Clear Sky Science · en
Structural study of monomeric and dimeric photosystem I-LHCI supercomplexes from a bryophyte
How Tiny Plants Handle Life on Land
Liverworts are among the earliest plants to venture from water onto land, long before trees or flowers existed. To survive harsh sunlight, drought, and ultraviolet radiation, they had to re‑engineer the tiny solar panels inside their cells. This study peeks into those solar panels at near‑atomic detail, revealing how a primitive land plant, the liverwort Marchantia polymorpha, arranges its light‑harvesting machinery and how that design may have helped plants conquer dry land. 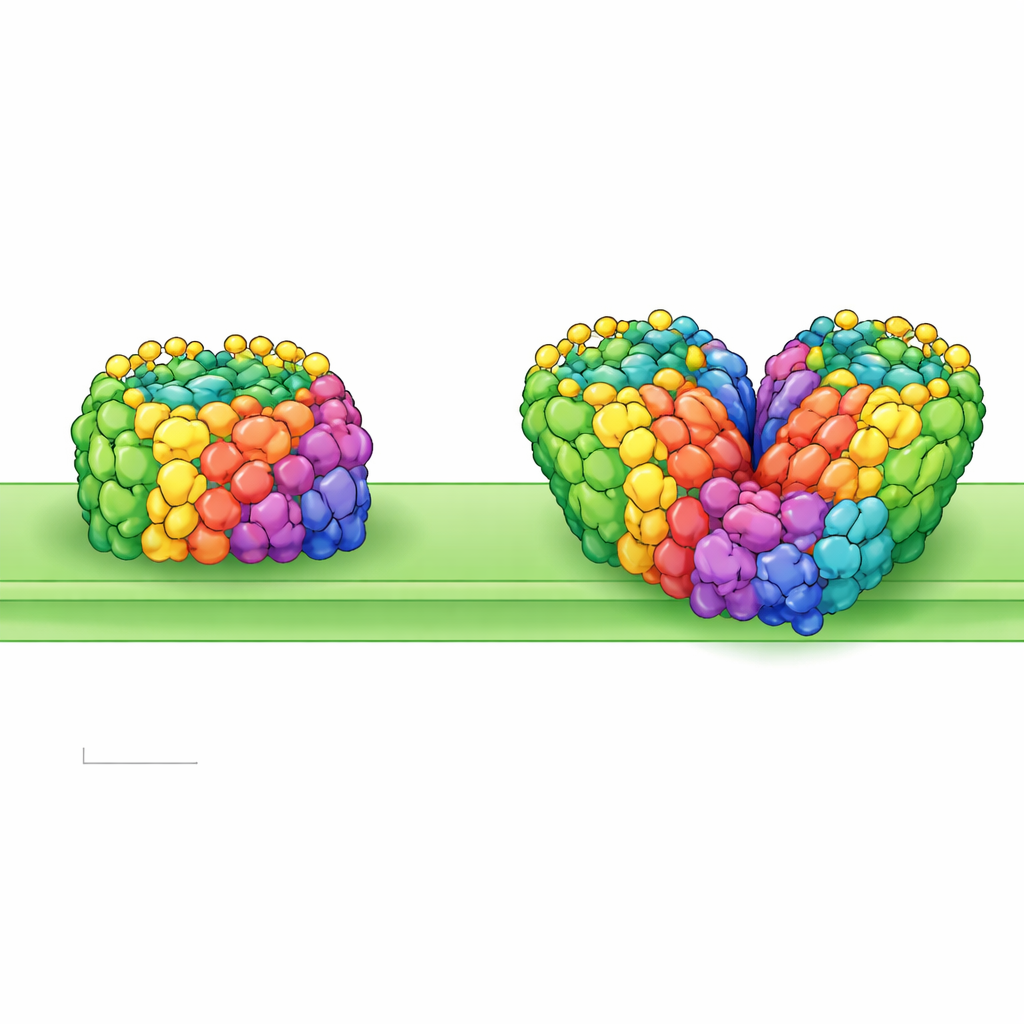
The Solar Engine Inside Green Cells
All green plants rely on microscopic machines called photosystems to turn sunlight into usable energy. One of the most important is Photosystem I, which passes electrons along a chain of pigments and iron–sulfur clusters, ultimately powering the cell’s “chemical battery.” Wrapped around this reaction center are light‑harvesting complexes, which act like antennae to catch extra light and funnel it inward. Together they form a large supercomplex, Photosystem I–LHCI, embedded in internal membranes of the chloroplast. Although this basic layout is shared from bacteria to flowering plants, the number and arrangement of antenna complexes vary by species and environment, hinting that evolution has tinkered with this design to cope with dim forests, bright fields, or aquatic light filtered through water.
Seeing Monomers and Dimers in Extreme Detail
The researchers purified Photosystem I–LHCI from Marchantia thylakoid membranes and imaged the particles using state‑of‑the‑art cryo‑electron microscopy, reaching resolutions fine enough to distinguish individual pigment molecules and even many water molecules. They found two main forms: a single unit, or monomer, and a pair of units stuck together, or homodimer. The monomer contains 13 core protein subunits and four antenna proteins, each loaded with chlorophylls and carotenoids that capture light. Its overall shape closely resembles that of a moss relative, suggesting a conserved blueprint among early land plants. The dimer, however, is more unusual: two complete monomers are joined face‑to‑face but slightly tilted and twisted, making the whole assembly look bent rather than flat. 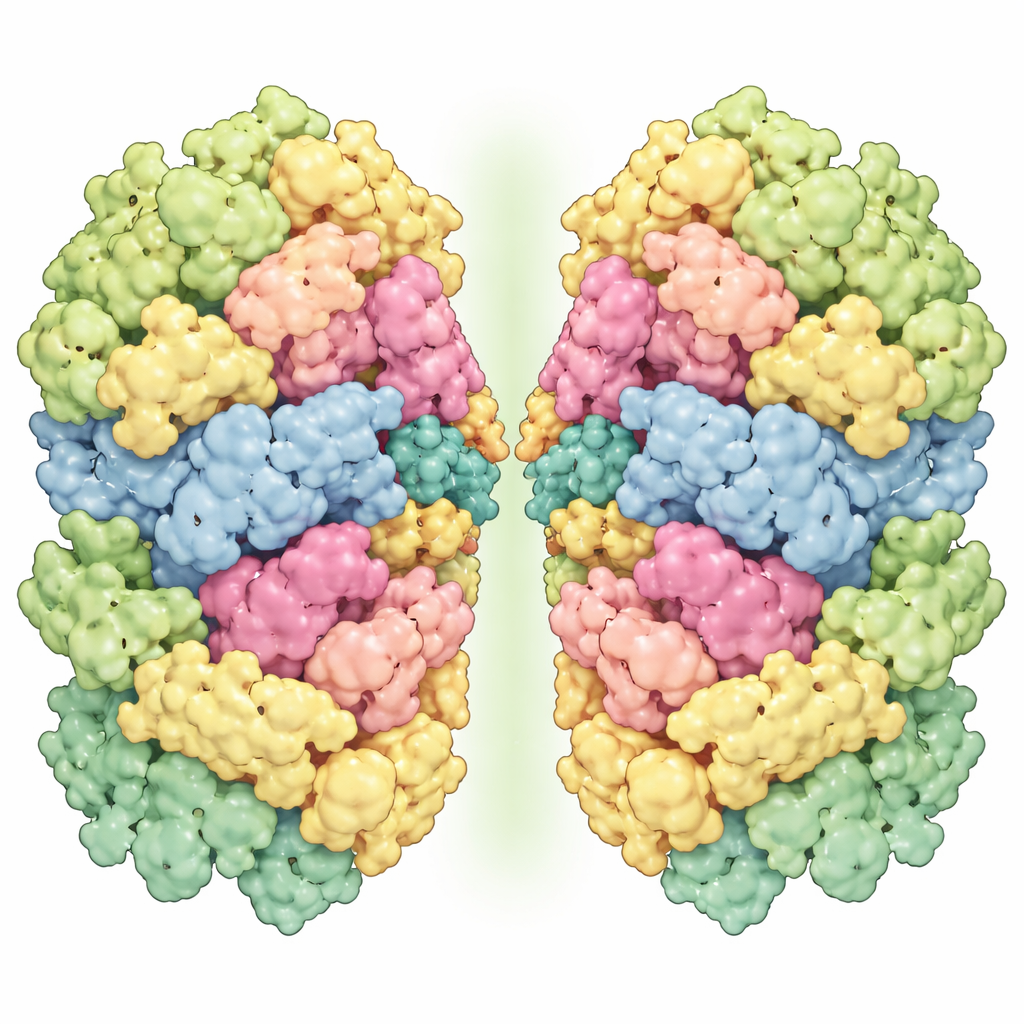
Fine Wiring of Pigments and Energy Flow
With such sharp maps, the team could trace the positions of nearly all the pigments that move energy toward the reaction center. They confirmed the classic chain of cofactors that carry electrons away from the special chlorophyll pair known as P700 and measured tiny differences—fractions of an angstrom—in distances compared with moss. These shifts are within experimental uncertainty but show that even closely related species fine‑tune the local environment around key pigments. The authors also cataloged which chlorophyll molecules sit where within the four antenna proteins and how subtle changes in loop regions of these proteins create or remove pigment‑binding pockets. Using theoretical calculations, they mapped out likely energy‑transfer routes between chlorophylls and showed that, despite the two monomers touching in a dimer, there is no strong pigment‑to‑pigment connection across the interface. In other words, the dimer seems to be a structural arrangement rather than a way to share light energy between partners.
How Two Solar Units Stick Together
The most distinctive feature of the liverwort complex is how its dimers are held together. The contact surface involves several core proteins—PsaB, PsaM, PsaI, PsaG, and PsaH—forming two main interaction zones. One extends from the top to the bottom of the membrane and relies mainly on snug, water‑excluding contacts between PsaM, PsaI, and PsaB, implying that PsaM is crucial for locking the two monomers into a pair. The other zone sits on the stromal side of the membrane, where PsaH reaches across to touch PsaG and nearby pigments on the neighboring monomer, helping to generate the observed tilt and twist. Intriguingly, PsaM is present in cyanobacteria, algae, and bryophytes but missing in flowering plants, while PsaG and PsaH are conserved in green algae and land plants. This pattern suggests that liverworts and mosses retain an older, PsaM‑based way of dimerizing Photosystem I that was later lost or remodeled in more advanced plants.
What This Means for Plant Evolution
By delivering the first high‑resolution structure of a Photosystem I–LHCI dimer from a land plant, this work traces a key step in the evolution of the photosynthetic “hardware.” Liverworts appear to bridge the gap between aquatic green algae, which can form various oligomers of Photosystem I, and vascular plants, where only monomers have been firmly established. The presence of a relatively fragile dimer in Marchantia, stabilized by PsaM and PsaH, supports the idea that dimeric forms were favored in wetter, lower‑light habitats but became unnecessary—or even disadvantageous—as plants adapted to brighter, drier terrestrial conditions. Understanding these structural solutions in a humble liverwort sheds light on how early plants tuned their solar machinery to meet the challenges of life on land.
Citation: Tsai, PC., La Rocca, R., Motose, H. et al. Structural study of monomeric and dimeric photosystem I-LHCI supercomplexes from a bryophyte. Commun Biol 9, 146 (2026). https://doi.org/10.1038/s42003-026-09631-w
Keywords: photosystem I, light harvesting, liverwort, cryo-electron microscopy, plant evolution