Clear Sky Science · en
Nighttime-restricted feeding disrupts spermatogenesis in mice via gut microbiota-derived KYNA homeostasis
Why When You Eat Might Matter for Future Fathers
Many people have heard that skipping late-night snacks or trying intermittent fasting can be good for weight and blood sugar. But this mouse study asks a different question: could limiting eating to certain hours actually harm male fertility if the timing clashes with the body’s internal clock? By tracking how feeding schedules reshape gut bacteria and a little-known chemical messenger, the researchers uncover a chain of events that ends in fewer, poorer-quality sperm.
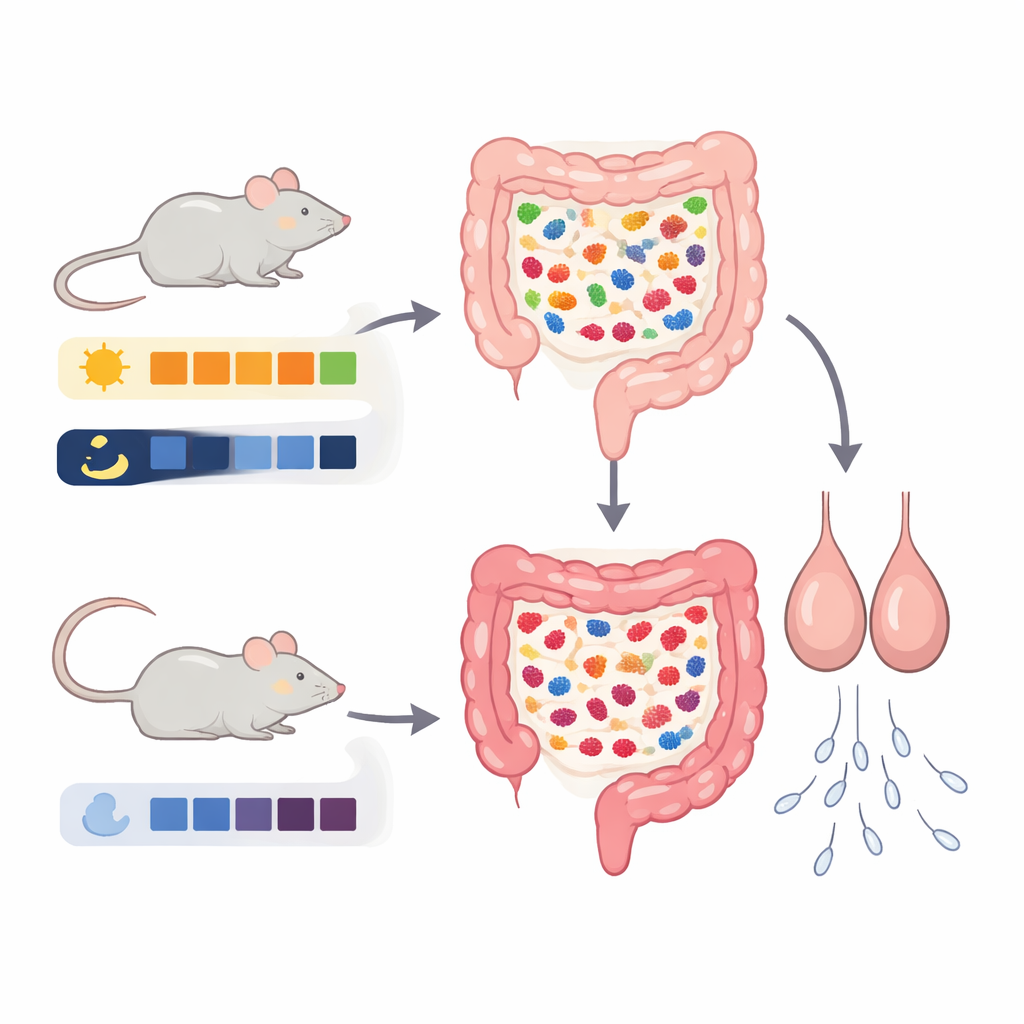
Feeding Windows and the Body’s Inner Clock
The team worked with male mice and set up three eating patterns. One group ate whenever they wanted. A second group could eat only in the daytime, and a third group could eat only at night. Importantly, these mice were not put on a diet; they could eat as much as they liked, just at different times. After four weeks, mice that ate only at night showed smaller testes and poorer sperm concentration and movement, even though they were not eating less overall. Stress-related hormone levels changed, but the usual sleep hormone stayed about the same, hinting that the harm came more from how metabolism and stress responded to mistimed meals than from a simple disruption of the brain’s master clock.
What Was Going Wrong Inside the Testes
Looking closely at testicular tissue under the microscope, the scientists found that night-fed mice had fewer developing sperm cells partway through their maturation process, while earlier stem cells and support cells were largely unchanged. Measures of chemical damage linked to oxidative stress were higher, suggesting that the testicular environment had become harsher and less balanced. Interestingly, the very final steps of sperm shaping—the formation of the acrosome structure on the sperm head—were not strongly affected. This points to the early and middle stages of sperm production as the weak points when feeding is misaligned with the body’s rhythms.
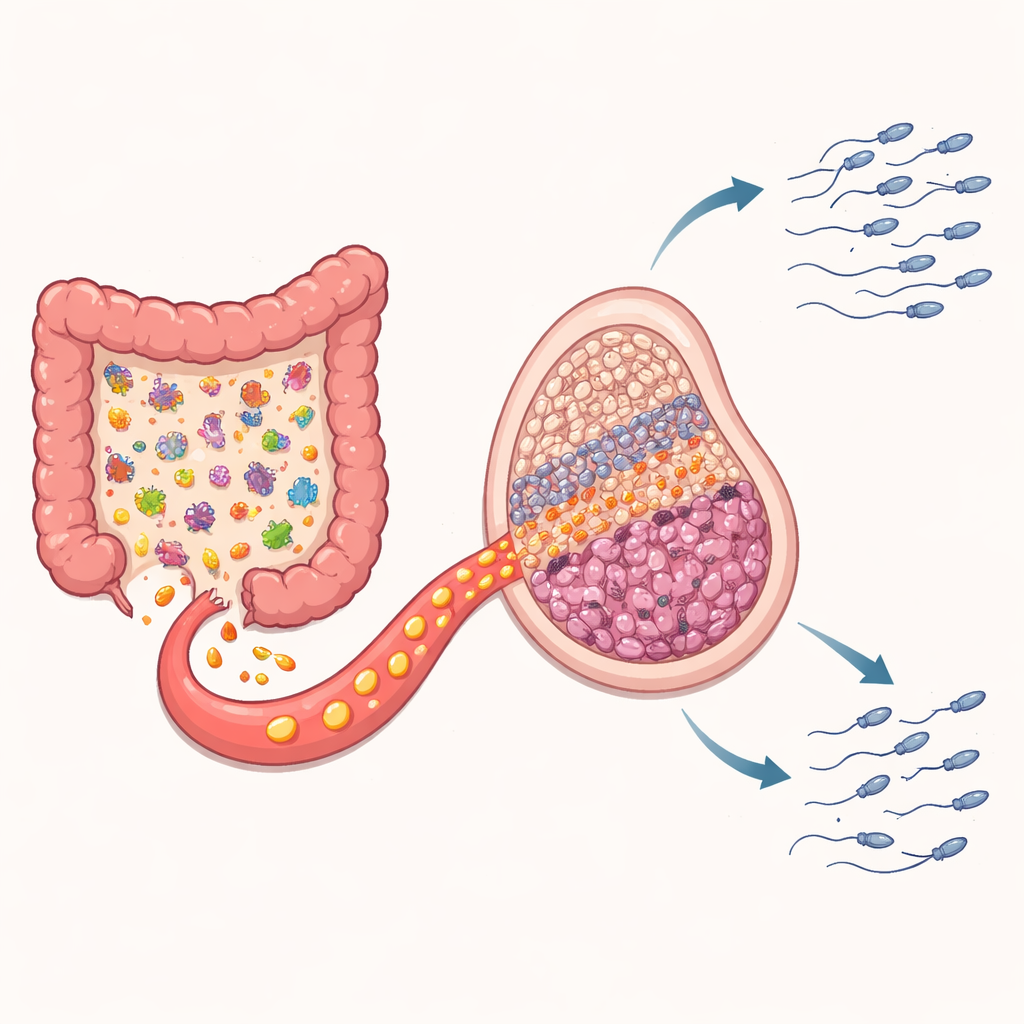
The Gut’s Hidden Role and a Key Chemical Messenger
Because gut microbes are known to shift with meal timing and to send signals throughout the body, the researchers next examined the animals’ intestinal communities. Mice fed only at night showed a distinct rearrangement of gut bacteria compared with both the free-feeding and daytime-restricted groups. Certain species linked in previous work to inflammation and poor sperm quality became more abundant. When the team profiled hundreds of small molecules produced in the gut, one stood out: kynurenic acid, or KYNA, a breakdown product of the amino acid tryptophan. Levels of KYNA were higher in the intestines and in the bloodstream only in the night-fed mice. Statistical analyses connected this rise in KYNA to the overgrowth of particular bacterial groups, supporting the idea that altered microbes were driving the change.
Testing Whether KYNA Itself Harms Sperm
To see if KYNA was just a bystander or an active troublemaker, the scientists gave healthy mice KYNA by mouth for four weeks, at doses designed to mimic the increased levels seen with nighttime feeding. The results were strikingly similar to those in the night-fed group: reduced testis size, fewer developing sperm, and lower sperm counts and motility, while testosterone levels stayed largely stable. In cell culture, KYNA proved directly toxic to developing sperm-like cells at higher doses, but was gentler on the hormone-producing cells. In testicular tissue, KYNA dialed down one receptor type and strongly turned on another pathway, known from previous work to trigger cell death in germ cells. Together, these findings argue that excess KYNA, generated in the gut and carried by the blood, plays a central role in stalling sperm production.
What This Means for Men and Meal Timing
For everyday readers, the message is not that all time-restricted eating is harmful. In fact, mice restricted to daytime eating in this study did not show the same reproductive problems, and some human trials suggest metabolic benefits from carefully timed fasting. Instead, the work highlights that “when” we eat can influence male fertility, at least in mice, by reshaping gut microbes and boosting a chemical messenger that quietly sabotages sperm formation. While more work is needed to confirm how closely this applies to men, especially given species differences and lifestyle complexity, the study adds a cautionary note: aligning eating patterns with natural daily rhythms and maintaining a healthy gut community may be important not only for weight and blood sugar, but also for protecting the ability to father children.
Citation: Yang, D., Xie, D., Li, C. et al. Nighttime-restricted feeding disrupts spermatogenesis in mice via gut microbiota-derived KYNA homeostasis. Commun Biol 9, 354 (2026). https://doi.org/10.1038/s42003-026-09619-6
Keywords: male fertility, time-restricted feeding, gut microbiome, kynurenic acid, circadian rhythm