Clear Sky Science · en
An inter-subunit path is required for entropically-driven and negatively cooperative binding of cyclic nucleotides in the HCN2 channel
How the Heart’s Tiny Pacemaker Switches Sense Chemical Signals
Every heartbeat and many brain rhythms depend on microscopic channels that open and close in our cells’ membranes. These channels, called HCN pacemaker channels, are controlled not only by electrical voltage but also by small messenger molecules known as cyclic nucleotides. This study asks a deceptively simple question with far‑reaching implications: how do these tiny molecules bind to the HCN2 channel in a coordinated way, and how can subtle faults in that process contribute to disorders such as epilepsy?
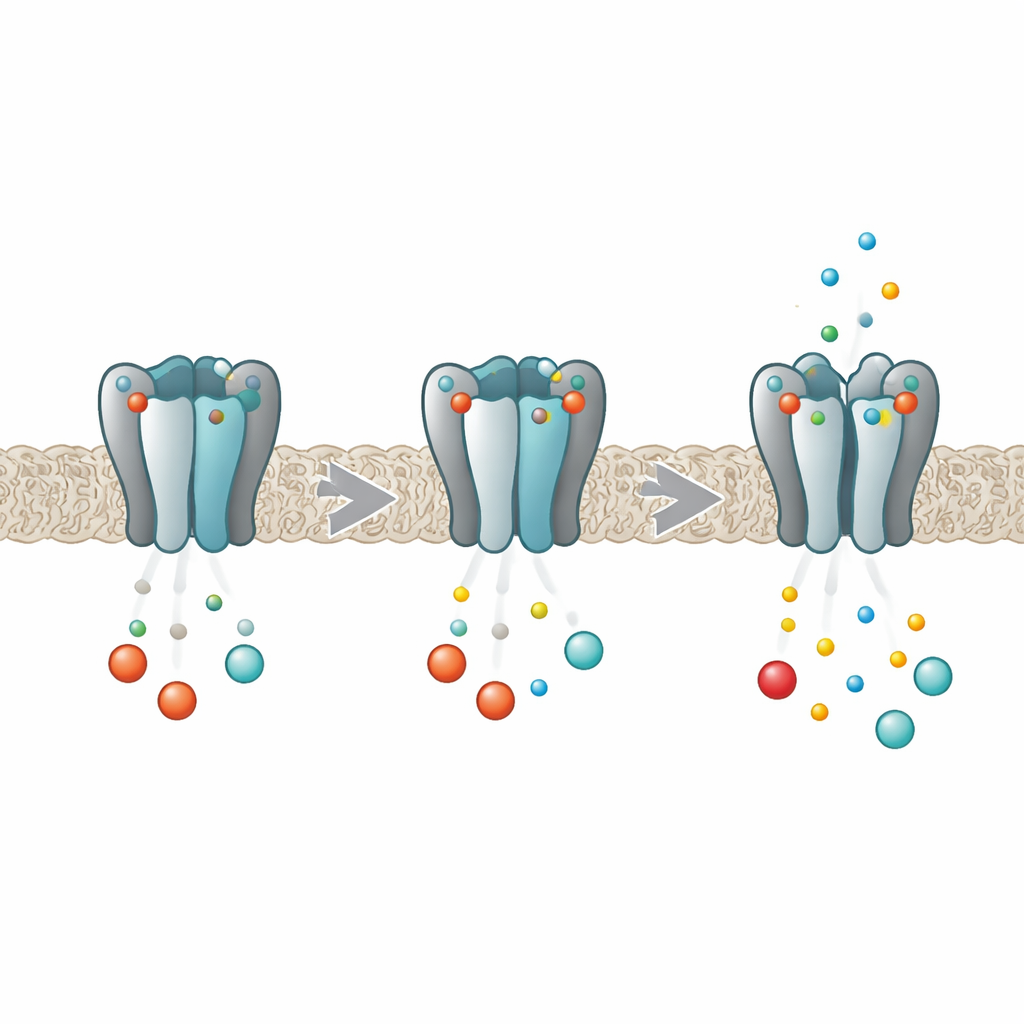
A Molecular Dimmer for the Body’s Rhythms
HCN channels act like dimmer switches for electrical activity in heart and brain cells. Each channel is built from four identical protein subunits that together form a central pore through which ions flow. Deep inside each subunit sits a pocket that can grab cyclic nucleotides such as cAMP and cGMP. When these messengers bind, they make it easier for the pore to open, boosting the pacemaker current. Earlier work showed that in the HCN2 variant, binding does not happen independently at each pocket; instead, the four sites influence one another so that the first molecule binds more readily than later ones, a behavior known as negative cooperativity. The new work set out to uncover exactly how the four subunits talk to each other during this process.
Subunits That Feel Their Neighbors
The researchers combined computer simulations with laboratory measurements on isolated pieces of the HCN2 channel. Using long molecular dynamics simulations, they watched how the channel’s tail region—the part that contains the cyclic nucleotide pockets and the so‑called C‑linker that connects to the pore—flexes and wobbles as different numbers of cAMP or cGMP molecules are bound. They found that when a messenger locked into one subunit, it did not simply stabilize that subunit alone. Instead, it subtly changed the stability of its neighbors, sometimes making them more rigid, sometimes more flexible, even though the overall protein shape barely shifted. These neighbor‑to‑neighbor influences depended on how many sites were occupied and where those sites sat around the ring, matching what would be expected from negative cooperativity.
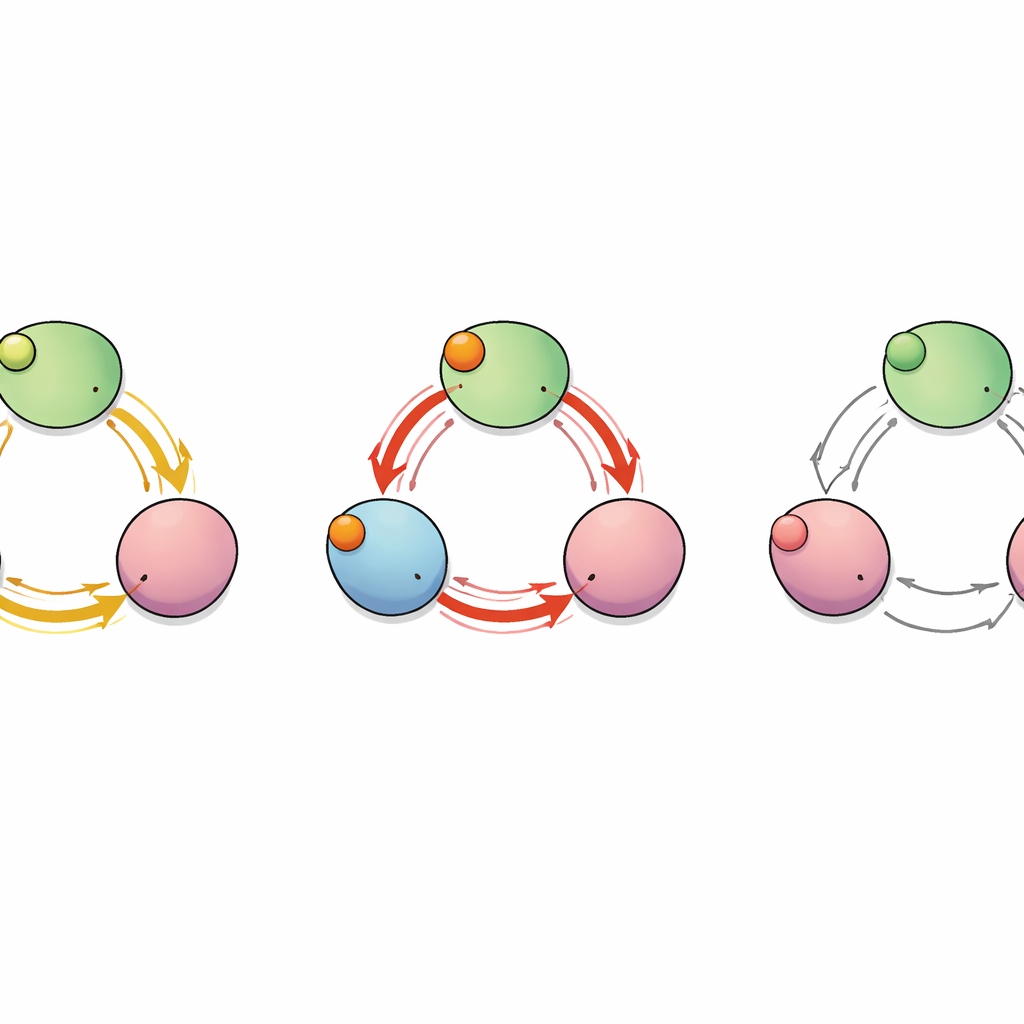
A Hidden Pathway Between Subunits
To map how the “signal” of binding travels through the protein, the team treated the channel as a network of interacting amino acids. They calculated which residues moved in a correlated way during the simulations and used graph‑theory tools to identify the shortest and most efficient communication paths from the binding pockets to the C‑linker segments that control the pore. This analysis revealed several possible routes, but two stood out: one that stays within a single subunit and another that jumps from one subunit to its neighbor. Along this inter‑subunit route, a single residue—glutamate at position 488 (E488)—acted as a critical hub through which many of the shortest paths passed, suggesting it could be a key relay point for cooperative binding.
Mutations That Break the Conversation
The authors then turned to experiments with purified channel tails to test the importance of this pathway. They altered E488 and nearby tyrosine 459 (Y459), which lies on the neighboring subunit where the path arrives. Using dynamic light scattering, they confirmed that the mutant proteins still assembled into four‑subunit complexes, but they no longer showed the strong ligand‑induced tightening between subunits seen in the normal protein. Isothermal titration calorimetry, which tracks the tiny heats of binding, revealed an even bigger change: wild‑type HCN2 displayed two distinct binding events, one high‑affinity step driven mainly by an increase in entropy (greater internal motion), followed by three lower‑affinity steps. In contrast, mutants at E488 or Y459 showed only a single, modest‑affinity binding mode that lacked this favorable entropic boost and lost the hallmark negative cooperativity.
Why Subtle Entropy Changes Matter
Putting these pieces together, the study paints a picture of the HCN2 channel as a dynamic ring of four subunits connected by specific “conversation paths.” When the first cyclic nucleotide binds, it tweaks the motions of distant parts of the ring through E488 and Y459, making later binding events less favorable and tuning how the pore responds. This effect is driven not by large structural rearrangements but by shifts in internal flexibility—changes in entropy that are invisible in static snapshots yet crucial for function. Mutations that disrupt the key residues along this inter‑subunit path silence the conversation, abolish negative cooperativity, and are linked to diseases such as idiopathic generalized epilepsy. For a lay reader, the takeaway is that life‑critical switches in the heart and brain rely on exquisitely choreographed molecular “whispers” between protein subunits, and understanding these whispers helps explain both normal rhythms and what happens when they go awry.
Citation: Costa, F., Ng, L.C.T., Chow, S.S. et al. An inter-subunit path is required for entropically-driven and negatively cooperative binding of cyclic nucleotides in the HCN2 channel. Commun Biol 9, 362 (2026). https://doi.org/10.1038/s42003-026-09626-7
Keywords: HCN2 ion channel, cyclic nucleotide binding, negative cooperativity, allosteric communication, epilepsy mutation