Clear Sky Science · en
A histidine pseudokinase modulates polar growth and cell shape in Streptomyces venezuelae
How Bacteria Build Delicate Branching Bodies
Filamentous bacteria of the genus Streptomyces live in soil and form branching networks of threads that resemble microscopic fungi. These bacteria are a major natural source of antibiotics, and their success depends on how precisely they control where and how their cells grow. This study uncovers a previously unknown protein that helps Streptomyces keep their growing tips stable and their branches orderly, offering new insight into how complex cell shapes are built and maintained.
Growing Only at the Tips
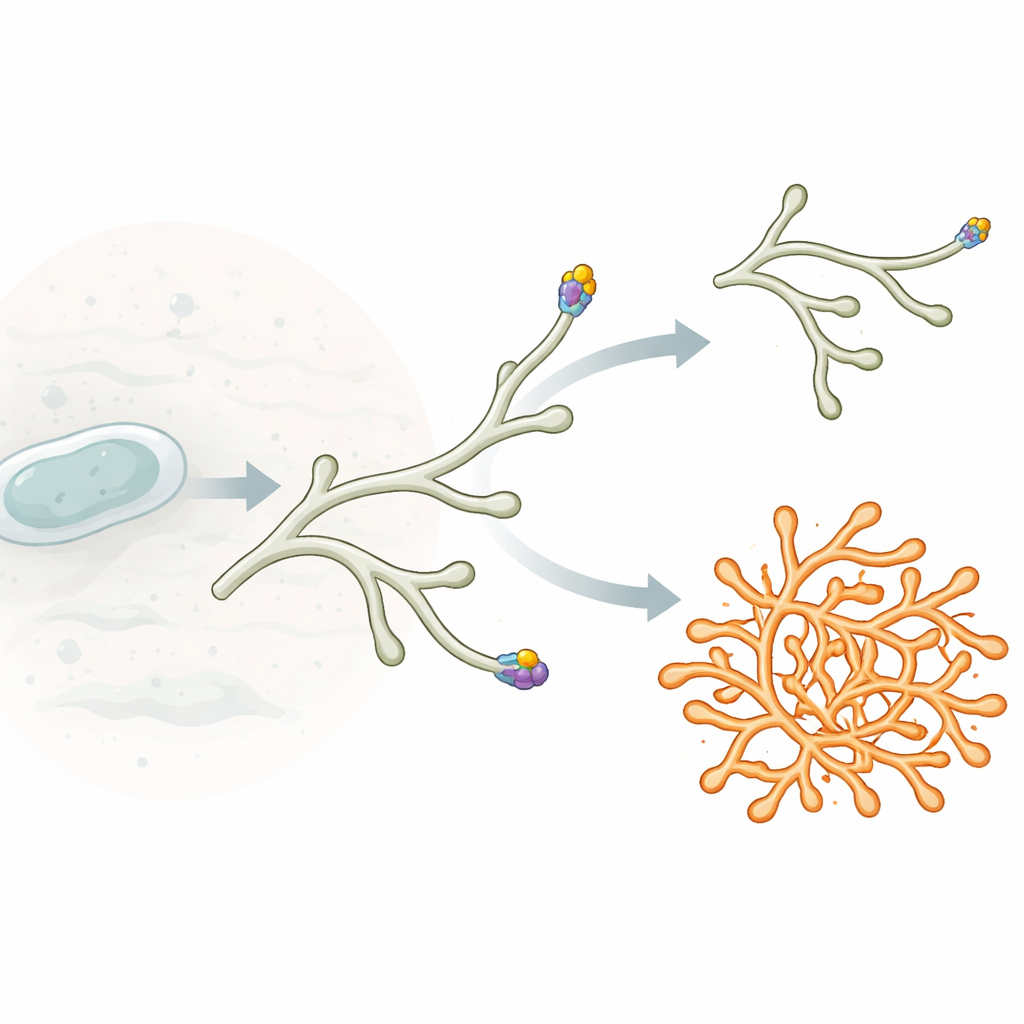
Unlike many familiar rod-shaped bacteria that expand along their entire length, Streptomyces grow mainly at their tips. Each thread, or hypha, extends by adding new cell wall material only at a small zone at the very end. This growth zone is organized by a protein assembly called the polarisome. At its heart lies a protein named DivIVA, which tends to gather at curved cell ends and marks where new wall material should be inserted. When small DivIVA clusters bud off from the main tip and settle along the side of a hypha, they can mature into new growth zones, giving rise to lateral branches and a finely branched mycelial network.
A New Shape Controller at the Tip
The authors set out to search for previously unknown components of the Streptomyces polarisome. By pulling down DivIVA from cell extracts and seeing what came along with it, they discovered a large protein, now named PsmA (for polar growth and shape modulator A). PsmA resembles a common bacterial signaling enzyme called a histidine kinase in its overall domain layout, with sensor-like regions, a central catalytic core and a receiver domain connected by a long flexible segment. However, closer analysis showed that PsmA is missing key amino acids needed to transfer phosphate groups and does not show detectable kinase activity, marking it as a “pseudokinase” that likely acts more as a structural or scaffolding partner than as a classic enzyme.
When the Shape Controller Is Missing
To probe PsmA’s role, the researchers deleted its gene in Streptomyces venezuelae. Colonies of the mutant were smaller and denser, with a pitted surface. Under the microscope, the vegetative hyphae were thicker, more irregular and showed dramatically increased branching at their tips. Instead of extending smoothly and occasionally producing side branches, many tips split into two or more growing ends, creating a hyperbranched, compact mycelium. Importantly, the ability to form aerial filaments and spores remained largely intact, indicating that PsmA mainly acts during vegetative tip growth rather than during the later sporulation stage.
Keeping the Growth Zone in One Piece
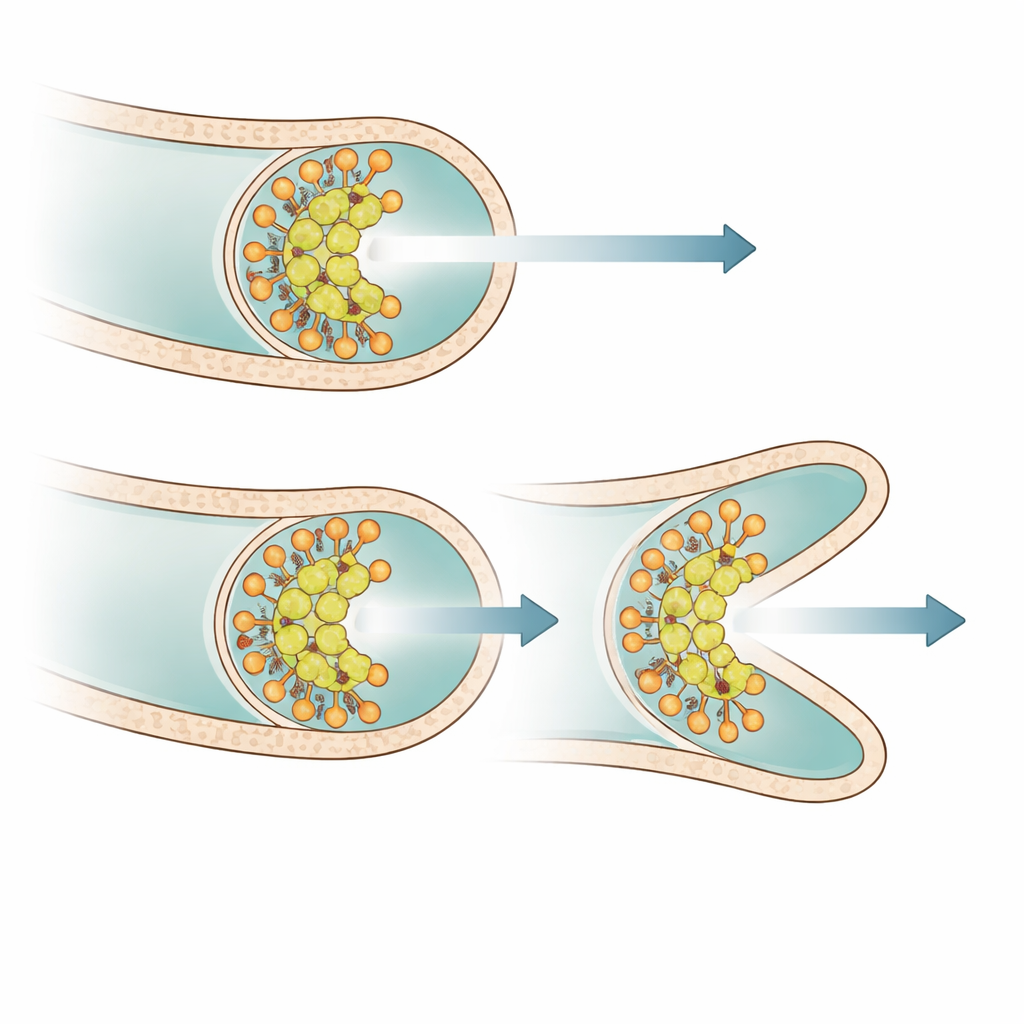
By tagging proteins with fluorescent markers, the authors visualized where PsmA sits in living cells. PsmA formed focused dots right at the very apex of growing hyphae, closely overlapping with DivIVA and another tip protein, Scy, but distinct from FilP, which sits just behind the tip. In the absence of PsmA, the DivIVA clusters at hyphal ends became broader and more irregular in shape. Time-lapse imaging revealed that these distorted clusters were much more prone to split into two similarly sized parts. Each part then drove growth of its own tip, leading to bifurcations very close to the original tip and explaining the hyperbranching pattern. When PsmA production was switched back on in the mutant, DivIVA clusters quickly narrowed, tip shape became smoother, and excessive splitting diminished.
Working in Parallel with Other Tip Factors
PsmA is not the only protein that helps stabilize Streptomyces tips. Earlier work had identified Scy and FilP, two elongated, coiled-coil proteins that associate with DivIVA and influence tip behavior. Double mutants lacking both PsmA and either Scy or FilP showed even more severe growth defects and denser, tangled mycelia than any single mutant, while still remaining viable. This pattern suggests that PsmA acts largely in parallel to Scy and FilP: all three contribute in partly independent ways to keeping the growth zone cohesive and preventing it from fragmenting into multiple competing tips.
What This Means for Bacterial Architecture
Taken together, the findings present PsmA as a non-enzymatic partner that fine-tunes the stability and dynamics of the DivIVA-based polarisome at Streptomyces hyphal tips. Rather than switching genes on and off through classical signaling pathways, PsmA appears to work directly at the growing end, likely as a structural organizer that helps DivIVA clusters maintain a focused, single growth zone. When PsmA is missing, the tip machinery becomes too unstable, splits too often, and produces excessive branching. Understanding this type of local architectural control not only deepens our basic knowledge of bacterial cell biology, but may also inform efforts to manipulate Streptomyces growth patterns for biotechnology and antibiotic production.
Citation: Singh Mavi, P., Flärdh, K. A histidine pseudokinase modulates polar growth and cell shape in Streptomyces venezuelae. Commun Biol 9, 345 (2026). https://doi.org/10.1038/s42003-026-09620-z
Keywords: Streptomyces, cell polarity, bacterial morphogenesis, pseudokinase, hyphal branching