Clear Sky Science · en
Negative regulation of the NF-κB pathway by the ubiquitin ligase Nedd4-1(NE)
Why shutting off inflammation matters
Our immune system relies on rapid bursts of inflammation to fight infections and heal injuries, but those same forces can damage tissues and fuel cancer if they stay switched on for too long. This article explores how human and other primate cells avoid that danger by using a newly characterized built-in "brake" that quiets one of the body’s central alarm systems once its job is done.
A central alarm switch in our cells
At the heart of this story is NF-κB, a family of proteins that acts like a master switch for hundreds of genes involved in inflammation, cell survival, and immunity. When cells sense stress signals such as the inflammatory molecule TNFα, a chain of events frees NF-κB from its protein "chaperones" in the cell’s fluid interior and allows it to move into the nucleus, where it turns on pro‑inflammatory and pro‑survival genes. Because this pathway is so powerful, cells must tightly tune both its activation and its shutdown. Failure to turn it off has been linked to chronic inflammatory diseases and many cancers.
A primate-only brake on the alarm
The researchers focused on Nedd4-1, a member of a large family of enzymes that tag other proteins with a small molecule called ubiquitin. This tag often marks proteins for destruction or relocation inside the cell, reshaping signaling networks. A previously discovered splice variant of Nedd4-1, called Nedd4-1(NE), is found only in primates and carries a long extra segment at its front end that changes where it sits in the cell and what it recognizes. In contrast to the standard Nedd4-1 form, which can actually boost NF‑κB activity through other partners, the team found that Nedd4-1(NE) has the opposite effect: it dampens NF‑κB signaling, especially after prolonged TNFα exposure.
Two ways to hold NF-κB in check
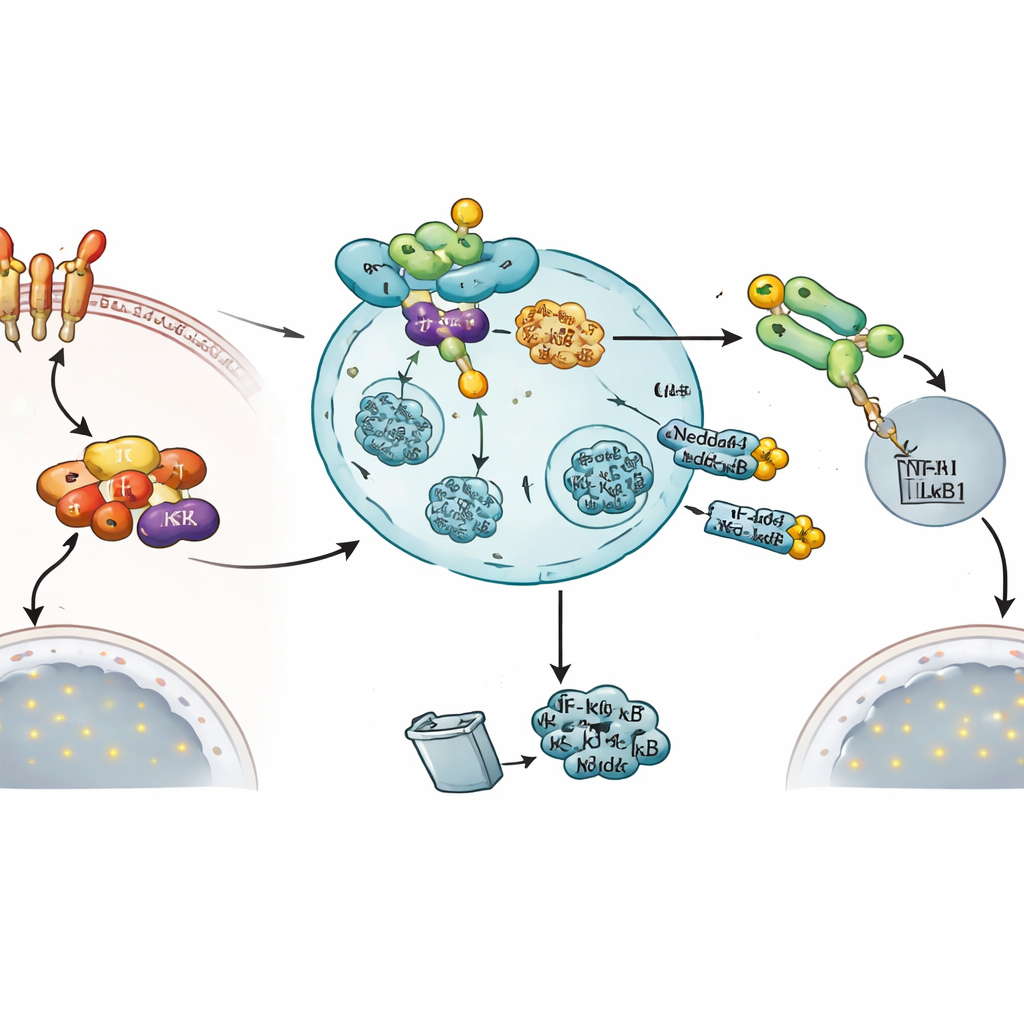
To uncover how this brake works, the authors used proximity labeling and protein pull‑down methods to see which molecules physically associate with Nedd4-1(NE). They identified key components of the NF‑κB pathway, including the kinase IKKβ and the NF‑κB precursor protein NF‑κB1 (also called p105). Follow‑up experiments showed that Nedd4-1(NE) adds ubiquitin tags to IKKβ, leading to its degradation. With less IKKβ around, its usual target IκBα is no longer phosphorylated and destroyed as efficiently. As a result, IκBα remains more stable and keeps the active NF‑κB subunit p50 sequestered in the cytosol, preventing it from entering the nucleus to switch on inflammatory genes.
An adaptor‑guided handoff to the brake
Nedd4-1(NE) also acts further downstream by limiting the levels of NF‑κB1 itself. Here, the enzyme does not work alone: it collaborates with another ubiquitin‑tagging protein called KPC1. KPC1 helps process the NF‑κB1 precursor p105 into the active p50 form and resides in cellular compartments known as late endosomes, where Nedd4-1(NE) is also found. The team showed that Nedd4-1(NE) can ubiquitinate KPC1 and that this modified KPC1 serves as an adaptor, recruiting NF‑κB1 to Nedd4-1(NE) on late endosomes. This three‑way complex promotes the destabilization of both p105 and p50, once again reducing the pool of NF‑κB that can reach the nucleus.
A built‑in feedback loop to end the signal
Strikingly, the very signal that activates NF‑κB also appears to induce this brake. When cells were treated with TNFα, levels of Nedd4-1(NE) messenger RNA rose dramatically, while the standard Nedd4-1 form did not change. Functional readouts supported the idea that Nedd4-1(NE) is a shutoff valve: increasing active Nedd4-1(NE) reduced the movement of NF‑κB1 into the nucleus, lowered the activity of an NF‑κB‑responsive reporter gene, and decreased the expression of an NF‑κB‑regulated inflammatory gene, TNFAIP3. Together, these findings outline a negative feedback loop in which activation of the NF‑κB pathway eventually boosts Nedd4-1(NE), which then targets two critical nodes—IKKβ and NF‑κB1—to bring the inflammatory response back down.
What this means for health and disease
For a general audience, the key message is that our cells incorporate an extra, primate‑specific safety mechanism to keep inflammatory signaling from running amok. By selectively tagging and trimming core components of the NF‑κB system, Nedd4-1(NE) helps terminate the response to TNFα and may contribute to long‑term immune balance. Understanding this built‑in brake could open new avenues for treating diseases where NF‑κB is chronically overactive, such as autoimmune disorders and certain cancers, by suggesting ways to mimic or enhance this natural shutdown process.
Citation: Persaud, A., Kefalas, G., Shteiman, A. et al. Negative regulation of the NF-κB pathway by the ubiquitin ligase Nedd4-1(NE). Commun Biol 9, 374 (2026). https://doi.org/10.1038/s42003-026-09634-7
Keywords: NF-kB signaling, inflammation, ubiquitin ligase, Nedd4-1(NE), TNF alpha