Clear Sky Science · en
PTBP1 supports mouse spermatogenesis by facilitating cytoskeletal organization through the mTORC2–PKCα pathway in Sertoli cells
Why this study matters for male fertility
Male infertility often seems mysterious, but at its core it depends on a finely tuned partnership between developing sperm and the “nurse” cells that support them. This study digs into one of the hidden molecular helpers inside those nurse cells and shows how its failure can bring sperm production to a halt. Understanding this hidden layer of control could point the way toward new diagnostics or treatments for certain forms of male infertility.
The support cells that make sperm possible
Sperm are produced inside long, coiled tubes in the testis called seminiferous tubules. Lining these tubes are Sertoli cells, a specialized support cell type that cradle developing germ cells, feed them, and guide them as they divide and mature into sperm. Sertoli cells also build a protective wall known as the blood–testis barrier, which separates early-stage cells from the immune system. To do all this, they rely on an internal scaffold of actin filaments and microtubules that constantly remodels as waves of germ cells move through the tubules. If this scaffold is disturbed, the barrier leaks and maturing germ cells can detach or die, threatening fertility.
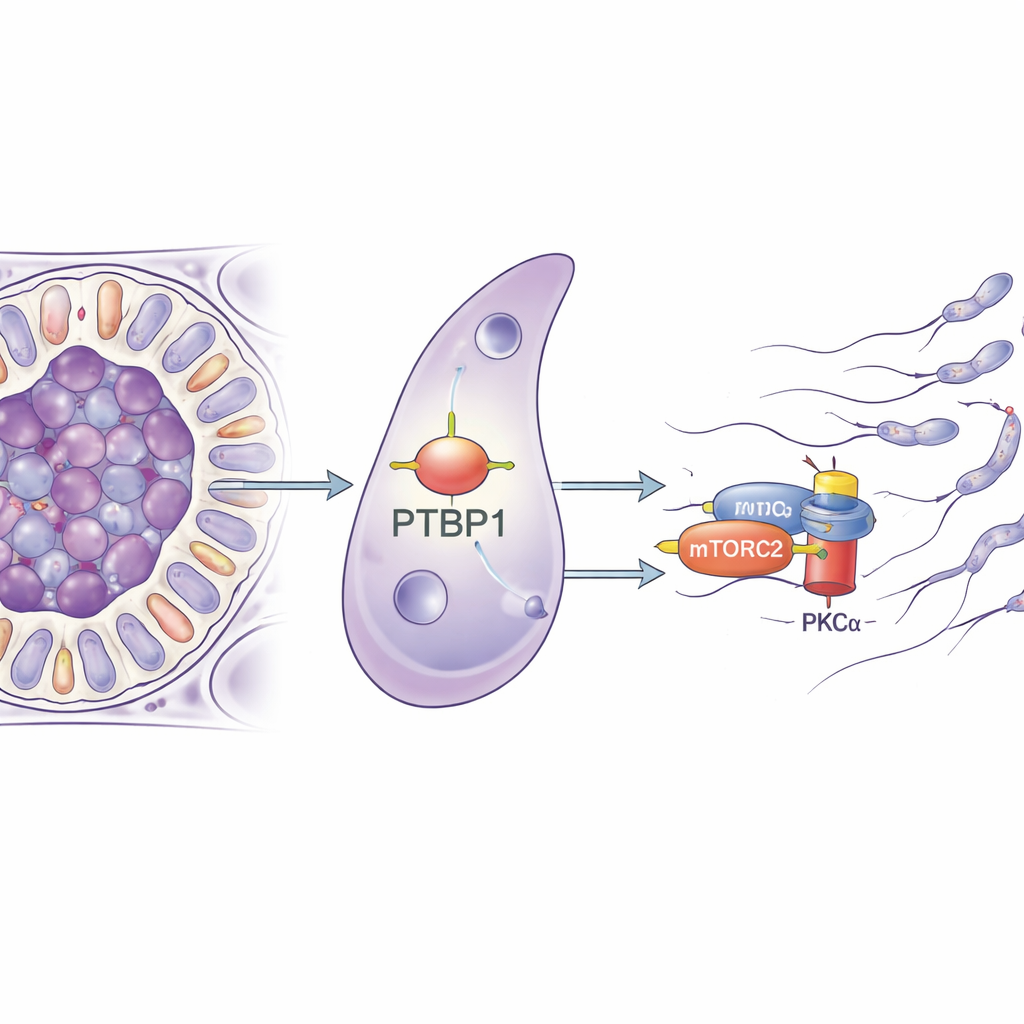
A tiny RNA manager with a big job
The researchers focused on a protein called PTBP1, best known for its role in managing RNA molecules inside cells—deciding how genes are spliced, how stable their messages are, and how efficiently they are turned into protein. PTBP1 is abundant in both germ cells and Sertoli cells, and earlier work showed that removing it from germ cells hampers sperm production. In this study, the team specifically deleted PTBP1 only in Sertoli cells in mice. At first glance, the young mice appeared normal, but as they reached sexual maturity their testes shrank, the number of sperm in the epididymis plummeted, and none of the males could sire offspring, revealing a complete loss of fertility.
When the cellular scaffold falls apart
Microscopic examination of the testes from these mice showed that spermatogenesis stalled halfway. Many tubules contained empty spaces, abnormal clumps of fused germ cell nuclei, and a shortage of fully elongated sperm. Sertoli cell nuclei, which normally hug the outer wall of the tubules, were often displaced toward the center, suggesting that these support cells were losing their grip on the surrounding structure. Staining for key junction components and structural proteins confirmed that the blood–testis barrier was weakened and that the usual tight bundles of actin filaments were replaced by disorganized, overly intense strands stretching from the base to the center of the tubule. A tracer molecule that normally stays on the “blood side” of the barrier instead leaked deep into the tubules, directly demonstrating barrier breakdown.
Tracing the failure back to a signaling hub
To understand how an RNA-binding protein could lead to such mechanical problems, the team purified Sertoli cells and analyzed which genes changed their activity when PTBP1 was missing. Many of the affected genes were involved in controlling the cell’s skeleton and in how cells adhere to each other, pointing to a broad remodeling defect. The researchers then pulled down the RNA molecules that physically bind PTBP1 and found that it latches onto the message for Rictor, a core part of a signaling complex called mTORC2. This complex, in turn, activates an enzyme called PKCα that shapes actin filaments. In Sertoli cells lacking PTBP1, the amount of RICTOR protein dropped, and PKCα was much less active, even though the underlying RNA level for Rictor barely changed. This strongly suggests that PTBP1 helps Sertoli cells make enough RICTOR protein from its RNA message, thereby keeping the mTORC2–PKCα pathway running.
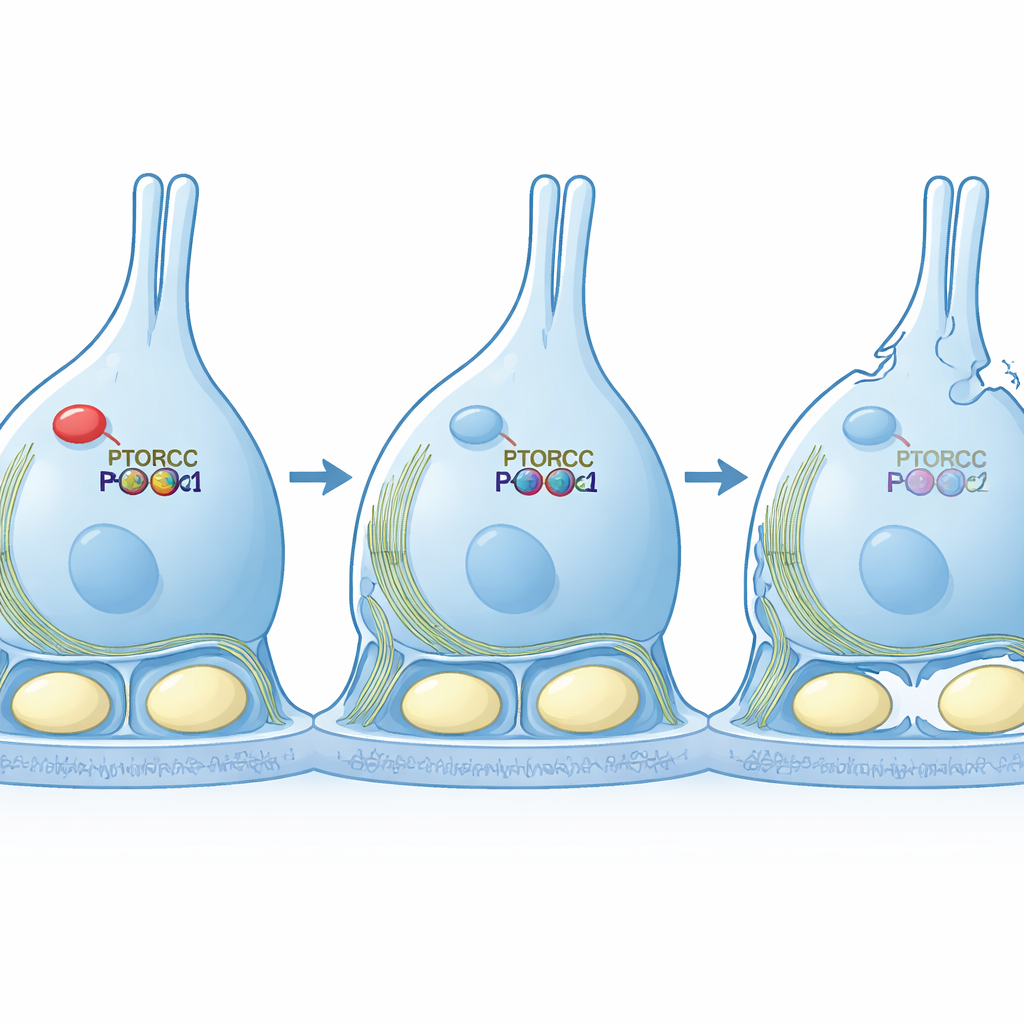
Rescuing the scaffold by restoring the signal
The team then moved to cell culture models to test causality. When they reduced PTBP1 in a Sertoli-like cell line, the cells stretched into long, thin shapes and displayed a scrambled actin network—hallmarks of weakened mTORC2 signaling. Importantly, when they forced these cells to produce a permanently active version of PKCα, the cells regained their normal, compact shape and more orderly actin structures, even though RICTOR itself remained low. This rescue experiment shows that the main job of PTBP1 in this context is to keep the mTORC2–PKCα pathway strong enough to organize the cell’s internal scaffold.
What this means for understanding infertility
In simple terms, this work reveals a chain of dependence inside the testis: PTBP1 helps Sertoli cells build a key signaling hub; that hub arranges their internal skeleton; and an intact skeleton lets them grip and protect developing sperm. Break the chain at PTBP1, and Sertoli cells lose their structure, the protective barrier fails, and sperm development collapses, resulting in male infertility. While this research was done in mice, the same molecules are present in humans, raising the possibility that subtle defects in RNA control within Sertoli cells could underlie some unexplained cases of male infertility and offering a new set of molecular targets for future study.
Citation: Ozawa, M., Taguchi, J., Mori, H. et al. PTBP1 supports mouse spermatogenesis by facilitating cytoskeletal organization through the mTORC2–PKCα pathway in Sertoli cells. Commun Biol 9, 341 (2026). https://doi.org/10.1038/s42003-026-09636-5
Keywords: male fertility, Sertoli cells, spermatogenesis, cytoskeleton, RNA-binding proteins