Clear Sky Science · en
Obesity impairs spermatogenesis via Leydig cell ferroptosis induced by liver-derived exosomal miR-122-5p
Why weight and fertility are linked
Obesity is usually discussed in terms of heart disease and diabetes, but it can also quietly undermine male fertility. This study in mice uncovers a surprising way that a fatty diet can lower testosterone and damage sperm production: the liver sends tiny "message bubbles" into the blood that trigger a special kind of cell death in the hormone‑producing cells of the testes. Understanding this hidden liver‑to‑testis communication may help explain why overweight men often struggle with low testosterone and infertility, and point toward new treatment targets.
Hidden mail between organs
Our organs constantly talk to each other using hormones and other chemical messengers. One form of this communication uses exosomes—nanometer‑sized bubbles released by cells that carry fats, proteins, and genetic material. The researchers started with mice fed either a normal diet or a high‑fat diet that caused obesity, insulin resistance, and poor blood sugar control. Obese mice had smaller, less active testes: their sperm counts, testosterone levels, and the size and number of sperm‑producing tubules were all reduced. When the team examined key cell markers in the testes, they found fewer testosterone‑producing Leydig cells and fewer early and late sperm cells, confirming that the whole sperm‑making assembly line was impaired. 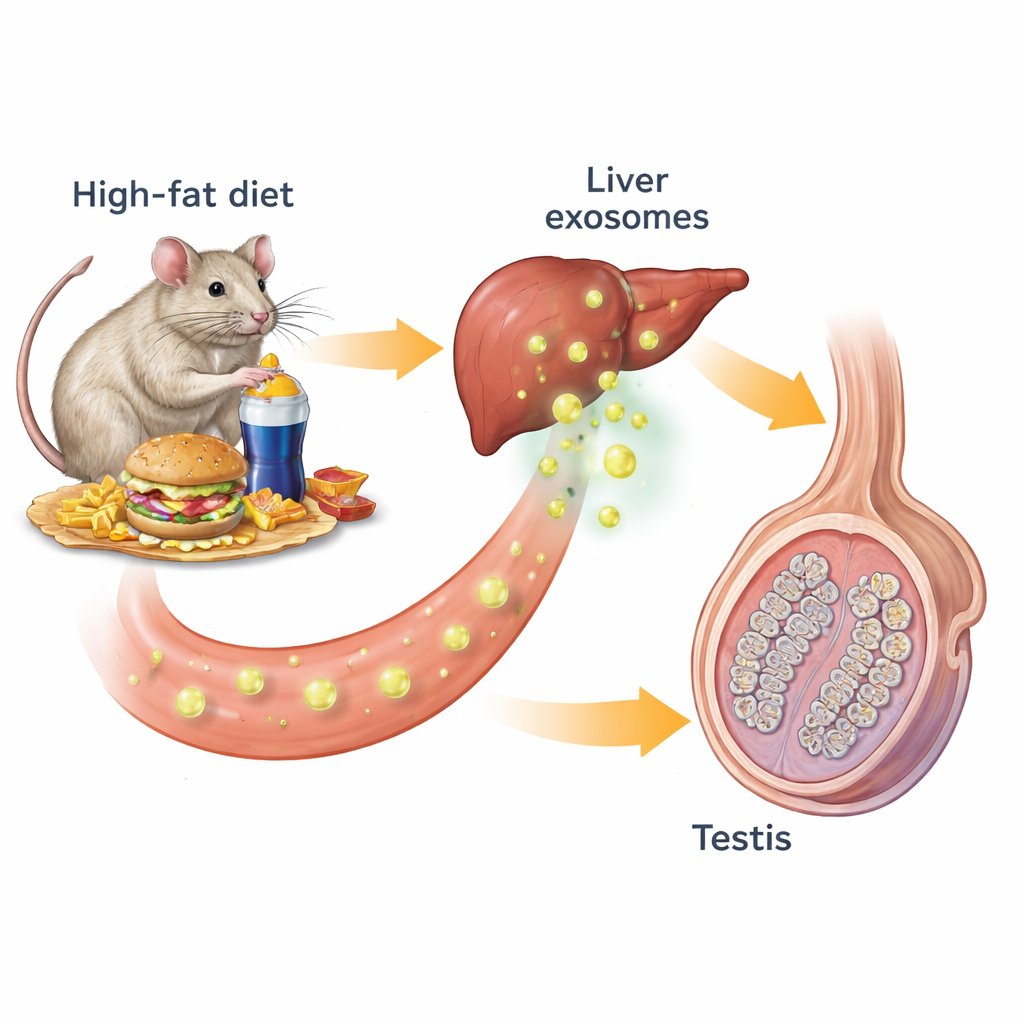
Exosomes spread the damage
To test whether circulating exosomes help drive this damage, the scientists purified exosomes from the blood of obese and lean mice. Injecting exosomes from obese mice into healthy, lean mice was enough to reproduce many of the problems: the recipients developed signs of metabolic trouble, their testosterone levels fell, and their sperm counts and testis structure worsened. Blocking exosome release in obese mice with a drug called GW4869 largely reversed these changes, restoring sperm counts, testosterone, and normal testis architecture. These results show that exosomes in the bloodstream are not just bystanders; they actively carry harmful signals from obesity to the reproductive system.
A toxic form of cell death
The team then looked for what, exactly, the exosomes were doing to Leydig cells. They focused on ferroptosis, an iron‑driven form of cell death marked by iron buildup, oxidative damage to fats, and injured mitochondria—the power plants of the cell. In obese mice and in healthy mice given obese‑derived exosomes, testes showed higher levels of iron and malonaldehyde, a by‑product of fat damage, as well as clear mitochondrial injury under the electron microscope. Treating obese mice with a ferroptosis‑blocking compound, Ferrostatin‑1, preserved Leydig cells, boosted testosterone and sperm counts, and improved mitochondrial health. In cell culture, the same drug protected isolated Leydig cells from death and oxidative stress induced by exosomes from obese animals. Together, these experiments reveal ferroptosis as a key link between obesity and failing testosterone production.
The liver’s message and the SCD2 switch
To track down the message inside exosomes, the researchers sequenced their small RNA cargo and found that a particular microRNA, miR‑122‑5p, was strongly increased in exosomes from obese mice. This microRNA was especially abundant in the liver and in liver‑derived exosomes, and those liver exosomes could be traced traveling to the testes. When miR‑122‑5p levels were experimentally lowered in exosomes, Leydig cells were less prone to ferroptosis and testosterone production improved, even though the animals remained metabolically unhealthy. Further tests showed that miR‑122‑5p directly shuts down a gene called Scd2, which helps convert certain fats into forms that protect cells from oxidative damage. Reducing Scd2 in Leydig cells made them more vulnerable to ferroptosis and lowered testosterone, while boosting Scd2 in obese mice partially restored hormone levels, sperm production, and mitochondrial structure. 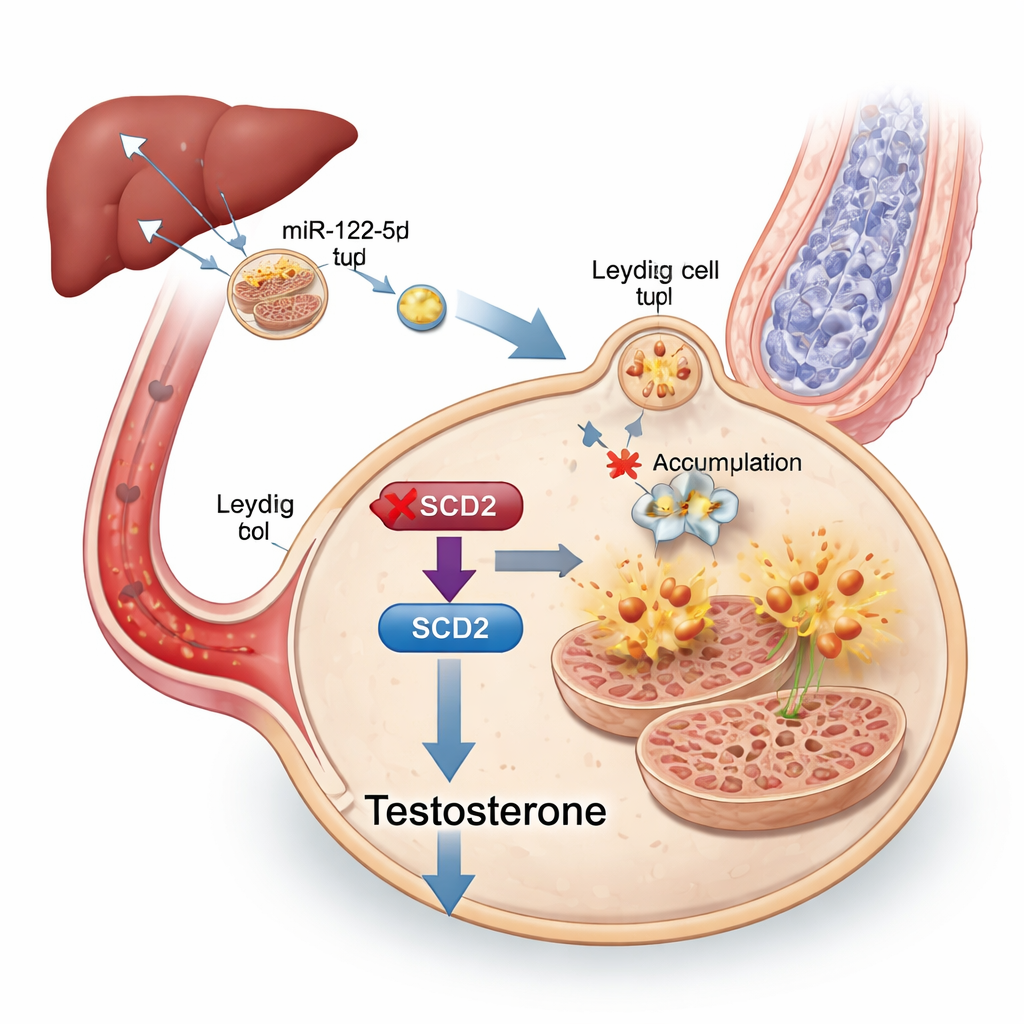
What this means for human health
In plain terms, this work shows that a high‑fat diet can cause the liver to load exosomes with an overabundance of miR‑122‑5p. These exosomes travel through the blood to the testes, where the microRNA flips off the Scd2 “fat‑handling” switch in Leydig cells. Without Scd2, these cells accumulate iron‑driven oxidative damage, undergo ferroptosis, make less testosterone, and ultimately support fewer healthy sperm. Although this research was done in mice and still needs to be confirmed in people, it identifies liver‑derived exosomes, miR‑122‑5p, ferroptosis, and Scd2 as promising biomarkers and potential drug targets for treating obesity‑related male infertility—and underscores yet another reason why long‑term high‑fat diets can quietly erode reproductive health.
Citation: Wang, N., Zhang, B., Chen, T. et al. Obesity impairs spermatogenesis via Leydig cell ferroptosis induced by liver-derived exosomal miR-122-5p. Commun Biol 9, 318 (2026). https://doi.org/10.1038/s42003-026-09612-z
Keywords: obesity and male fertility, testosterone, exosomes, Leydig cells, ferroptosis