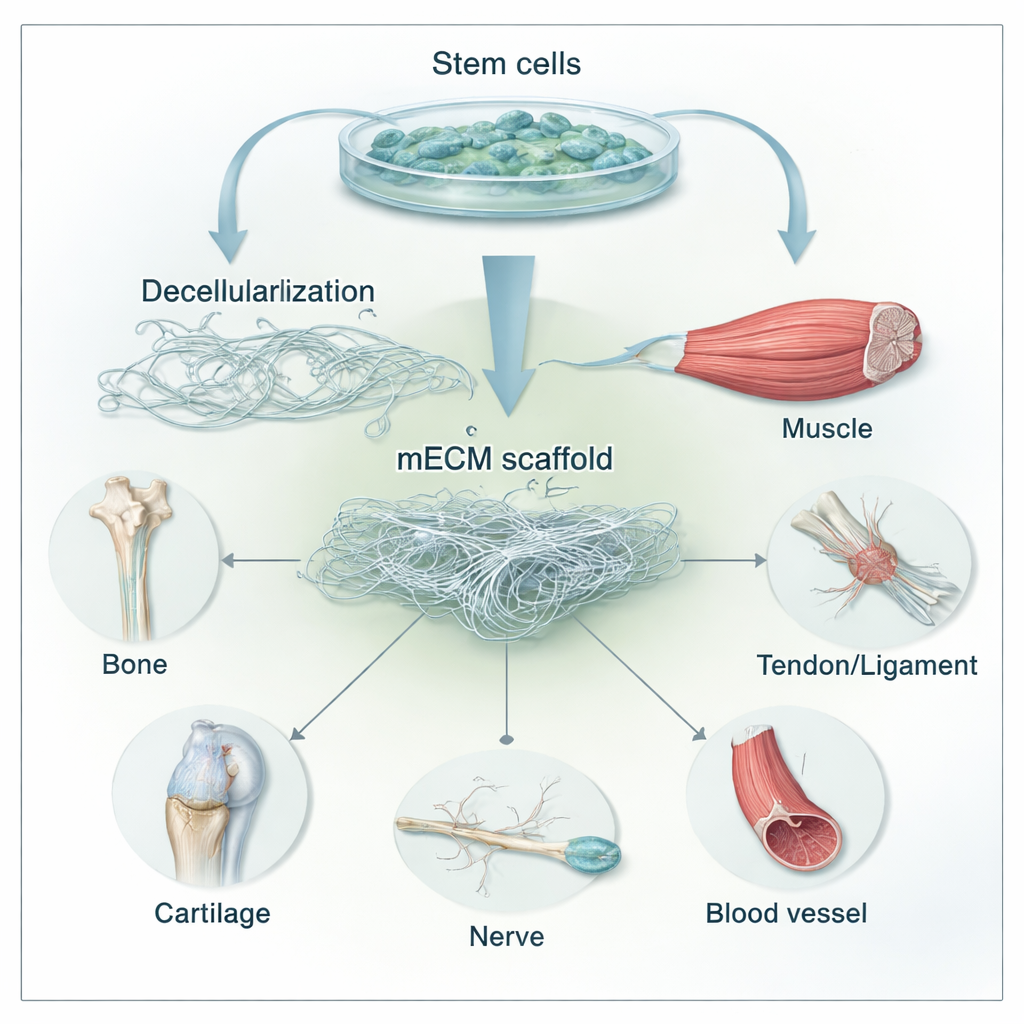
The Body’s Hidden Building Scaffold
Every cell in the body lives within a supportive mesh known as the extracellular matrix, or ECM. It is not just passive glue: its fibers, pores, and bound molecules constantly signal to cells how to grow, move, and mature. In aging and disease, this matrix stiffens, frays, and loses its springiness, directly weakening our ability to repair bone and cartilage. Scientists have learned to strip cells out of tissues like skin, intestine, or cartilage, leaving behind “decellularized” ECM scaffolds that can be implanted to guide healing. But these tissue-derived materials can vary from donor to donor, may still trigger immune reactions, and are hard to customize for different patients or injuries.
Growing Custom Repair Material from Stem Cells
The review focuses on ECM made not from whole organs but from mesenchymal stem cells (MSCs), versatile cells found in bone marrow, fat, umbilical cord, and other tissues. In the lab, MSCs are allowed to spread across a surface and lay down their own matrix—a delicate three-dimensional web of collagen, fibronectin, laminin, sugars, and growth factors. The cells are then gently removed with mild detergents and enzymes, leaving a cell-free mECM. Because the ECM molecules are highly conserved across species and lack DNA and other strong immune triggers, these scaffolds are often well tolerated. Importantly, researchers can tune the composition and stiffness of mECM by choosing the stem cell source (for example, bone marrow vs. fat) and by “preconditioning” the cells with different cues before decellularization.
How This Living Scaffold Talks to Cells
When fresh cells are reseeded onto mECM, they behave very differently than on flat plastic. Studies show that mECM boosts cell growth while helping cells stay “younger,” delaying the onset of cellular aging and preserving their ability to turn into bone or cartilage. Young mECM, with softer mechanics and richer hyaluronic acid, appears especially powerful at rejuvenating older cells. Specific components, such as type I collagen and fibronectin, influence whether cells resist aging, form bone-resorbing osteoclasts, or respond to inflammation. The matrix also activates internal signaling routes linked to longevity and stress resistance, including SIRT1 and NF-κB, and can tilt immune cells toward a more calming, tissue-healing state. In short, mECM acts as both a physical nest and a biochemical instruction manual for cells.
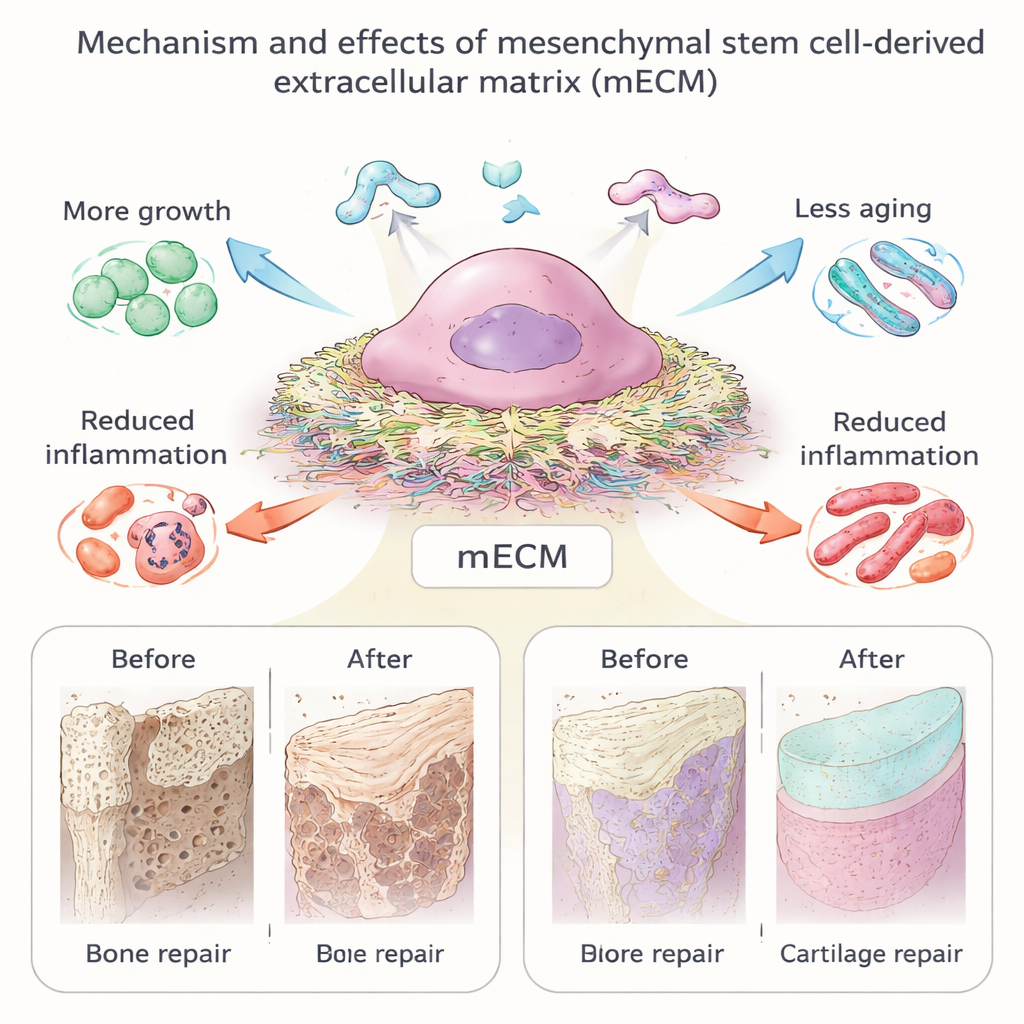
Repairing Bone, Cartilage, Muscle, Tendon, Nerves, and Vessels
Because mECM so closely mimics the tissues it is meant to repair, it has been tested in many forms: thin sheets wrapped around implants, powders blended into hydrogels and microspheres, and coatings on synthetic scaffolds. In bone repair, cells grown on mECM gain bone-forming ability and help build stronger, better-vascularized tissue in animals, even without heavy drug cocktails. In cartilage, mECM supports the formation of smooth, hyaline-like cartilage and helps chondrocytes (cartilage cells) avoid drifting into scar-like states—especially when the matrix comes from young or early-differentiated stem cells. Early work in muscle and tendon suggests that stem-cell-derived matrices can attract host cells, reduce scarring, and better align new fibers. In nerves and blood vessels, mECM integrated into guiding tubes or nanofibers accelerates nerve regrowth and stimulates new vessel formation, underscoring its broad regenerative potential.
From Lab Bench to Real-World Treatments
Despite encouraging results, mECM is not yet ready for routine clinical use. The field still lacks standard recipes for safely removing cells while preserving delicate matrix details, and the exact “active ingredients” that control cell behavior remain only partly understood. Different stem cell sources and ages produce noticeably different matrices, raising questions about which combinations are best for each injury type. Manufacturing mECM at scale is costly and technically demanding, and scientists must prove that large, long-lasting implants are safe, stable, and mechanically strong enough for load-bearing sites like joints and tendons. The authors argue that advances in proteomics, bioinformatics, and biofabrication will be crucial to map, standardize, and industrialize these living scaffolds. If these hurdles can be cleared, patient-specific mECM could become a new gold standard for repairing the body’s framework from within.
Citation: Lv, S., Wang, J., Chen, J. et al. Mesenchymal stem cell-derived extracellular matrix for musculoskeletal tissue regeneration. Commun Biol 9, 147 (2026). https://doi.org/10.1038/s42003-026-09638-3
Keywords: extracellular matrix, mesenchymal stem cells, bone and cartilage repair, tissue engineering, regenerative medicine