Clear Sky Science · en
Notch3 regulates pericyte phenotypic plasticity in colorectal cancer
Why the Blood Vessels Around Tumors Matter
Colorectal cancer is one of the most common and deadly cancers worldwide, yet most therapies are aimed directly at tumor cells and often overlook the surrounding support system that helps tumors grow. This paper focuses on a lesser-known group of support cells called pericytes, which wrap around tiny blood vessels. By uncovering how a signaling switch named Notch3 changes pericyte behavior, the authors show how the tumor’s blood supply can be pushed toward either leaky, chaotic vessels that favor cancer, or more normal vessels that may slow disease and improve treatment. 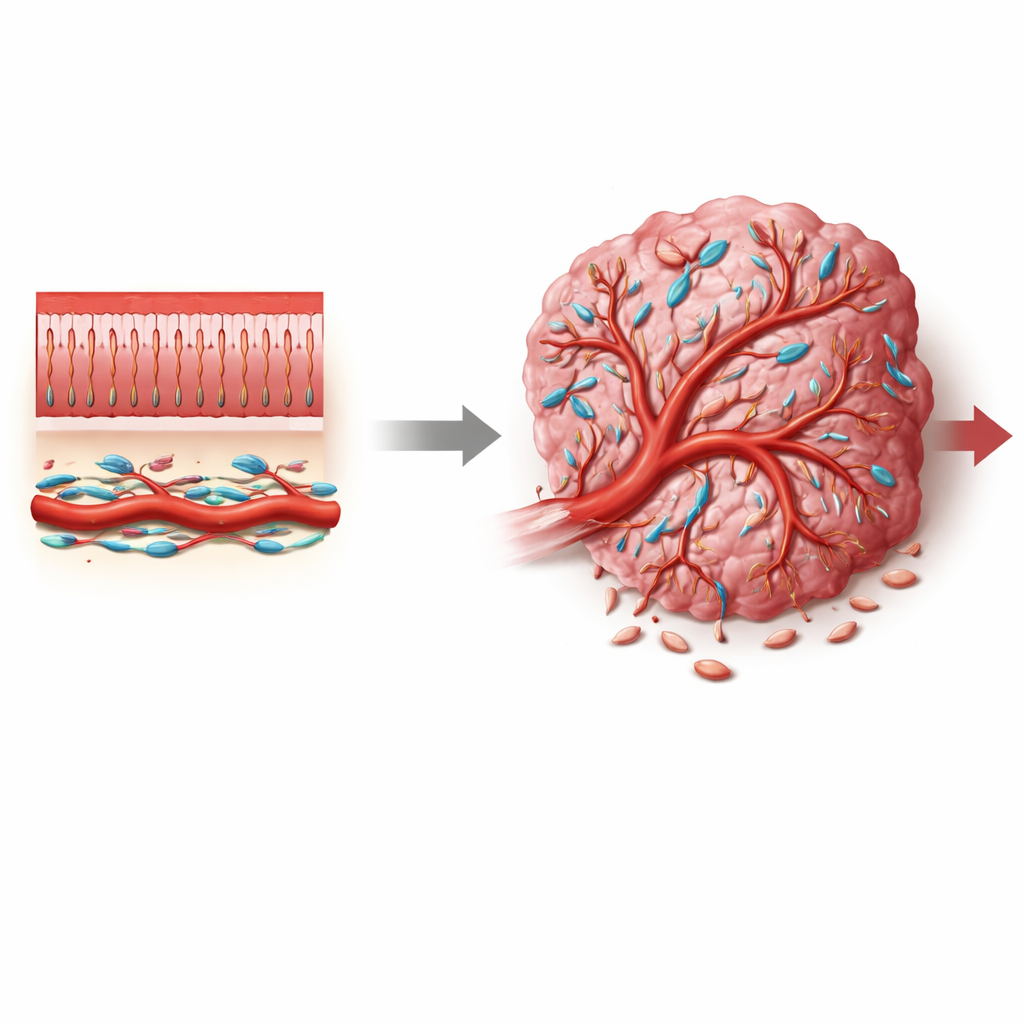
The Hidden Helpers Around Tumor Vessels
Blood vessels are more than just hollow tubes. In healthy tissues, small vessels are tightly hugged by pericytes—specialized cells that stabilize vessel walls, control blood flow, and keep leakage in check. In tumors, however, these pericytes often look and behave differently: they may detach, multiply, or change their identity, contributing to the abnormal, leaky vasculature that is typical of cancers. In colorectal tumors, where the microenvironment is crowded with fibroblasts, immune cells, and vessel cells, it has been unclear where tumor-associated pericytes come from, what states they adopt, and which molecular signals push them toward tumor-promoting roles.
Tracking Pericytes as Tumors Form
To follow pericytes over time, the researchers used mouse models of colitis-associated colorectal cancer in which they could genetically “label” specific cell types and watch their fate. They showed that the pericytes found in tumors largely arise from normal, tissue-resident pericytes that expand and proliferate as cancer develops, rather than arriving from distant sources such as the bone marrow. Using advanced single-cell RNA sequencing, they cataloged tens of thousands of stromal cells from mouse tumors and normal colon and found that pericytes are significantly enriched in the tumor microenvironment. They also identified similar pericyte expansion in several human colorectal cancer datasets, underscoring that this is not just a mouse-specific phenomenon.
A Molecular Switch That Changes Pericyte Behavior
The team then asked which signals shape pericyte behavior inside tumors and found that Notch3—a receptor that enables cells to talk to their neighbors—is particularly active in tumor pericytes. Computational analysis of cell–cell communication suggested that pericytes both send and receive Notch-related signals, especially through partners on blood vessel cells. By artificially turning Notch3 on in pericytes, the researchers saw these cells proliferate more but express fewer contractile proteins typically associated with vessel-stabilizing behavior. Tumor blood vessels in these mice became larger, leakier, and lined with more proliferating endothelial cells, indicating less stable vasculature. Conversely, deleting Notch3 reduced endothelial cell proliferation, shrank vessel size, and lowered leakage, suggesting a shift toward more normalized blood vessels without dramatically changing early tumor number or size. 
Many Faces of Pericytes Inside Tumors
Beyond simple on–off changes, the study revealed that tumor pericytes exist in several distinct states. Some subgroups showed features of highly contractile cells, others produced large amounts of extracellular matrix, some displayed inflammatory traits, and a subset even carried signatures usually seen in endothelial cells. Notch3 activity was not evenly distributed: it was enriched in a “synthetic” or matrix-producing pericyte population and low in the more contractile subset. Trajectory analyses suggested that pericytes can shift from a contractile state toward a synthetic, proliferative one as Notch3 activity rises. When the authors examined human colorectal cancer data, they found a comparable spectrum of pericyte states and again saw higher activity of a Notch3 target gene in tumor-associated and proliferating pericytes, mirroring the mouse results.
Turning Vessel Chaos into a Therapeutic Opportunity
Overall, the work paints Notch3 as a key dial that tunes pericyte identity and, through them, the structure and function of tumor blood vessels. High Notch3 activity pushes pericytes toward a proliferative, less contractile state linked with enlarged, leaky vessels that favor tumor progression. Low Notch3 activity is associated with more contractile pericytes, calmer endothelial cells, and more normalized vasculature that may restrain tumor growth and improve drug delivery. By targeting Notch3 specifically in pericytes, future therapies might steer tumor vessels away from chaos and toward stability, offering a new angle to treat advanced colorectal cancer in combination with existing drugs.
Citation: Chalkidi, N., Stavropoulou, A., Arvaniti, VZ. et al. Notch3 regulates pericyte phenotypic plasticity in colorectal cancer. Commun Biol 9, 343 (2026). https://doi.org/10.1038/s42003-026-09629-4
Keywords: colorectal cancer, tumor microenvironment, pericytes, Notch3 signaling, tumor blood vessels