Clear Sky Science · en
Periodic confined cell migration drives partially reversible chromatin reorganization in cancer cell lines
How Squeezed Cancer Cells Remember Being Stressed
Cancer cells that break away from a tumor must crawl through tiny gaps in the body’s tissues and blood vessels. To do this, they squeeze their soft insides—and especially their nucleus, which houses DNA—through spaces sometimes smaller than the nucleus itself. This study explores what happens to the DNA’s packaging during those repeated squeezes and asks a key question: does the nucleus simply spring back to normal, or does it keep a kind of “mechanical memory” that might help cancer spread?
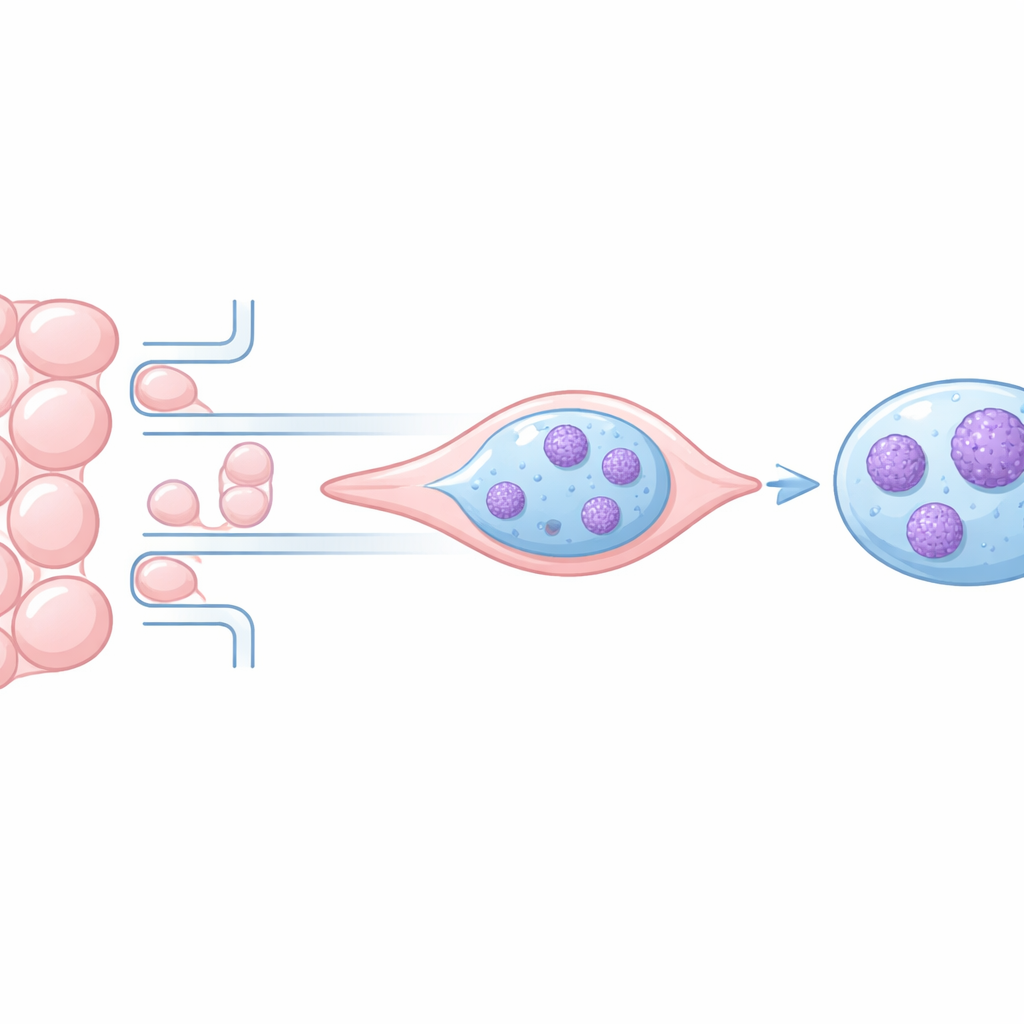
Why DNA Packing Matters When Cells Move
Inside the nucleus, DNA is wrapped around proteins and folded into chromatin, which can be loosely packed or tightly condensed. Looser regions tend to be active, hosting genes that are frequently used, while tightly packed regions are more silent. When cells migrate through narrow spaces, the nucleus deforms, and previous work showed that this can temporarily compact chromatin and even damage DNA. But it wasn’t clear whether those changes fully reverse or leave lasting marks that might alter how aggressive cancer cells become.
A Tiny Maze to Squeeze Single Cancer Cells
The researchers built a custom microfluidic device—a transparent chip containing dozens of microscopic channels that repeatedly alternate between wide and narrow segments. Two different cancer cell lines, both engineered so that their chromatin glows under the microscope, were guided to crawl through these channels using a chemical attractant. High-resolution time-lapse imaging allowed the team to track each cell nucleus as it moved, measuring nuclear size and mapping bright, highly condensed chromatin clusters frame by frame in more than 100 individual cells.
What Happens to the Nucleus Under Pressure
Whenever a cell entered a narrow constriction, its nucleus abruptly shrank in projected area, reflecting a reversible reduction in nuclear volume. At the same time, the fraction of the nucleus occupied by highly condensed chromatin spiked—often two- to threefold—then dropped back to its baseline once the cell emerged into a wider region. This pattern repeated across several successive constrictions and in both cancer cell types, suggesting that the overall amount of dense chromatin is a flexible, short-term response to mechanical stress rather than a permanent change. However, a closer look at the fluorescence patterns revealed that pixels with both very high and very low intensity increased during squeezing, hinting at a more complex internal rearrangement than a simple uniform compaction.
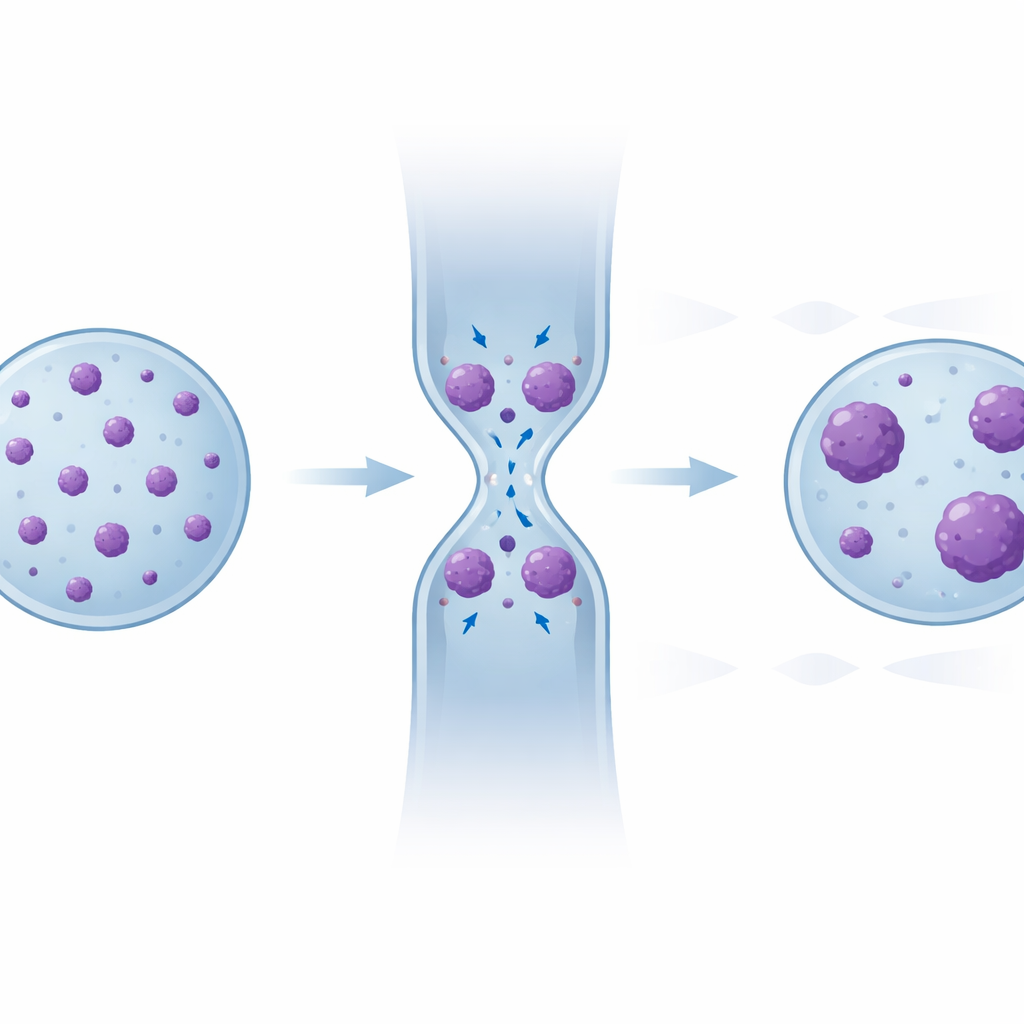
Hidden Memory in the Pattern of DNA Clusters
To uncover that hidden reorganizing, the team examined each individual condensed chromatin cluster—its size and how many such clusters each nucleus contained—before, during, and after confinement. During squeezing, small clusters tended to come together into larger ones. After the nucleus relaxed, the total condensed area returned to its original level, but the pattern did not: compared to the starting state, cells that had passed through several constrictions showed fewer clusters overall, biased toward larger sizes. Statistical comparisons confirmed that distributions in later “recovery” regions looked more and more like those seen during confinement. Immunofluorescence staining further showed that many of these dense regions overlapped with established “silent” chromatin marks, and a computer model of chromatin as an interacting polymer reproduced this partial but not complete relaxation simply from the physics of sticky DNA segments brought together by deformation.
What This Means for Cancer Spread and Future Therapies
In everyday terms, the study finds that when cancer cell nuclei are repeatedly squeezed, their DNA packing behaves like a stress ball with a memory. The nucleus can bounce back in size, and the overall amount of tight packing can reset, but the detailed pattern of dense DNA clumps does not fully return to its original state. Over many squeezing cycles, the nucleus ends up with fewer, larger dense regions, potentially altering which genes are easier or harder to access. This “mechanical memory” could help cancer cells better tolerate future deformations as they invade new tissues. Understanding and eventually targeting this coupling between physical forces and DNA organization might open new ways to limit metastasis by disrupting how cancer cells adapt to being mechanically stressed.
Citation: Blazquez-Romero, M.d.V., Mendivil-Carboni, M., Sarasquete-Martinez, M. et al. Periodic confined cell migration drives partially reversible chromatin reorganization in cancer cell lines. Commun Biol 9, 366 (2026). https://doi.org/10.1038/s42003-026-09637-4
Keywords: confined cell migration, chromatin condensation, nuclear mechanics, mechanical memory, cancer metastasis