Clear Sky Science · en
Glycolipid recognition and binding by Siglec-6 hinges on interactions with the cell membrane
How Our Cells Tell Friend from Foe
The immune system constantly scans our own cells, deciding when to stay calm and when to attack. A key part of this surveillance involves recognizing sugary structures on cell surfaces. This study reveals that one human immune receptor, called Siglec-6, uses not just those sugars but also the surrounding cell membrane itself to make especially precise decisions about what to bind. That unusual strategy could help explain how our bodies fine‑tune immune signals and may open new avenues for targeted therapies.
A Sugar-Sensing Gatekeeper on Immune Cells
Siglecs are a family of receptors that sit on immune cells and recognize sialic-acid–containing sugars, helping the immune system distinguish “self” from “non-self.” Most Siglecs rely on a single, highly conserved building block—an arginine amino acid—to latch onto these sugars. If that arginine is removed, binding usually fails. Siglec-6, however, is an oddball: earlier work showed that it can still bind some sugar-bearing lipids even when this key arginine is mutated. The new study set out to uncover how Siglec-6 gets away with breaking this apparent rule and what that means for its role on mast cells, memory B cells, and human placental cells.
The Special Lipids Siglec-6 Is Looking For
On cell surfaces, relevant sugars can be displayed on both proteins and lipids. This work focuses on a group of sugar-bearing lipids called gangliosides, especially three closely related ones named GM1, GM2, and GM3. All three poke a single sialic-acid–capped “head” out of the membrane. Earlier experiments showed that Siglec-6 binds strongly to GM1 when it is part of a membrane, but barely at all to GM2 or GM3, even though their sugar heads are very similar. Using detailed computer simulations of realistic membranes, the authors confirmed that the sialic acid in GM1 and GM3 is equally exposed and accessible. In other words, simple physical access is not what gives GM1 its special status. Instead, an additional sugar at the tip of GM1—the terminal galactose—emerged as a critical piece that helps position Siglec-6 correctly against the membrane.
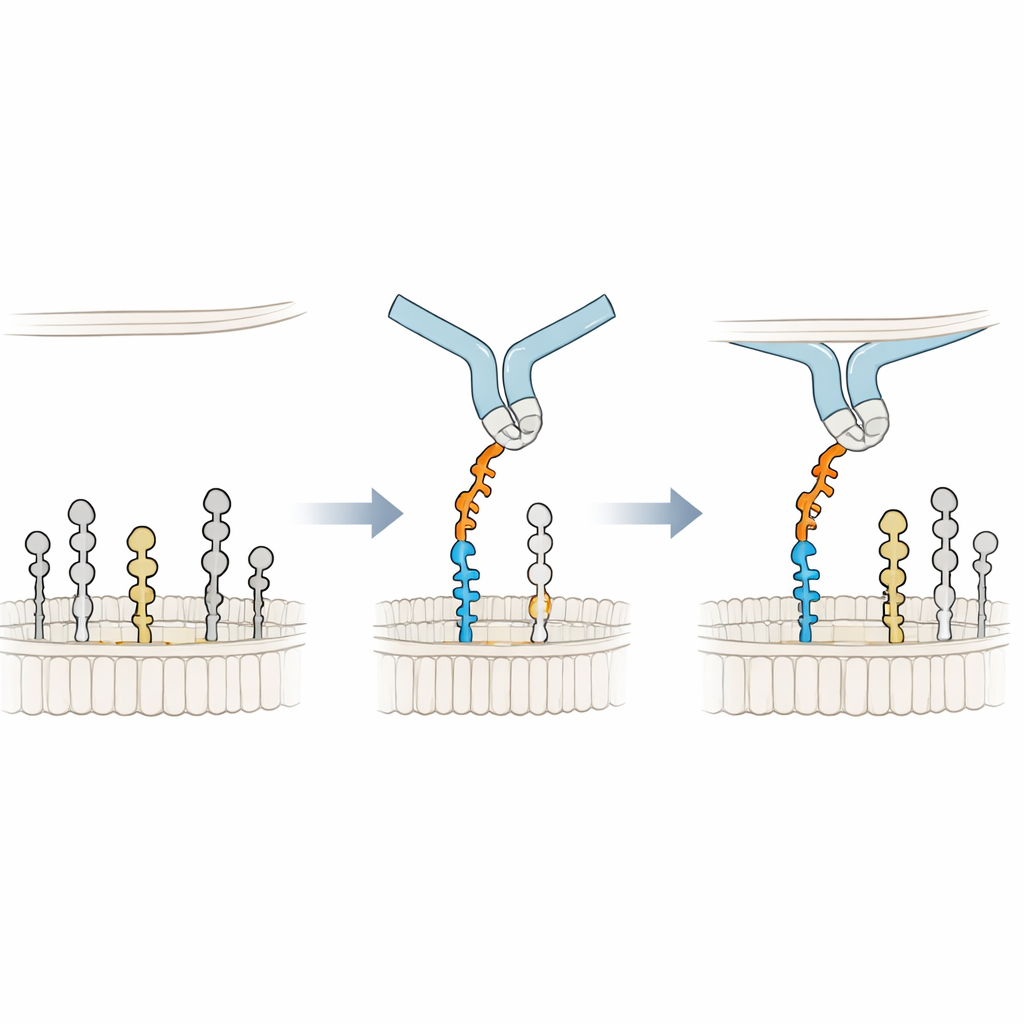
Leaning on the Membrane for a Better Grip
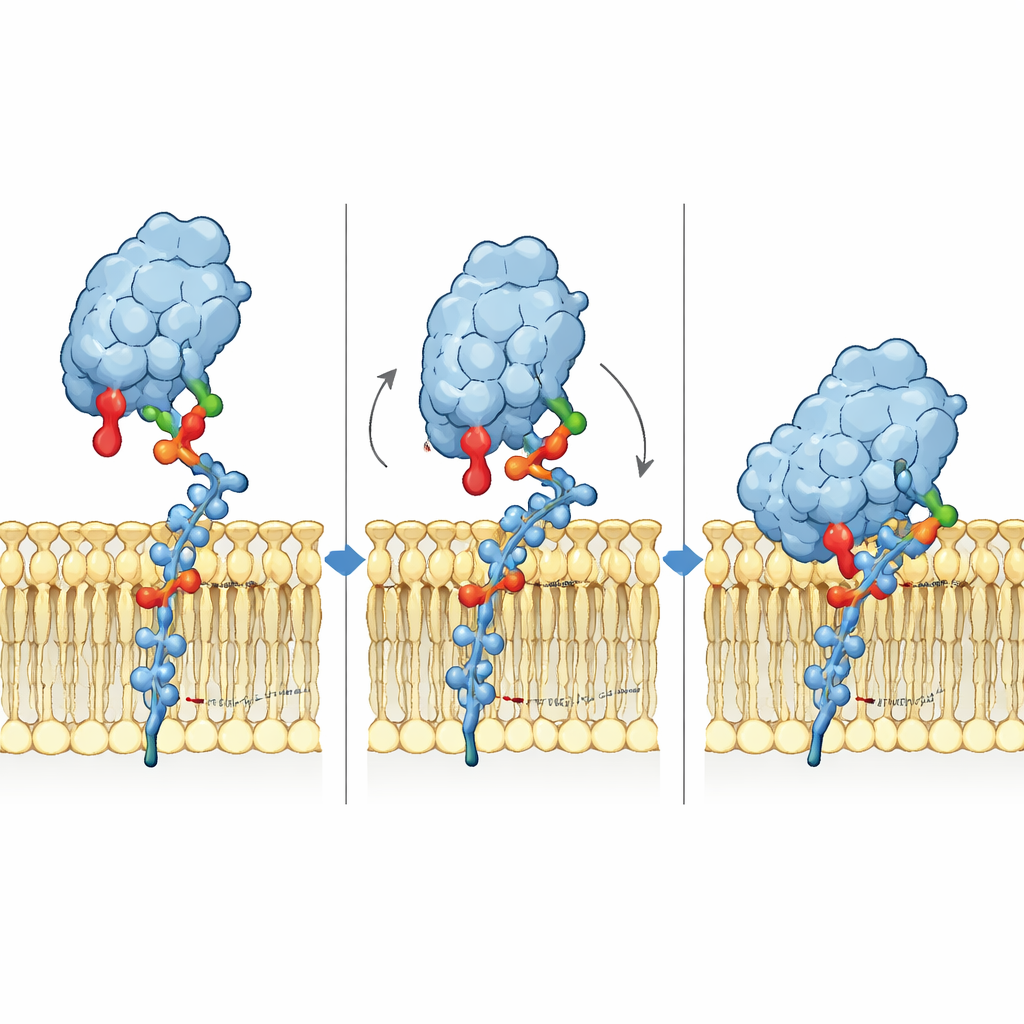
To understand the binding mechanism in atomic detail, the researchers built 3D models of Siglec-6 engaging GM1 in a membrane and ran long molecular dynamics simulations. They found that Siglec-6 does still use its canonical arginine (Arg122) to contact the sialic acid, but that contact flickers on and off over time. What keeps the complex stable is something new: a nearby tryptophan (Trp127) pushes into the fatty part of the membrane, while an adjacent lysine (Lys126) interacts with the charged head groups of surrounding lipids. This “wedge” into the membrane supplements the usual sugar–arginine interaction, effectively borrowing binding energy from the lipid environment. When the terminal galactose of GM1 is removed to mimic GM2, a flexible loop in Siglec-6 swings into the vacant space, pulls the receptor away from the membrane, and disrupts this membrane-assisted grip, explaining the loss of stable binding.
Experiments That Put the Mechanism to the Test
The team then tested these computer-based insights in living cell and biochemical assays. They engineered cells to express normal or mutant Siglec-6 and measured how well these cells bound to fluorescent liposomes and tiny lipid discs carrying GM1. Mutating the canonical arginine only modestly reduced binding when GM1 was in a membrane, confirming that Siglec-6 does not depend solely on this residue in that setting. In contrast, mutating Trp127 almost completely abolished binding to GM1-containing liposomes, and mutating both Trp127 and Lys126 nearly wiped out binding altogether. The same mutants, however, bound normally to GM1-like sugars presented outside of a membrane, showing that the basic structure of Siglec-6 was intact. Native mass spectrometry experiments further revealed that Siglec-6 can bind not only GM1 but also ordinary phospholipids, and that this lipid interaction disappears when Trp127 is removed. Remarkably, Siglec-6 even sticks to “naked” liposomes lacking GM1, again in a Trp127‑dependent manner, suggesting that it may first sample the membrane and then lock in when it encounters GM1.
Free Sugars Tell a Different Story
When the same three gangliosides were tested as free-floating sugar fragments rather than as part of a membrane, Siglec-6 behaved more like its relatives. In solution, it bound GM1, GM2, and GM3 with similarly weak affinity, and now the canonical arginine was essential: mutating Arg122 sharply reduced binding, while mutating Trp127 had little effect. This contrast shows that Siglec-6 effectively switches mechanisms depending on whether it encounters sugars in a membrane or in free form. In membranes, it relies on a cooperative partnership between the sugar head, the terminal galactose of GM1, and direct membrane contact; in solution, it reverts to the classic arginine-centered recognition motif.
Why This Matters for Immune Control
Taken together, the study reveals Siglec-6 as a finely tuned sensor that uses the physical context of the membrane to sharpen its specificity. By partially anchoring itself in the surrounding lipids, it can selectively recognize GM1 among very similar gangliosides, effectively turning a general “sialic acid reader” into a high-precision detector of a particular surface pattern. This membrane-assisted strategy appears unique among Siglecs studied so far and may help Siglec-6 scan cell surfaces for specific glycolipid signatures that regulate immune responses or mark particular tissues, such as the human placenta. Understanding this dual mode of recognition could aid the design of therapies and diagnostic tools that exploit Siglec-6’s unusual combination of sugar and membrane sensing.
Citation: D’Andrea, S., Schmidt, E.N., Bui, D. et al. Glycolipid recognition and binding by Siglec-6 hinges on interactions with the cell membrane. Commun Biol 9, 333 (2026). https://doi.org/10.1038/s42003-026-09609-8
Keywords: Siglec-6, gangliosides, cell membrane, glycolipid recognition, immune regulation