Clear Sky Science · en
ATM Inhibition Enhances Knock-in Efficiency by Suppressing AAV-Induced Activation of Apoptotic Pathways
Making Gene Editing Work Better
CRISPR gene editing promises treatments for genetic diseases, stronger crops, and powerful research tools. But one of its most valuable tricks—precisely inserting new DNA into a chosen spot in the genome, known as a “knock-in”—still works inefficiently. This study explores why some cells accept new DNA more readily than others and reveals a way to coax cells to perform precise edits more often, especially when using adeno-associated virus (AAV), a leading vehicle for gene therapy.
Watching DNA Repair in Real Time
To understand what helps or hinders precise gene insertion, the researchers built an intricate “dashboard” inside mouse embryonic stem cells. This triple-reporter system uses three fluorescent markers to track, in the same cells, whether CRISPR has cut the DNA, whether a precise knock-in has occurred, and whether the cell has instead glued the donor DNA into place using a rougher repair method. By reading out different color combinations with flow cytometry, they could separate clean, template-based edits from error-prone insertions and unedited cells. This side-by-side tracking allowed them to map how different DNA repair pathways contribute to each outcome with far more resolution than older PCR-based tests.
Two DNA Helpers, Two Very Different Responses
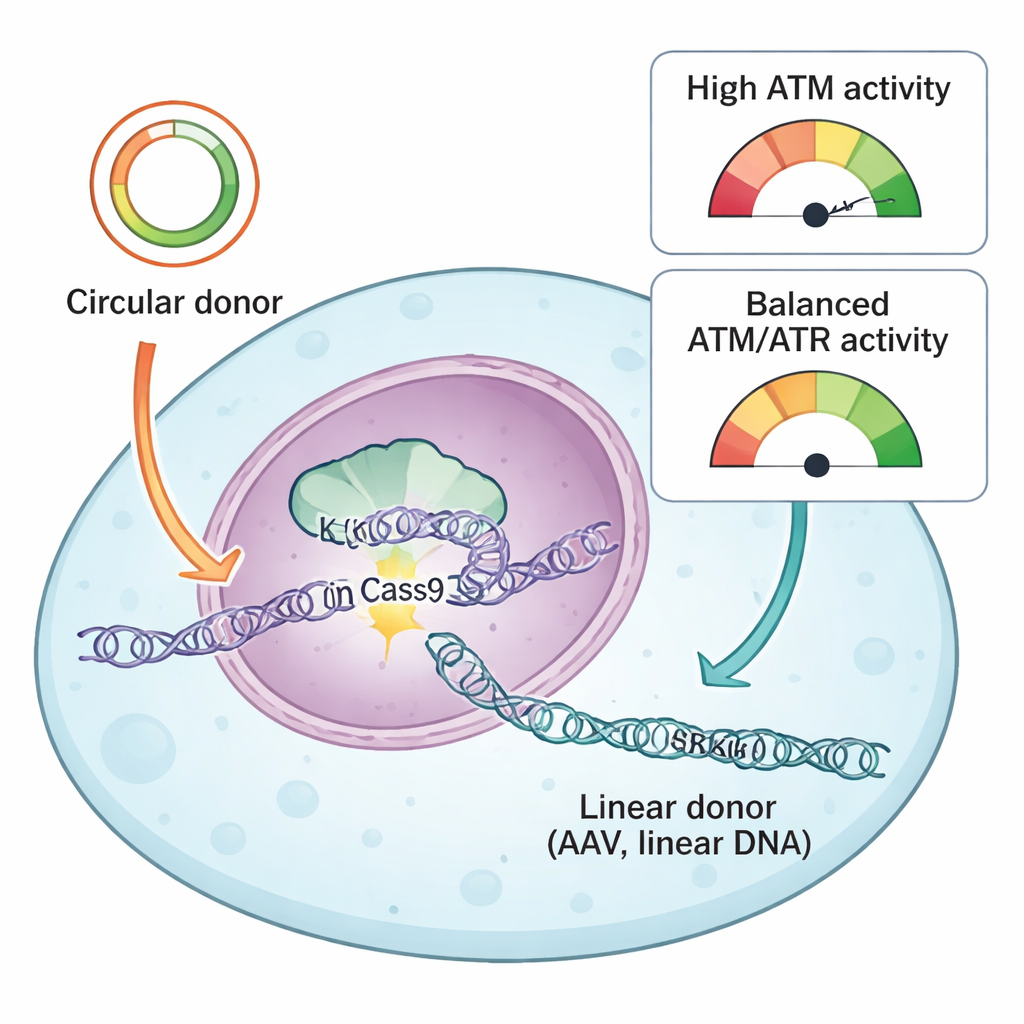
The team compared two common types of donor DNA used to guide CRISPR repairs: circular plasmids and linear AAV-derived DNA. Both carry matching “homology arms” that tell the cell where to insert the new sequence, but they enter the cell in different physical forms. Circular plasmids are closed loops of double-stranded DNA, whereas AAV donors typically arrive as single linear molecules. Using their reporter system and a library of drugs that target DNA damage–response proteins, the authors found that one key protein, ATR, was uniformly essential for successful knock-ins with both donor types. Inhibiting ATR sharply reduced precise insertion, while gently boosting ATR activity via a KEAP1–NRF2 pathway made knock-ins more efficient, especially with AAV donors.
When DNA Protection Backfires
A different DNA damage sensor, ATM, turned out to behave in a strikingly donor-dependent way. With circular plasmid donors, blocking ATM reduced both precise knock-in and end-joining–mediated insertions, matching its classic role in helping cells repair double-strand breaks. But with linear DNA—whether from AAV, self-complementary AAV, or plasmids cut into straight pieces—ATM inhibition did the opposite: it increased knock-in efficiency. The researchers traced this effect to how cells interpret bursts of linear DNA. High doses of AAV strongly activated ATM, which in turn switched on p53 and caspase 3, central players in programmed cell death. Cells that had taken up many copies of the donor DNA, and therefore had the highest chance of a successful knock-in, were also the most likely to be killed by this alarm system.
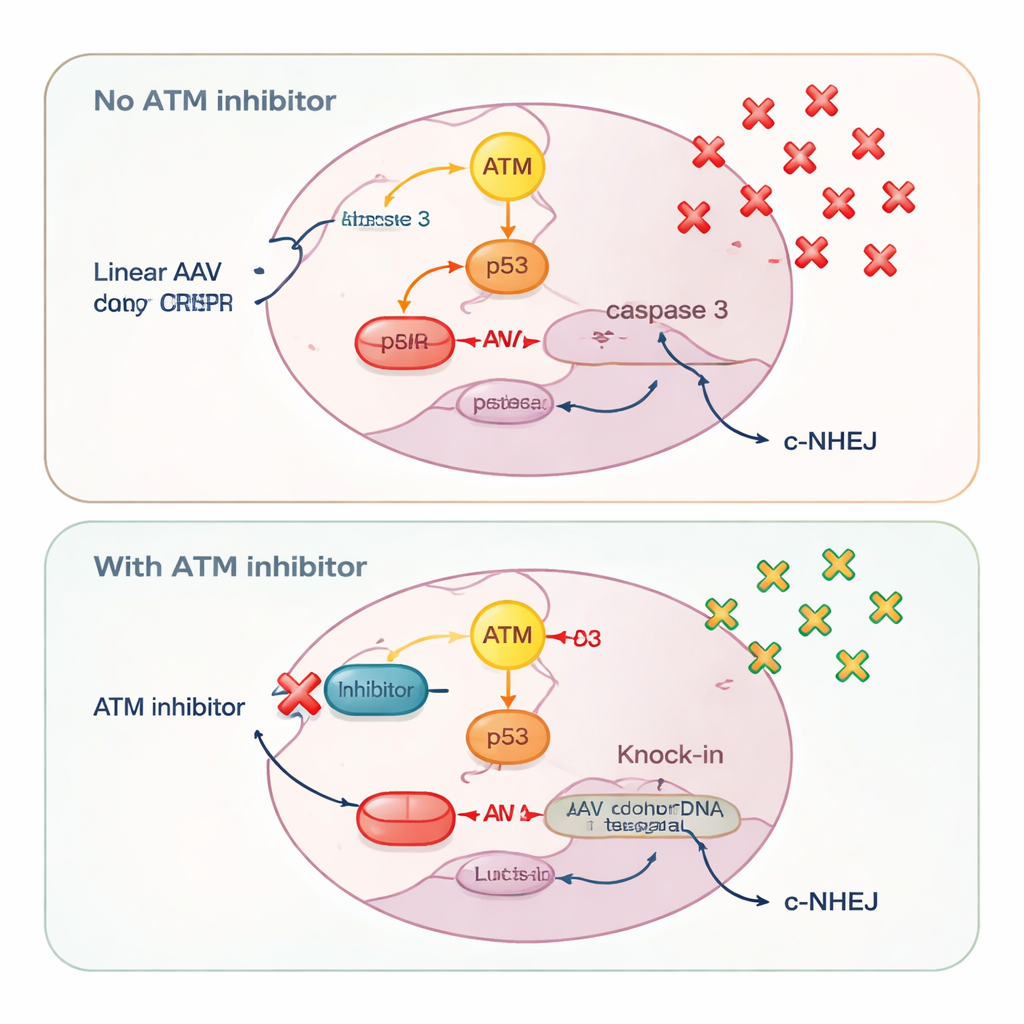
Saving the Best-Equipped Cells
By adding ATM inhibitors, the scientists quieted the ATM–p53–caspase 3 death pathway. This spared cells carrying high amounts of donor DNA, allowing more of them to survive long enough to complete a precise knock-in. Measurements of viral genome copies confirmed that ATM inhibition increased the number of AAV molecules retained inside cells. At the same time, ATM blockade slightly dampened a fast, blunt repair route called classical non-homologous end-joining, as shown by reduced activation of DNA-PK, a core enzyme in that pathway. Weakening this competing repair option nudged more CRISPR cuts toward homology-based knock-in rather than quick patch jobs that ignore the donor template.
What This Means for Future Gene Therapies
For non-specialists, the message is that the same cell safeguards that protect us from DNA damage can unintentionally sabotage precise gene editing, especially when large amounts of viral donor DNA are used. This work shows that gently tuning those safeguards—keeping ATR active but temporarily restraining ATM when linear donors like AAV are present—can make CRISPR knock-ins more reliable and efficient. Such a strategy could improve gene therapies, cell engineering, and disease models by helping more cells accept the intended change while avoiding unnecessary cell loss.
Citation: Natsagdorj, ME., Hara, H., Uosaki, H. et al. ATM Inhibition Enhances Knock-in Efficiency by Suppressing AAV-Induced Activation of Apoptotic Pathways. Commun Biol 9, 177 (2026). https://doi.org/10.1038/s42003-026-09604-z
Keywords: CRISPR knock-in, AAV gene editing, DNA damage response, ATM inhibition, genome engineering