Clear Sky Science · en
Two-in-one protein tag enables the soluble expression and calcium-dependent purification of single-chain antibody fragments
Making Powerful Antibodies Easier to Manufacture
Modern medicines increasingly rely on antibody fragments—small, precise “guided missiles” that can seek out cancer cells, inflamed tissues, or disease-causing molecules. Yet these promising drugs are surprisingly hard to manufacture: in bacterial factories, they tend to clump, misfold, and resist standard purification methods. This study describes a new protein add-on, called the CSQ-tag, that tackles both problems at once, making these fragments easier to produce and clean up in a simple, low-cost way.
Why Shrunken Antibodies Matter
Full-size antibodies have transformed the treatment of cancer, autoimmune disease, and eye disorders, but their large size and complex structure come with drawbacks. They can be slow to clear from the body, may not penetrate tumors or tissues efficiently, and are expensive to produce. Single-chain variable fragments, or scFvs, keep only the business end of an antibody—the part that recognizes a target—linked into a single chain. These compact molecules enter tissues more readily and can be engineered into sophisticated therapies and diagnostics. However, when made in common bacteria such as Escherichia coli, many scFvs pile up in insoluble clumps called inclusion bodies, and there is no universal, easy way to purify them.
A Two-in-One Helper Tag
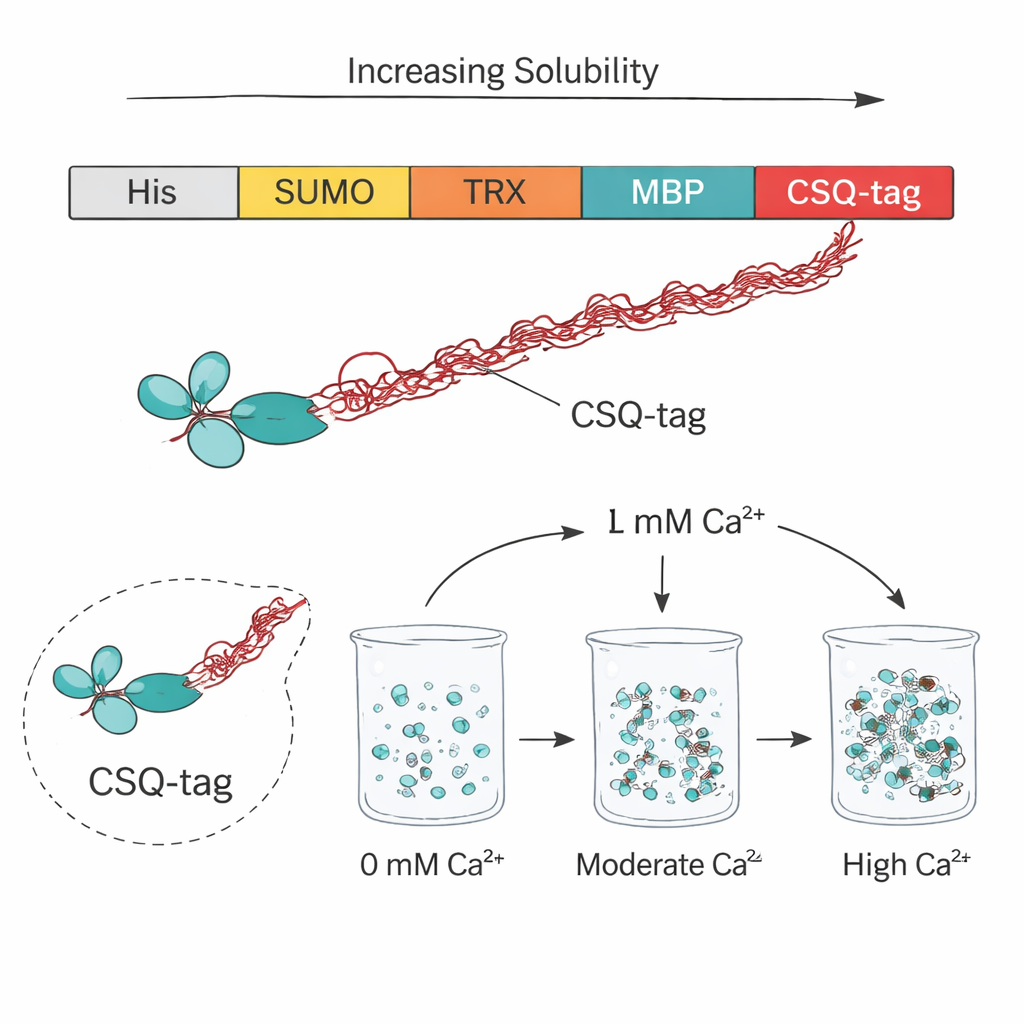
The researchers turned to calsequestrin, a natural calcium-storing protein in muscle cells, to design a helper tag they call CSQ-tag. This tag is unusually acidic and highly flexible, with long, disordered regions that behave like “entropic bristles” sweeping out space around the attached protein. When fused to four different therapeutic scFvs that normally aggregate, the CSQ-tag dramatically boosted how much stayed dissolved inside standard E. coli cells. While the same scFvs with only a simple His-tag were almost entirely insoluble, the CSQ-tagged versions reached an average solubility of about 84 percent—even in a bacterial strain that usually provides a chemically challenging environment for proper folding.
Beating Established Tag Technologies
To see how CSQ-tag stacked up against popular protein tags, the team compared it to SUMO, thioredoxin, GST, and MBP, which are widely used to coax troublesome proteins into solution. In this head-to-head test with an anti-VEGF scFv drug fragment (the basis of the eye drug brolucizumab), only MBP and CSQ noticeably improved solubility, and CSQ clearly came out on top. Across four different scFvs, CSQ increased soluble production about 1.8-fold more than MBP, long considered one of the best options. Further experiments showed that the most acidic and disordered portion of calsequestrin, known as domain 3, was crucial for this effect: removing it sharply reduced solubility, underscoring how the tag’s negative charge and structural looseness help keep partner proteins from sticking together.
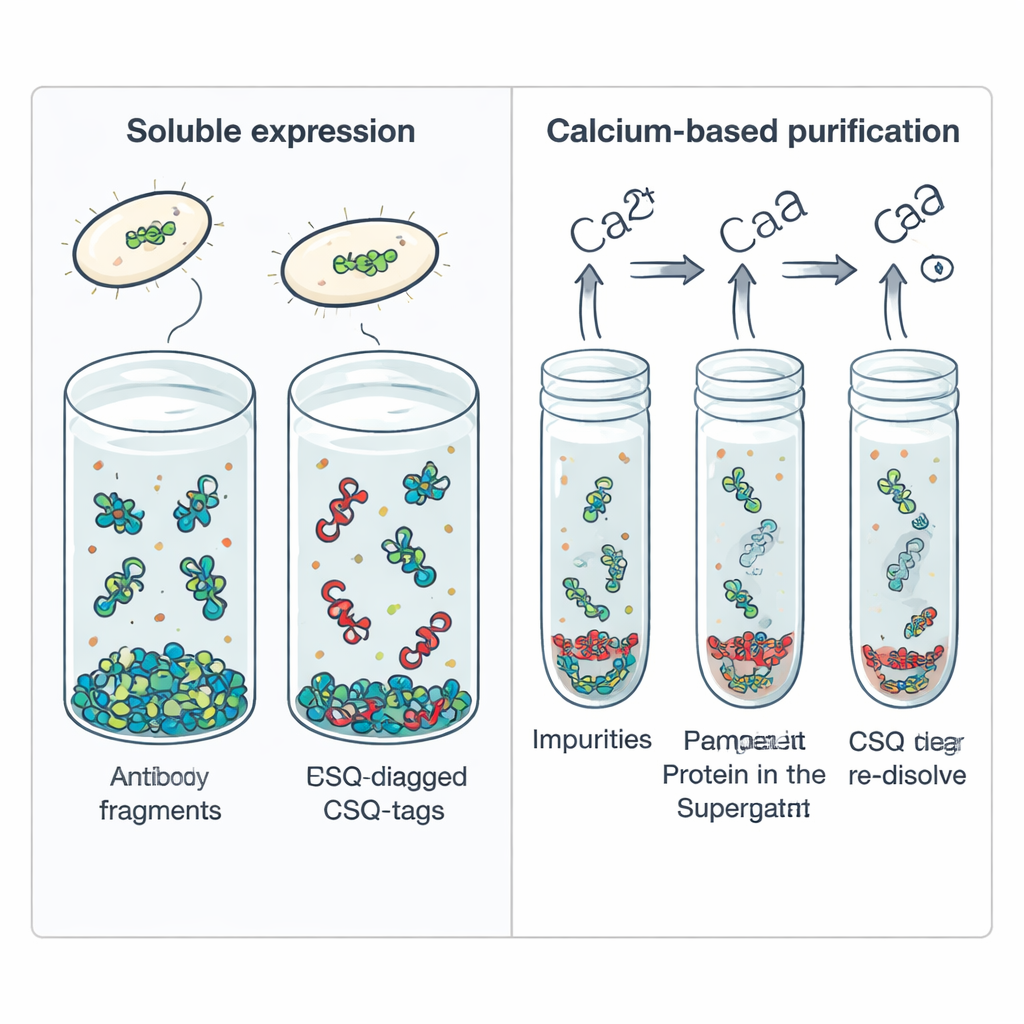
Using Calcium as a Simple Purification Switch
Beyond helping scFvs dissolve, CSQ-tag offers a built-in purification trick. Calsequestrin naturally clumps together when it binds calcium and falls apart again when calcium is removed. The authors harnessed this behavior by adding calcium to a cell extract containing CSQ-tagged scFv, which caused the tagged fusion proteins to aggregate while most bacterial contaminants stayed in solution. A quick spin in a centrifuge pulled down the CSQ-tagged protein; adding the calcium-chelating agent EDTA then redissolved it at high purity, over 95 percent, without expensive chromatography resins. This calcium-driven phase transition could be repeated without damaging the protein, making it attractive for large-scale manufacturing.
Keeping the Medicine Potent
For any therapeutic, enhancing production only matters if the final product still works. After using a standard enzyme to cut off the CSQ-tag and a final polishing step, the team tested the free anti-VEGF scFv. It bound its target with a strength essentially identical to the approved drug brolucizumab, and detailed chemical assays confirmed correct internal bonds and a lack of damaging aggregates. A second, particularly troublesome scFv aimed at the CD3 receptor showed similar preservation of activity. Overall yields from the CSQ-tag process were several times higher than those reported for traditional refolding methods, while using simpler equipment and cheaper reagents.
What This Could Mean for Future Treatments
By combining improved solubility and an easy purification handle into a single tag, the CSQ system addresses two long-standing bottlenecks in antibody fragment production. It allows drug developers to use fast-growing, inexpensive E. coli strains while still obtaining high-quality scFvs that retain their binding power. Although some optimization is still needed, especially to reduce losses after tag removal, this calcium-tunable tag could streamline manufacturing not only for antibody fragments but also for other sensitive therapeutic proteins. For patients, that could eventually translate into a broader range of targeted biologic drugs produced more efficiently and at lower cost.
Citation: Lee, J., Park, H., Jeong, S. et al. Two-in-one protein tag enables the soluble expression and calcium-dependent purification of single-chain antibody fragments. Commun Biol 9, 326 (2026). https://doi.org/10.1038/s42003-026-09611-0
Keywords: single-chain antibody fragments, protein expression, calsequestrin tag, calcium-dependent purification, therapeutic antibodies