Clear Sky Science · en
STING controls glycolysis and histone lactylation to drive macrophage metabolic reprogramming in postoperative ileus
When Surgery Makes the Gut Hit Pause
After abdominal surgery, many patients find that their intestines simply refuse to wake up. This slowdown, called postoperative ileus, can mean days of nausea, bloating, and delayed recovery. The study summarized here asks a deceptively simple question: why do certain immune cells in the gut keep the bowel stalled, and could dialing down a single molecular switch help the intestine start moving again?
How a Routine Operation Sparks Gut Inflammation
Abdominal surgery inevitably disturbs the intestines. In mice, gentle handling of the bowel was enough to cause swelling, fluid buildup, and blocked contents in the small intestine. When researchers tracked how far a harmless fluorescent dye traveled through the gut, they saw that surgery sharply slowed its progress, a sign of impaired motility. Microscopic examination of the intestinal wall showed that the muscular layer—the tissue that actually contracts to push food along—became inflamed and crowded with incoming immune cells, especially macrophages and neutrophils. These findings suggest that the gut’s own muscular coat becomes an inflamed battlefield after surgery, and this local inflammation is closely tied to the bowel’s paralysis.
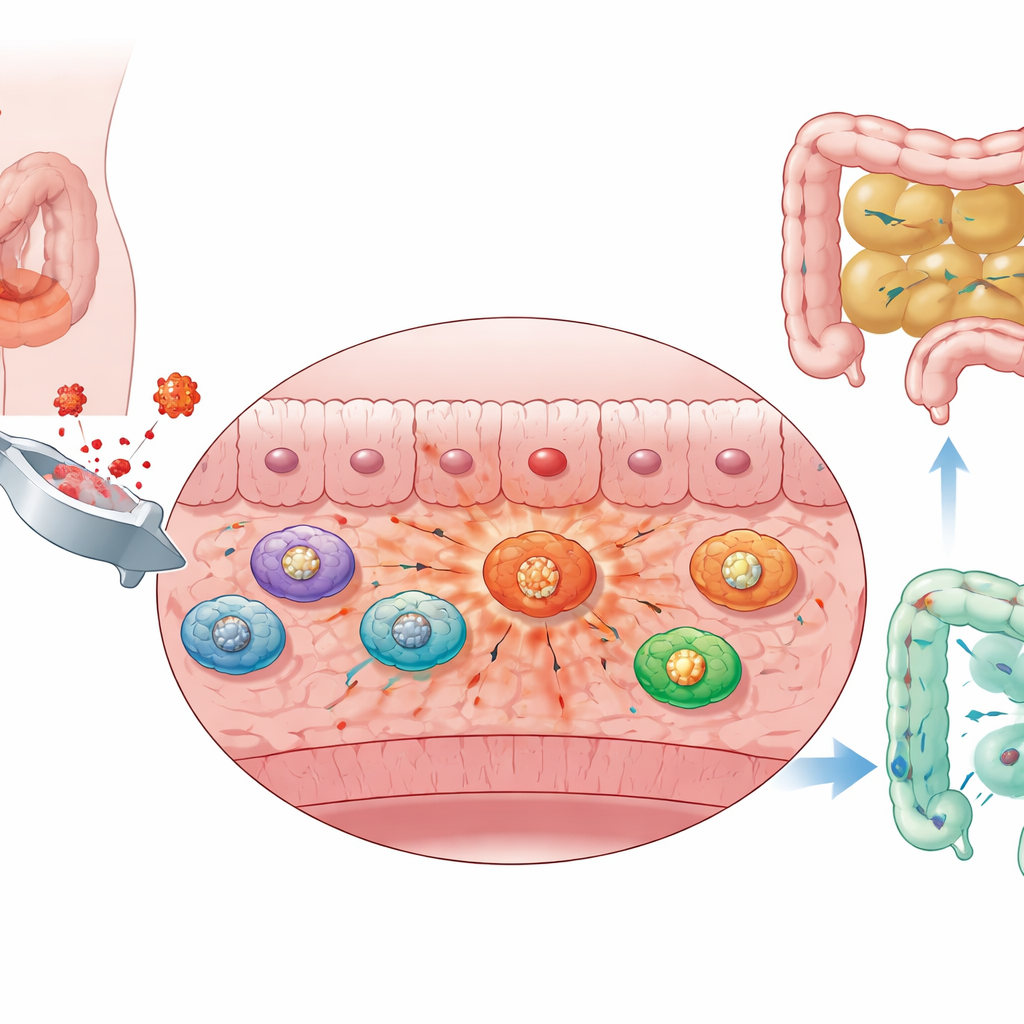
Macrophages Switch Their Fuel and Fan the Flames
To understand what these infiltrating macrophages were doing, the team mined large-scale RNA and single-cell sequencing data from the intestinal muscle layer. They found that macrophages in operated animals strongly boosted a fast, sugar-burning pathway known as glycolysis. Compared with other immune cells in the same tissue, infiltrating macrophages showed the highest glycolysis scores and expressed elevated levels of key glycolytic enzymes. A distinct subset of these cells relied heavily on glycolysis, carried strong inflammatory signatures, and responded vigorously to bacterial components such as lipopolysaccharide. In essence, the macrophages had flipped into a high-sugar, high-inflammation gear that appears to worsen the muscle-layer inflammation keeping the bowel from moving.
A Molecular Switch That Couples Sensing Danger to Burning Sugar
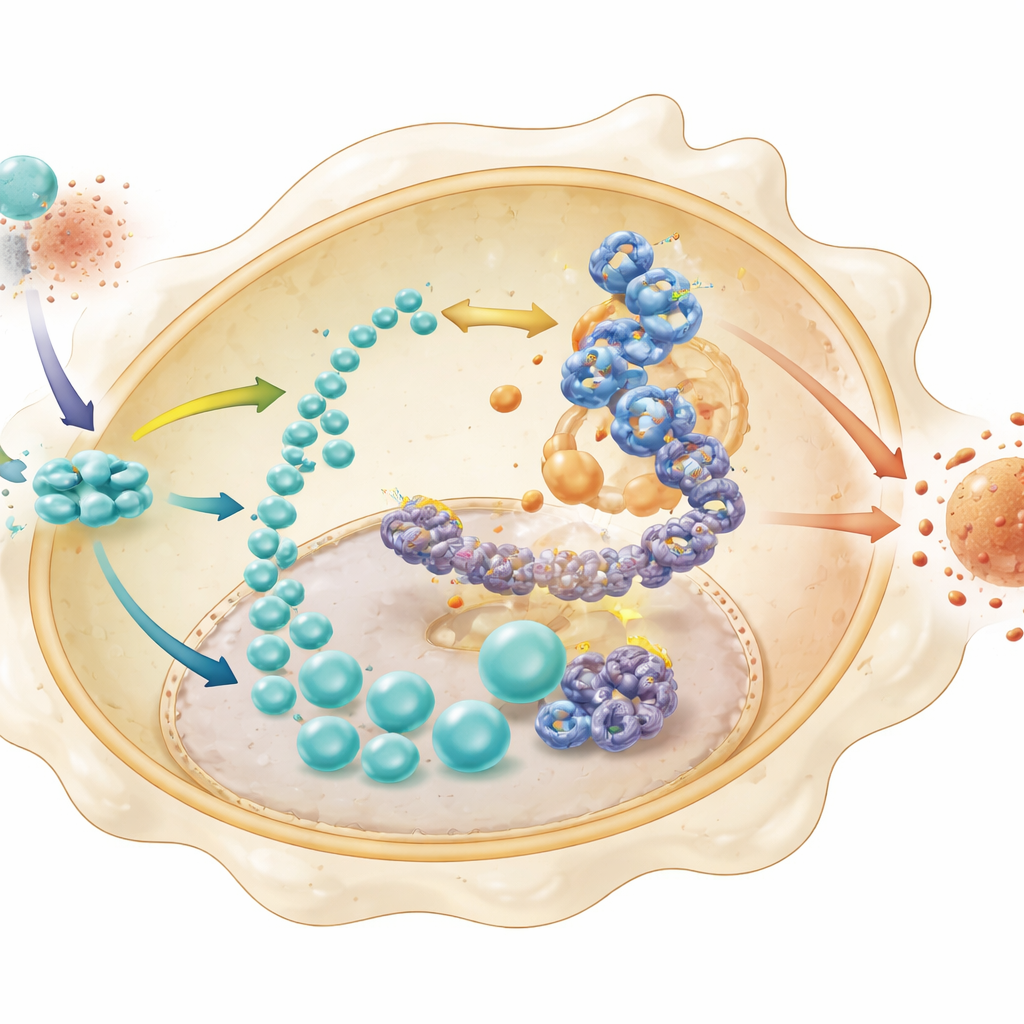
The researchers then focused on STING, a protein best known as an alarm system for foreign DNA but increasingly recognized as a metabolic regulator. In the inflamed gut muscle, the highly glycolytic macrophage subset showed markedly higher STING levels, a pattern confirmed in tissue staining. In cell culture, exposing macrophages to bacterial lipopolysaccharide drove up both STING activity and the glycolytic enzymes that help convert glucose into energy and lactate. When the scientists knocked out the STING gene, this sugar-burning burst collapsed: glycolytic intermediates dropped, acidification of the culture medium decreased, and fewer reactive oxygen species were produced. These STING-deficient cells also generated less lactate, implying that the alarm pathway and the metabolic engine are tightly coupled.
From Sugar Waste to Epigenetic Memory
Lactate is often viewed as a metabolic waste product, but here it plays a more subtle role. The team showed that in normal macrophages, the flood of lactate is used to chemically modify histones—the proteins that package DNA—through a process called lactylation. This modification was especially prominent at a site called H4K8 on histones near the gene for HK2, a gatekeeping glycolytic enzyme. In STING-deficient cells, histone lactylation and chromatin accessibility at the HK2 promoter were both reduced, and specialized sequencing methods confirmed that these changes curtailed HK2’s activation. At the same time, a downstream transcription factor, IRF3, was found to bind directly to the HK2 promoter more readily when this histone lactylation mark was present. Together, these steps create a self-reinforcing loop: STING stabilizes factors that boost glycolysis, glycolysis produces lactate, lactate decorates histones to open the HK2 gene, and IRF3 ramps up HK2 expression, further fueling glycolysis and inflammation.
Breaking the Loop to Let the Bowel Recover
Finally, the team asked whether breaking this loop could actually help animals recover from postoperative ileus. In mice lacking STING, intestinal muscle tissue expressed fewer glycolytic enzymes, contained fewer inflammatory macrophages, and showed a shift away from an aggressive, pro-inflammatory macrophage state toward a more resolving profile. Neutrophil infiltration was lower, and gut transit improved. Pharmacologically blocking STING in normal mice had similar benefits, while re-activating the HK2 enzyme in STING-deficient animals partially restored inflammation and motility problems. To a lay reader, the message is clear: a single danger-sensing switch in gut macrophages can rewire their metabolism in a way that locks the bowel into a prolonged pause after surgery. Targeting STING—or the sugar-burning, lactate-writing feedback loop it controls—offers a promising path toward speeding up recovery and easing one of the most stubborn complications of abdominal operations.
Citation: Chen, K., Li, G., Cheng, Y. et al. STING controls glycolysis and histone lactylation to drive macrophage metabolic reprogramming in postoperative ileus. Commun Biol 9, 358 (2026). https://doi.org/10.1038/s42003-026-09602-1
Keywords: postoperative ileus, macrophage metabolism, STING pathway, glycolysis, gut inflammation