Clear Sky Science · en
Reverse transcriptase inhibitors enable the generation of fertile spermatids from fetal mouse testes in vitro
Turning Early Germ Cells into Working Sperm
Infertility treatments and basic research both depend on understanding how immature germ cells in the testis turn into sperm. This study shows that it is now possible to coax very early fetal mouse testes, taken just after an embryo’s sex is determined, to complete this transformation entirely in a dish. By carefully tuning the chemical environment and oxygen level, the researchers not only produced mature sperm-like cells, but also used them to create healthy, fertile offspring.
Why Early Testis Development Is Hard to Copy
Growing sperm outside the body has been achieved before using testis tissue from newborn or older mice, but efficiency was low and attempts using much earlier fetal tissue largely failed. At this young stage, the testis is still assembling its internal architecture, and germ cells are undergoing sweeping changes in how their DNA is packaged and marked. Reproducing this intricate choreography in a dish is challenging, and previous cultures rarely progressed beyond early steps when starting from testes collected around embryonic day 12.5, shortly after the embryo’s sex is set.
Borrowing Antiviral Drugs to Protect Developing Cells
The team focused on a hidden obstacle: mobile DNA elements called retrotransposons, which can copy and insert themselves into new places in the genome. In normal testes in the body, these elements are tightly controlled, especially in germ cells. In culture, however, the authors found that retrotransposon activity was much higher, while the cell’s own defense genes were quieter. This imbalance correlated with poor sperm development. Because retrotransposons use a reverse transcriptase enzyme, similar to some viruses, the researchers tested several drugs originally designed as reverse transcriptase inhibitors. A specific three-drug mix, termed AEC, roughly doubled the fraction of spermatogenic cells and more than tripled the share of haploid, sperm-like cells in cultured neonatal testes compared with untreated controls.
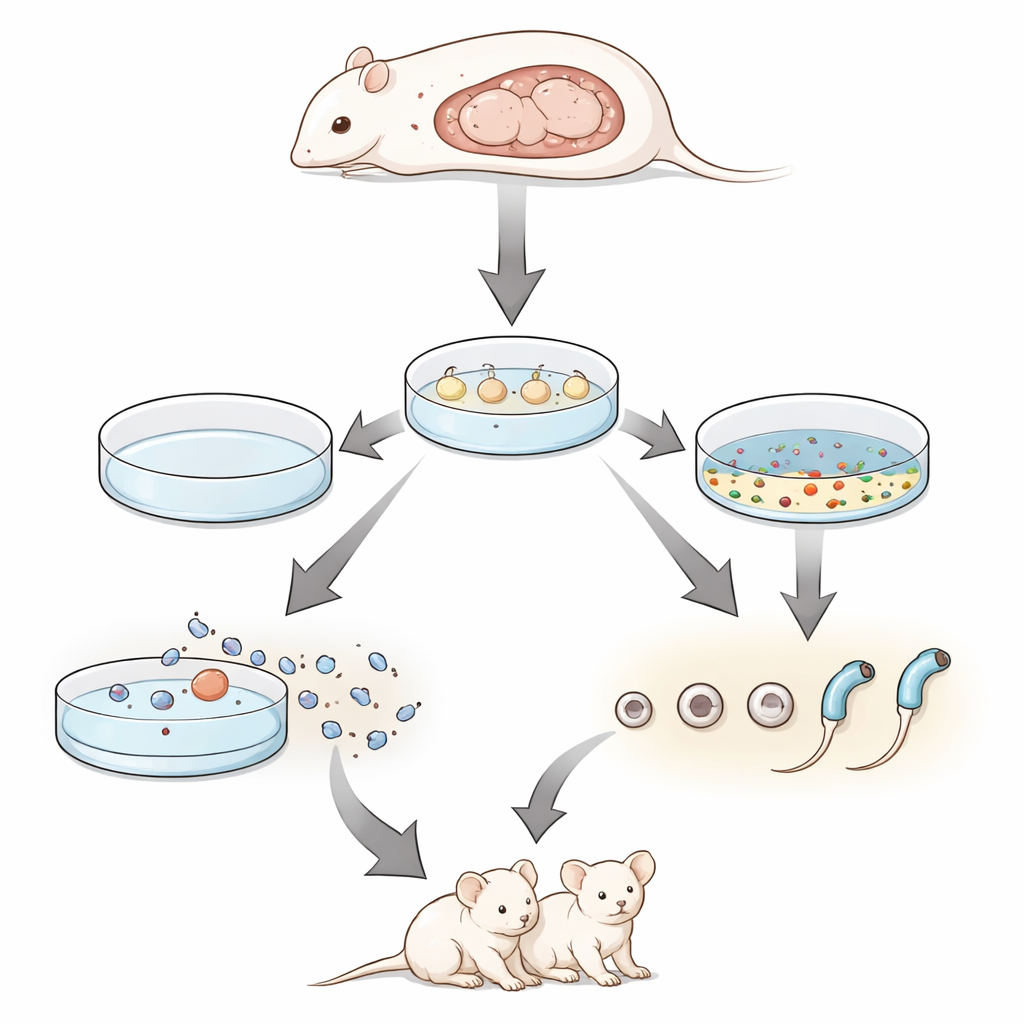
From Tiny Fetal Testes to Functional Sperm
Armed with this drug cocktail, the scientists turned to fetal testes. In tissue taken slightly before birth, the inhibitors boosted the appearance of advanced spermatids. More strikingly, when they cultured testes from embryos at day 12.5, a stage never before shown to complete spermatogenesis in vitro, they could now generate round and elongated spermatids. The biggest gains came when they combined the inhibitor mix with a low-oxygen atmosphere that better mimics conditions in the developing body. Under these hypoxic conditions, about a third of the tissue area showed markers of advanced germ cell development, and sperm with tails could be recovered from the cultures.
Testing Whether Lab‑Made Spermatids Really Work
To find out if these in vitro–derived cells were truly functional, the team used a technique called round spermatid injection. They isolated round spermatids from the cultured fetal testes and injected them into mouse eggs that had been gently activated. The resulting embryos were transferred into foster mothers. From just two cultured testis fragments, the procedure yielded multiple live pups, many of which carried a fluorescent marker gene that confirmed their cultured origin. When these first-generation animals were allowed to mate, they produced normal-sized litters of healthy second-generation offspring, demonstrating that the lab-derived spermatids could support not only development to birth but also full fertility.
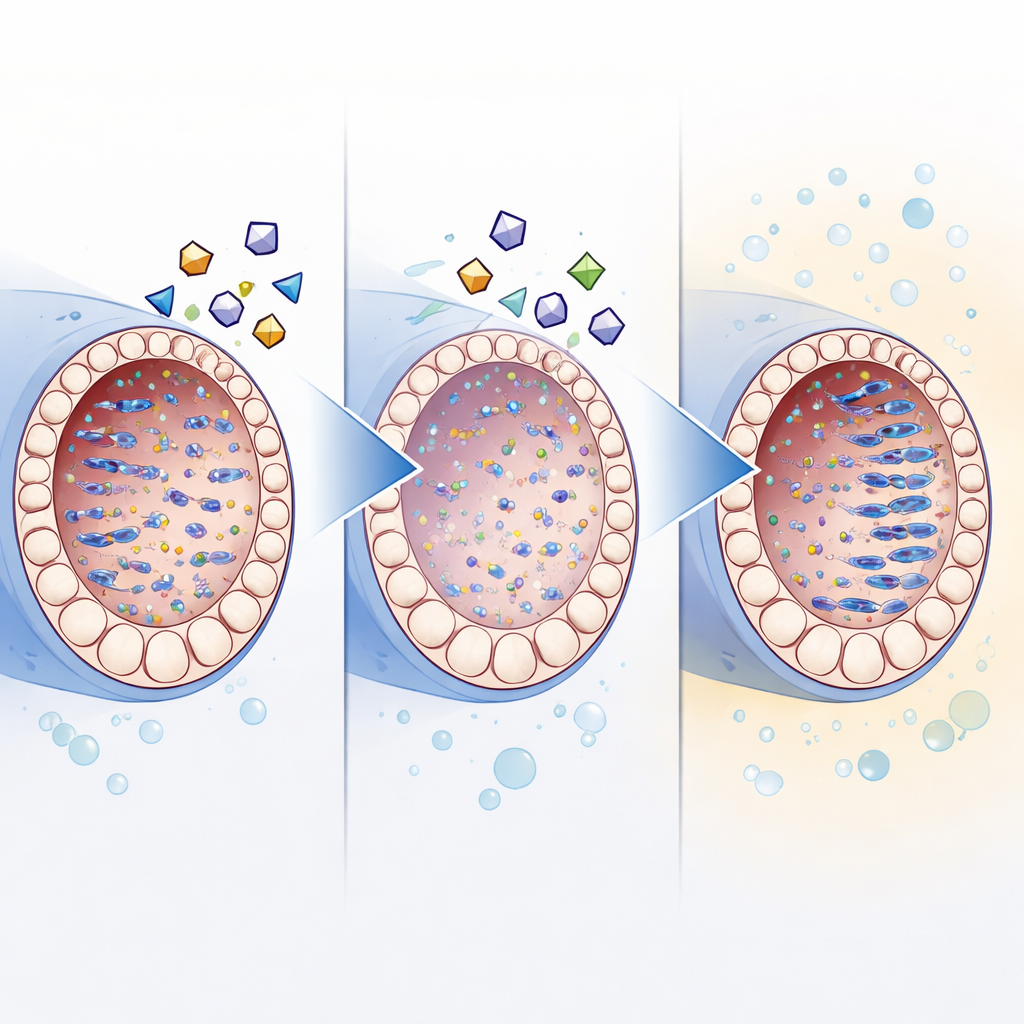
How Blocking Mobile DNA May Enable Future Therapies
Further experiments supported the idea that reverse transcriptase inhibitors help mainly by keeping mobile DNA in check. Compared with age-matched testes in the body, cultured testes showed higher levels of retrotransposon proteins and extra copies of their DNA, along with reduced expression of the cells’ natural silencing machinery. Adding the inhibitor cocktail reduced this accumulation, while simultaneously improving germ cell maturation. Although the drugs slightly slowed overall tissue growth, attempts to counter this with serum-based additives did not improve sperm output, suggesting that growth and proper differentiation must be balanced carefully.
What This Means for Reproductive Science
This work shows that even very immature fetal testes already contain all the ingredients needed to build a functioning sperm factory, provided the environment protects their genomes and mimics key aspects of life in the womb. By combining antiviral-style drugs with low oxygen, the researchers created a culture system that recapitulates complete spermatogenesis from one of the earliest stages yet tested and yields offspring that grow and reproduce normally. While the study is in mice and far from clinical use, it opens a powerful window into the earliest steps of male germ cell development and offers clues that may one day help preserve or restore fertility in patients whose reproductive cells are damaged early in life.
Citation: Nishida, M., Ono-Sunagare, Y., Kato, S. et al. Reverse transcriptase inhibitors enable the generation of fertile spermatids from fetal mouse testes in vitro. Commun Biol 9, 329 (2026). https://doi.org/10.1038/s42003-026-09613-y
Keywords: in vitro spermatogenesis, fetal testis culture, reverse transcriptase inhibitors, retrotransposons, male fertility