Clear Sky Science · en
Bone mineral loss damages renal tubules in mice
When Weak Bones Put Stress on the Kidneys
Most people think of bones and kidneys as separate players in the body, but this study shows they are tightly linked. When bones lose mineral—for example during bed rest, osteoporosis, or even spaceflight—the extra minerals released into the blood can silently irritate and damage the kidney’s tiny tubes. Understanding this hidden connection may help protect kidney health in people who are losing bone mass.
Bone Loss Floods the Bloodstream
Bones store most of the body’s phosphate, a mineral that teams up with calcium to keep skeletons strong. The authors worked with mice and triggered rapid bone breakdown using a signal called RANKL, which turns on bone-resorbing cells. As bone mineral dissolved, both phosphate and calcium levels in the blood rose. The body responded by forming tiny mineral–protein complexes and by boosting a hormone called FGF23, which tells the kidneys to dump more phosphate into the urine.
Kidney Tubes Face a Mineral Overload
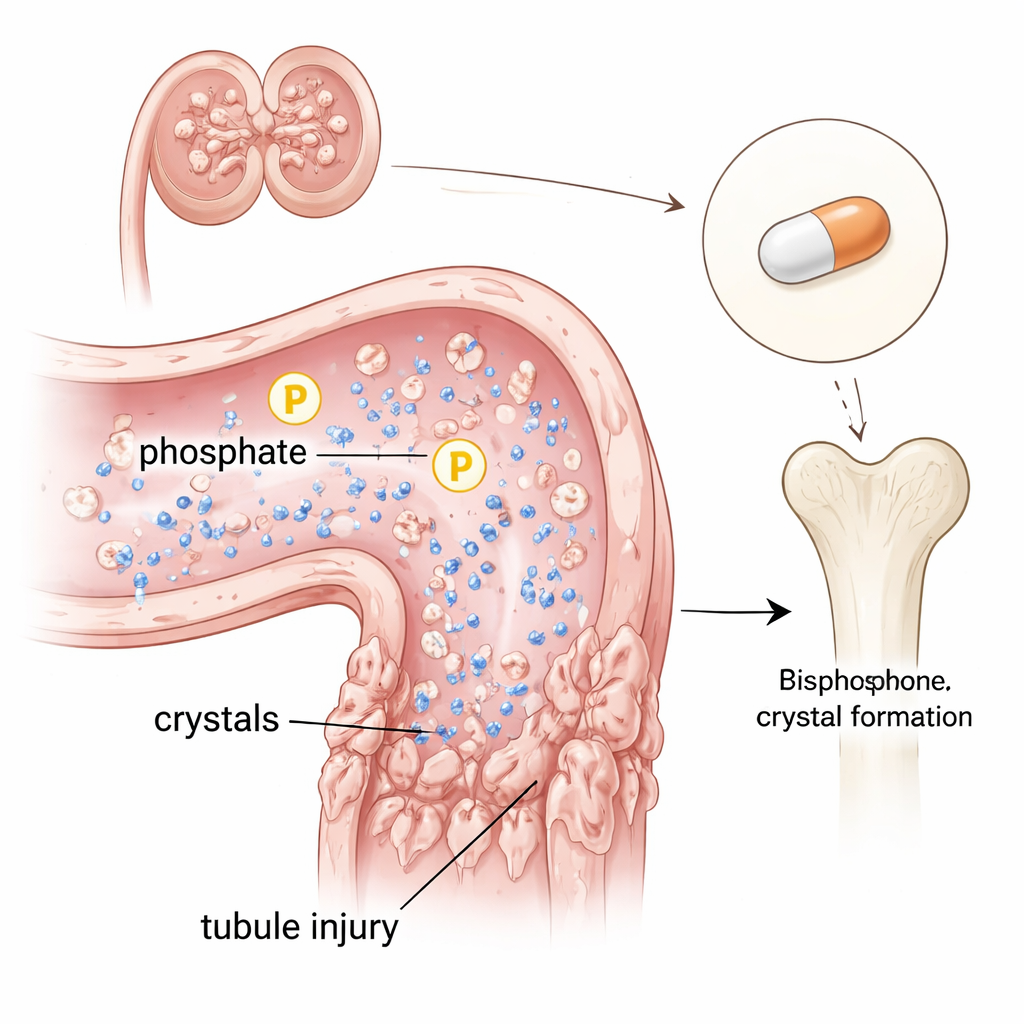
That emergency response helps keep blood phosphate in check, but it has a downside. When FGF23 pushes the kidneys to excrete more phosphate, the fluid flowing through the kidney’s filtering tubes becomes overloaded with mineral. In this harsh environment, calcium and phosphate can crystallize into microscopic particles. The study shows that when bone breakdown was driven hard and repeatedly, these particles appeared in the kidney tubules of mice and set off local injury signals, inflammation, and scarring—changes similar to those seen when animals are fed very high-phosphate diets.
Diet, Bone Loss, and a Dangerous “Second Hit”
The researchers then asked what happens when stressed kidneys face both a high-phosphate diet and bone loss at the same time. In mice eating a phosphate-rich diet, the kidneys already showed subtle signs of damage and low-grade inflammation. Adding bursts of bone-derived phosphate on top of this acted as a “second hit”: kidney injury markers stayed high, inflammatory cells accumulated, and fibrous tissue began to form. This suggests that in people who already have a heavy phosphate load—such as many patients with chronic kidney disease—episodes of rapid bone loss could accelerate long-term kidney harm.
Insights from Spaceflight and a Possible Protective Drug
To see whether a natural form of bone loss has similar effects, the team studied mice that spent about 10 days on the International Space Station. Even over this short stay, spaceflight mice showed increased bone resorption, higher blood levels of calcium, phosphate, and mineral particles, and early signs of kidney tube stress compared with matched mice kept on Earth. Finally, the scientists tested a common bone drug, a bisphosphonate, which kills overactive bone-resorbing cells. In mice, pretreating with this drug prevented bone mineral loss, blunted the surge in phosphate and FGF23, and sharply reduced kidney injury signals, hinting that protecting bone can also shield the kidneys.
What This Means for Everyday Health
To a lay reader, the take-home message is that bone health and kidney health are two sides of the same coin. When bones lose mineral quickly—during prolonged bed rest, after major fractures, in osteoporosis, or in the low-gravity conditions of space—the minerals do not just disappear. They move into the bloodstream, forcing the kidneys to work harder and exposing delicate kidney tubes to harmful crystal deposits. Over time, and especially in people whose kidneys are already strained, this extra burden may hasten kidney damage. Strategies that slow bone loss, whether through movement, diet, or carefully chosen medications, may therefore help protect not only our skeletons but also our kidneys.
Citation: Hayashi, H., Miura, Y., Iwazu, Y. et al. Bone mineral loss damages renal tubules in mice. Commun Biol 9, 304 (2026). https://doi.org/10.1038/s42003-026-09603-0
Keywords: bone loss, kidney injury, phosphate, osteoporosis, spaceflight