Clear Sky Science · en
Endothelial TRIM47 regulates blood-brain barrier integrity and cognition via the KEAP1/NRF2 signalling pathway in mice
Why protecting tiny brain vessels matters
Cerebral small vessel disease is a quiet troublemaker in the brain. It damages the tiniest blood vessels, raises the risk of stroke, and slowly erodes memory and thinking, yet doctors still lack treatments that tackle its root biological causes. This study digs into one specific protein, TRIM47, found in the cells lining brain blood vessels, to ask a simple but crucial question: does it help keep the brain’s protective blood–brain barrier intact, and does that matter for cognition?
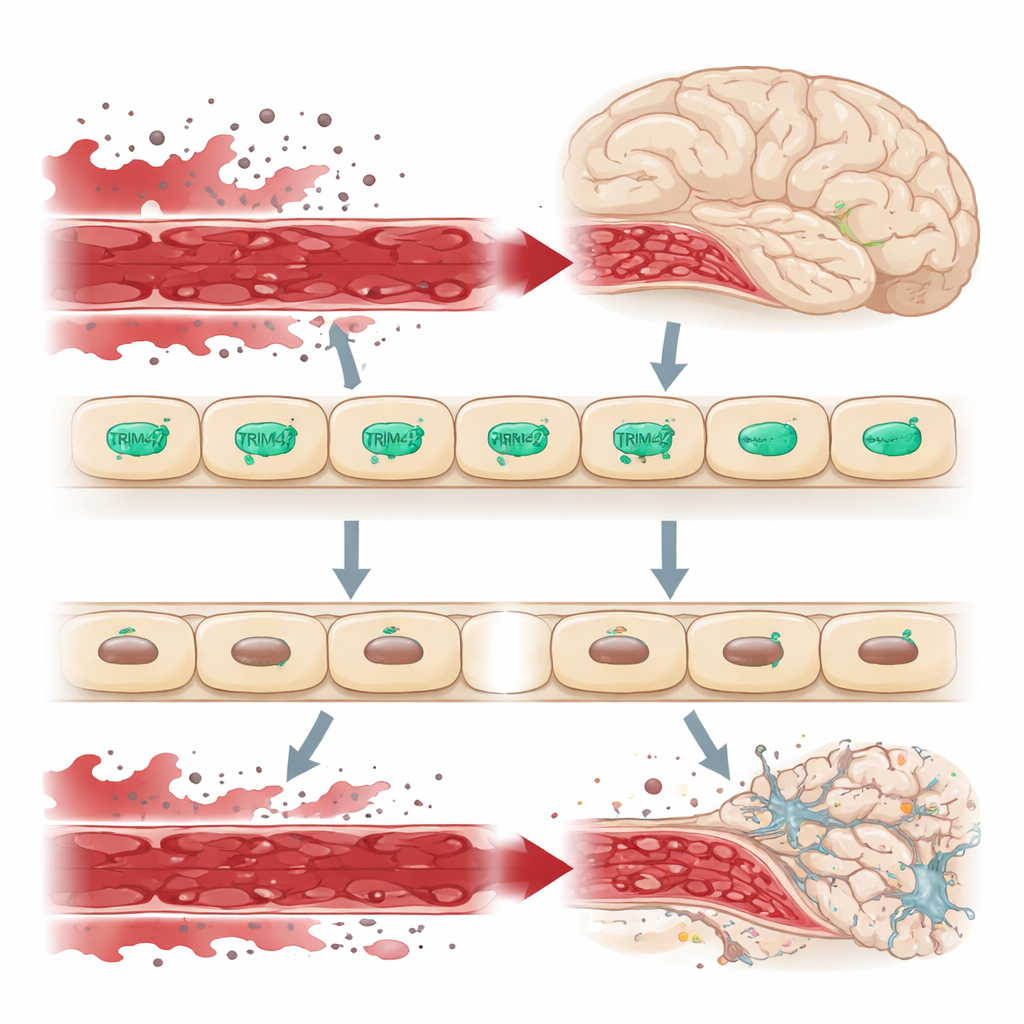
A gatekeeper at the brain’s border
The brain is shielded from the rest of the body’s circulation by the blood–brain barrier, a tightly sealed lining of endothelial cells that strictly controls what can enter brain tissue. When this barrier leaks, harmful molecules from the blood seep into the brain, stressing nerve cells and supporting cells and contributing to conditions like dementia. Genetic studies in large human populations had pointed to a stretch of DNA containing the TRIM47 gene as a risk region for cerebral small vessel disease. TRIM47 is especially active in brain endothelial cells, hinting that it might be an important guardian of this barrier.
How TRIM47 arms cells against oxidative stress
The authors first studied human brain endothelial cells grown in the lab and reduced TRIM47 levels using RNA interference. This triggered broad changes in gene activity, with the most strongly affected network centered on NRF2, a master regulator of the cell’s antioxidant defenses. Under normal conditions, NRF2 is held in check by another protein, KEAP1, which directs it toward destruction. The team showed that TRIM47 binds to KEAP1 and helps prevent NRF2 from being broken down. With TRIM47 present, NRF2 can accumulate, move into the nucleus, and switch on genes that detoxify reactive oxygen species. When TRIM47 is lost, NRF2 levels fall, antioxidant genes are less active, and endothelial cells become more vulnerable to oxidative stress.
From leaky vessels to memory problems in mice
To see how this mechanism plays out in a living brain, the researchers engineered mice lacking the Trim47 gene throughout the body, and another line in which Trim47 could be deleted only in endothelial cells. Adult animals of both models developed clear problems in spatial learning and memory, failing tasks such as the Y-maze and Morris water maze despite normal movement. Examination of their brains revealed that the blood–brain barrier had become leaky: small fluorescent tracers and blood proteins escaped from vessels into surrounding tissue. Key “sealing” proteins that form tight and adherens junctions between endothelial cells, especially Claudin-5 and Occludin, were reduced. Nearby astrocytes, the star-shaped support cells that react quickly to injury, became activated, even though there was little sign of broad inflammation or nerve cell loss at this stage.
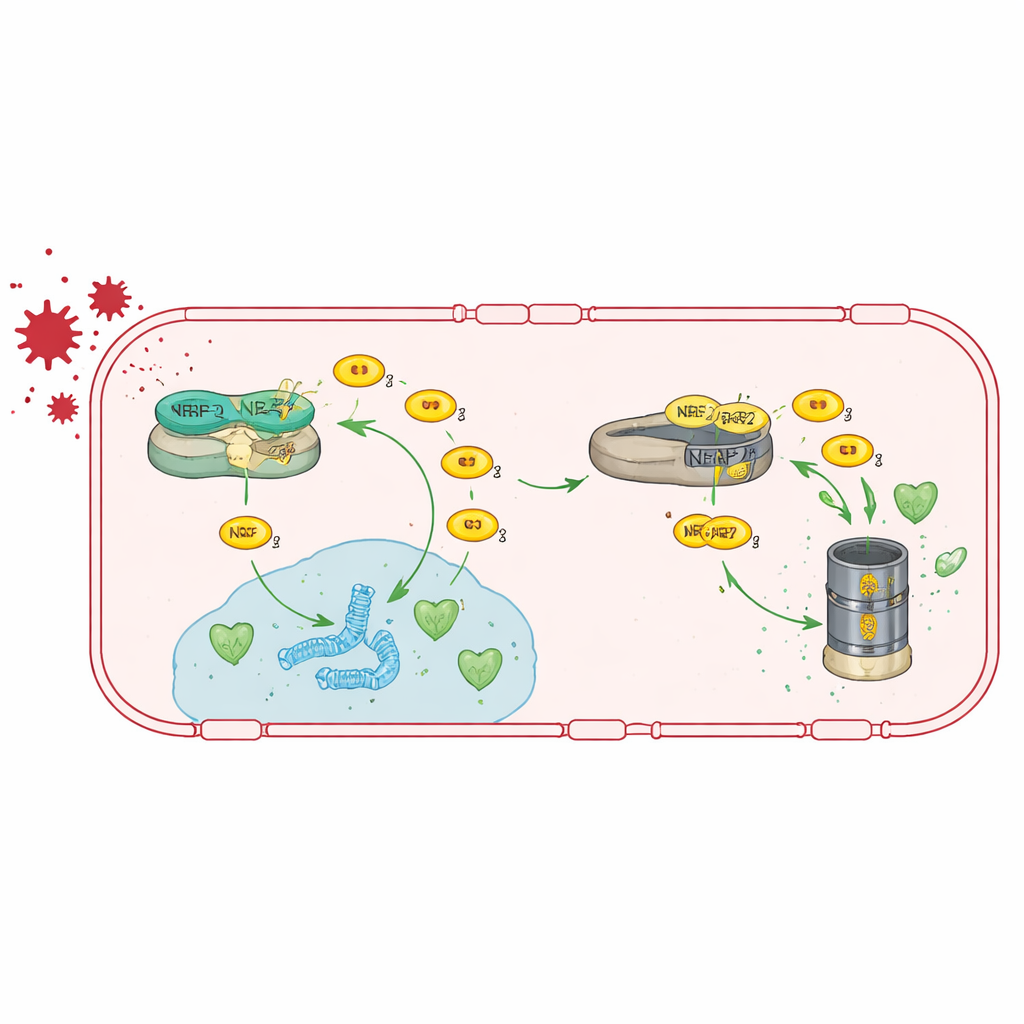
Switching the antioxidant pathway back on
Because TRIM47 works mainly by boosting NRF2 activity, the team tested whether they could bypass its loss by directly stimulating NRF2. They fed Trim47-deficient mice a diet containing tert-butylhydroquinone (tBHQ), a compound known to stabilize NRF2 and turn on its target genes. This treatment restored antioxidant gene activity in brain endothelial cells, brought tight junction gene expression back toward normal, and reduced blood–brain barrier leakage. Strikingly, it also normalized astrocyte activation and rescued the animals’ performance in memory tests, suggesting that reinforcing this antioxidant pathway can reverse both vascular and cognitive problems, at least in mice.
Clues from human blood and what comes next
To bridge their mouse experiments to human disease, the researchers analyzed proteins related to the NRF2 pathway in blood samples from thousands of people who had brain MRI scans. Levels of several NRF2-regulated proteins were linked to imaging markers of small vessel disease, such as enlarged perivascular spaces and changes in white matter. This supports the idea that altered TRIM47–NRF2 signaling is relevant in people, not just in experimental animals, and that these proteins might serve as biomarkers of disease severity or progression.
What this means for brain health
Taken together, the work portrays TRIM47 as a molecular bodyguard that helps brain blood vessels withstand oxidative damage by stabilizing the NRF2 antioxidant system. When TRIM47 is missing or impaired, the blood–brain barrier weakens, subtle brain injury accumulates, and memory starts to fail. Reactivating the NRF2 pathway can patch the barrier and restore cognition in mice, highlighting this signaling axis as a promising therapeutic target for vascular forms of dementia. While tBHQ itself is not suitable as a drug for humans, safer NRF2-boosting compounds or strategies that mimic TRIM47’s protective role may one day help preserve brain health by strengthening its smallest, most fragile vessels.
Citation: Delobel, V., Grenier, C., Boulestreau, R. et al. Endothelial TRIM47 regulates blood-brain barrier integrity and cognition via the KEAP1/NRF2 signalling pathway in mice. Commun Biol 9, 399 (2026). https://doi.org/10.1038/s42003-026-09628-5
Keywords: cerebral small vessel disease, blood–brain barrier, TRIM47, NRF2 pathway, vascular dementia