Clear Sky Science · en
A small molecule allosterically activates SecA dependent secretion
Turning Bacteria’s Shipping Dock into Overdrive
Bacteria survive by constantly exporting proteins through tiny gateways in their membranes. These exports help them build cell walls, secrete toxins, and adapt to stress—processes at the heart of many infections. This study reports a small chemical, called HSI#6, that does something unusual: instead of blocking this export machinery, it flips it into a hyperactive, less selective mode. Understanding how this switch works could both reveal new antibacterial strategies and offer biotech a way to boost protein secretion on demand.
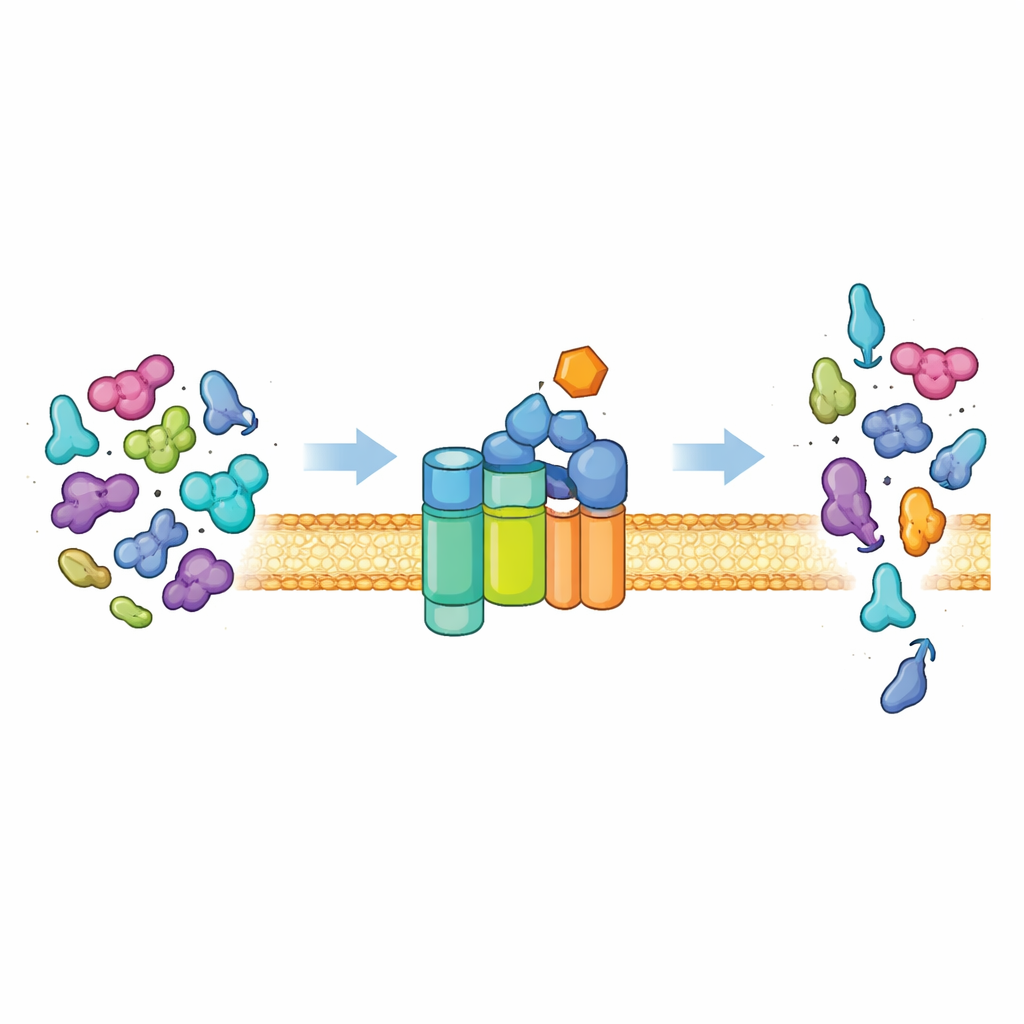
The Cell’s Protein Exit Gate
Most bacteria ship proteins out of the cell using a universal pathway known as the Sec system. At its core is a membrane channel (SecYEG) and a motor protein (SecA) that burns cellular fuel (ATP) to push unfolded protein chains through the channel. Under normal conditions, this system is picky. It primarily exports “preproteins” that carry special address tags, known as signal peptides, plus additional features in their main body. These features are needed to switch SecA from a quiet, low-activity state into a powerful motor. Only proteins that present the right combination of signals can unlock full activity, ensuring that the cell exports the right cargo and keeps ordinary cytoplasmic proteins safely inside.
A Small Molecule that Wakes Up the Motor
The researchers discovered HSI#6 in a screen for compounds that interfere with bacterial protein secretion and growth. Biochemical tests showed that HSI#6 binds directly to SecA with micromolar strength and changes how efficiently it uses ATP. In solution, it makes SecA hold onto ATP more tightly; when SecA is attached to the channel, HSI#6 both improves ATP binding and speeds up ATP turnover. This pattern is the hallmark of an allosteric activator: a molecule that binds at one site on a protein but alters activity at another, far-away site. Unlike the natural protein clients, HSI#6 does not need to act as a cargo to stimulate the motor—it simply tunes SecA’s internal dynamics so that the entire translocase becomes catalytically primed.
From Selective Filter to Promiscuous Nanomachine
Once HSI#6 is bound, the Sec system no longer insists on the usual export tags. In test-tube experiments, the compound roughly doubled secretion of a standard preprotein and made the complex more resistant to sodium azide, a known SecA inhibitor. More strikingly, HSI#6 allowed the normal, “wild-type” machinery to move proteins that completely lack signal peptides, as well as proteins that normally reside in the cytoplasm. In living bacterial cells, the same effect was seen using alkaline phosphatase–based reporters: when cytoplasmic proteins were fused to a secreted enzyme, the presence of HSI#6 caused a sharp rise in secreted activity without changing how much protein was made. In other words, HSI#6 effectively decouples export from the usual client identity checks, turning the Sec system into a broadly acting export pump for any sufficiently unfolded protein.
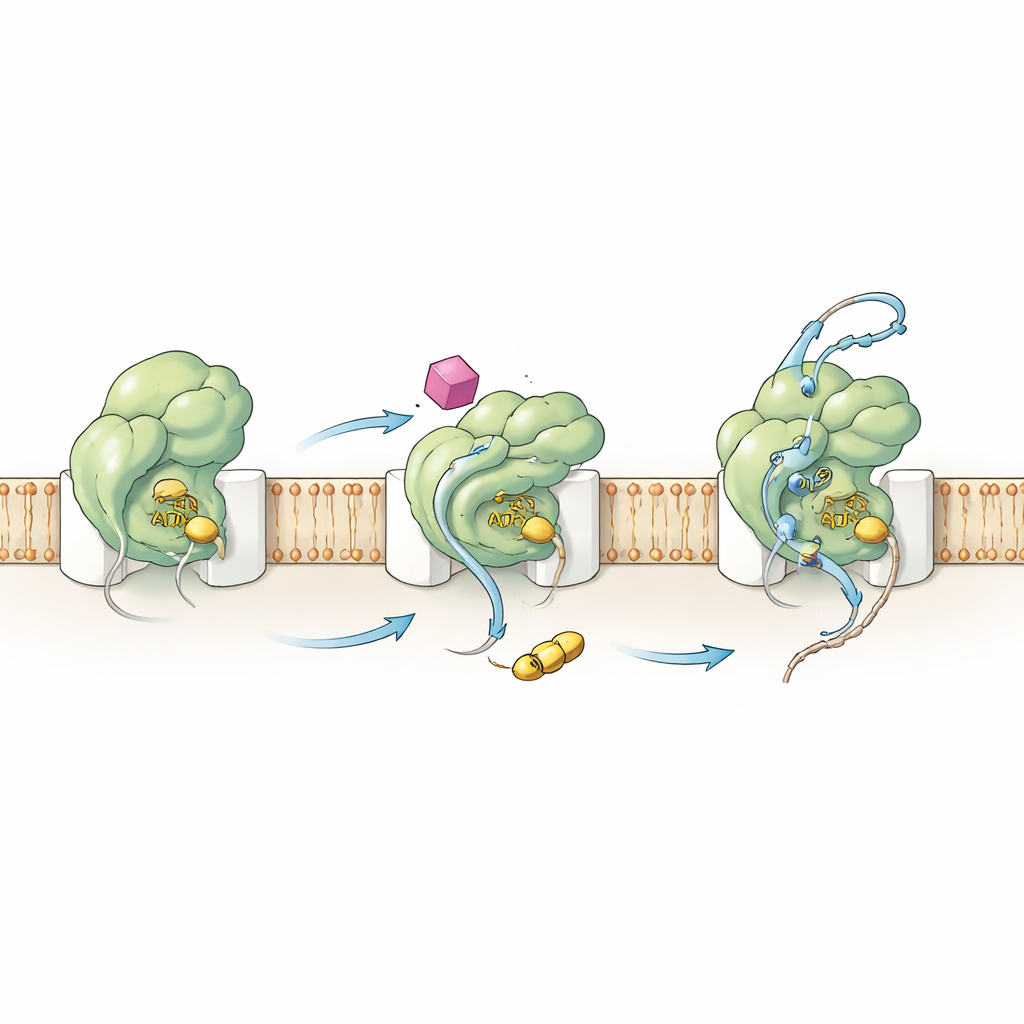
Rewiring the Motor from Within
To understand how HSI#6 triggers this behavior, the team examined both the chemistry and the motions of SecA. Kinetic measurements showed that the compound shifts the key activation step upstream: the SecA–channel complex becomes catalytically active even before a protein client binds. HSI#6 also helps SecA release spent fuel (ADP), a normally slow, rate-limiting step, and thereby supports faster ATP cycling. Using hydrogen–deuterium exchange mass spectrometry, the authors mapped how HSI#6 reshapes the flexibility of different SecA regions. The compound selectively loosens parts of the protein that sense clients and connect to the channel, while stiffening a scaffold region that likely hosts its binding site. The resulting motion pattern resembles an already-engaged, “on” state of the translocase, suggesting that HSI#6 stabilizes an activated form—possibly even favoring a monomeric SecA bound to the channel—without any client present.
Why This Matters for Medicine and Biotechnology
By showing that a small molecule can allosterically activate SecA and strip away its normal client selectivity, this work reveals a new way to control a central bacterial machine. For infectious disease, such forced, uncontrolled secretion could weaken pathogens by draining energy and disrupting their finely tuned export programs, making HSI#6-like activators attractive starting points for antibiotic design—or as guides for crafting future inhibitors that block the same regulatory features. For biotechnology, the same principle could be repurposed to enhance secretion of useful recombinant proteins, without the need for engineered signal peptides or complex genetic tweaks. In essence, the study demonstrates that the bacterial protein “shipping dock” can be reprogrammed chemically, opening new avenues for both combating microbes and harnessing them as microscopic factories.
Citation: Sedky, H., Hamed, M.B., Krishnamurthy, S. et al. A small molecule allosterically activates SecA dependent secretion. Commun Biol 9, 368 (2026). https://doi.org/10.1038/s42003-026-09623-w
Keywords: bacterial protein secretion, SecA translocase, allosteric activation, small molecule regulators, antibacterial drug discovery