Clear Sky Science · en
TGFBI promotes liver fibrosis through remodeling the profibrotic microenvironment by a positive feedback regulatory loop
Why this matters for liver health
Liver scarring, or fibrosis, underlies many common liver diseases and can silently progress to cirrhosis and cancer. Yet doctors still have few drugs that actually halt or reverse this scarring. This study uncovers how one little-known protein, called TGFBI, helps drive liver fibrosis by coordinating a damaging conversation between immune cells and scar‑forming cells. Understanding this cross‑talk could open the door to new treatments that interrupt the scarring process at its source. 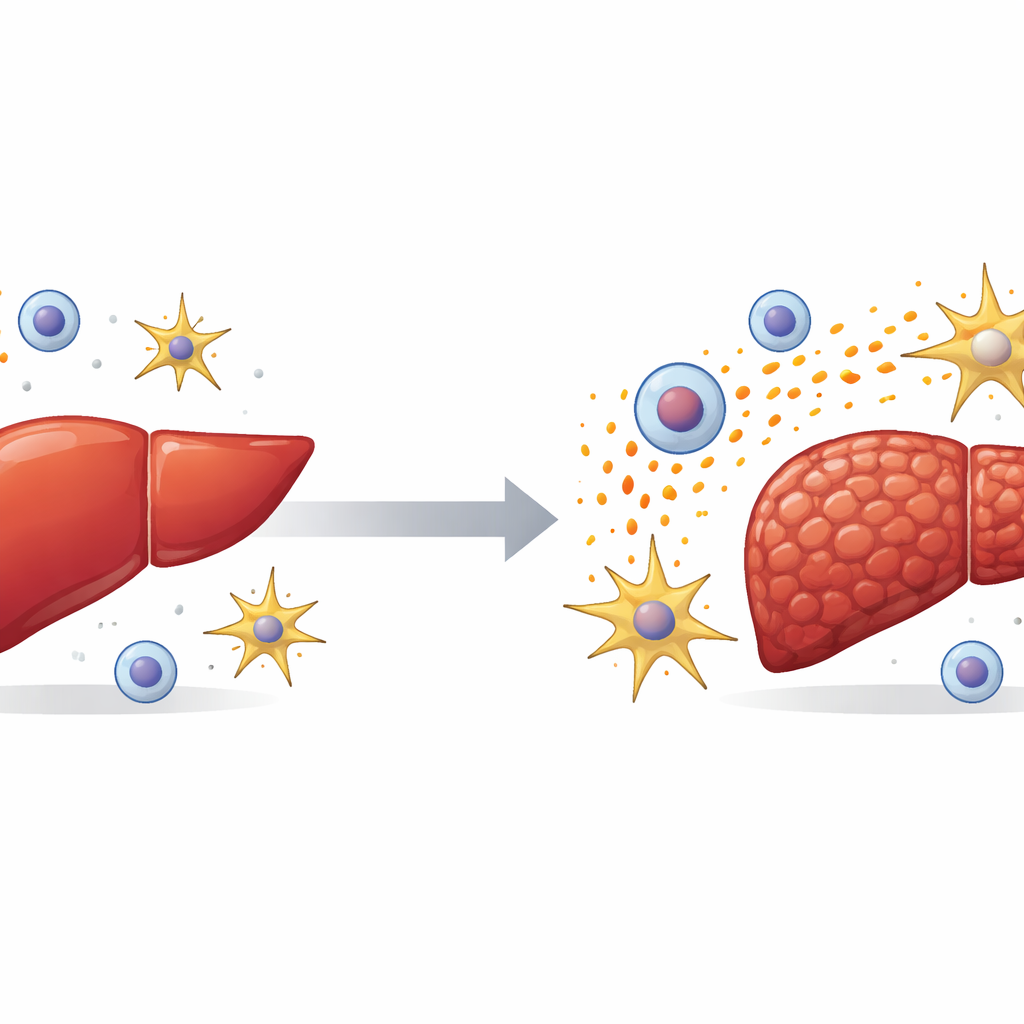
A hidden driver in a scarred liver
The researchers began by examining human liver samples and large public datasets. They found that TGFBI levels were much higher in people with severe liver scarring than in those with healthy livers. Mice with experimental liver injury showed the same pattern: TGFBI surged in damaged livers created by either a toxic chemical or bile duct blockage. By isolating different liver cell types, the team discovered that TGFBI is produced mainly by non‑parenchymal cells—especially immune cells called macrophages and star‑shaped support cells known as hepatic stellate cells. These are exactly the cells known to orchestrate inflammation and scar formation.
What happens when TGFBI is missing or added
To test whether TGFBI is merely a bystander or an active culprit, the scientists engineered mice that lacked the Tgfbi gene. When these mice were exposed to liver‑damaging treatments, they developed far less scarring than normal animals. Their livers accumulated less collagen, had fewer dying cells, and showed lower levels of inflammatory molecules and infiltrating macrophages. The opposite experiment was just as revealing: when healthy mice were given extra TGFBI protein, their livers began to show signs of inflammation and fibrosis even without another obvious trigger. Together, these experiments show that high TGFBI is not just associated with liver disease—it is sufficient to worsen it.
How TGFBI turns stellate cells into scar factories
Hepatic stellate cells are the liver’s main source of scar tissue once they switch from a quiet, vitamin‑storing state into an active, muscle‑like form. The team showed that TGFBI pushes stellate cells toward this harmful identity. In dishes, stellate cells from Tgfbi‑deficient mice were less active and divided more slowly. Adding purified TGFBI to mouse or human stellate cells had the opposite effect, boosting their growth, movement, and production of a classic activation marker. Digging deeper, the researchers found that TGFBI binds to a surface receptor called integrin αvβ3 on stellate cells, triggering an internal signaling chain (involving FAK and STAT3 proteins) that ramps up another receptor, PDGFRβ. This receptor makes stellate cells especially responsive to growth cues, and blocking PDGFRβ largely shut down TGFBI’s ability to activate them. 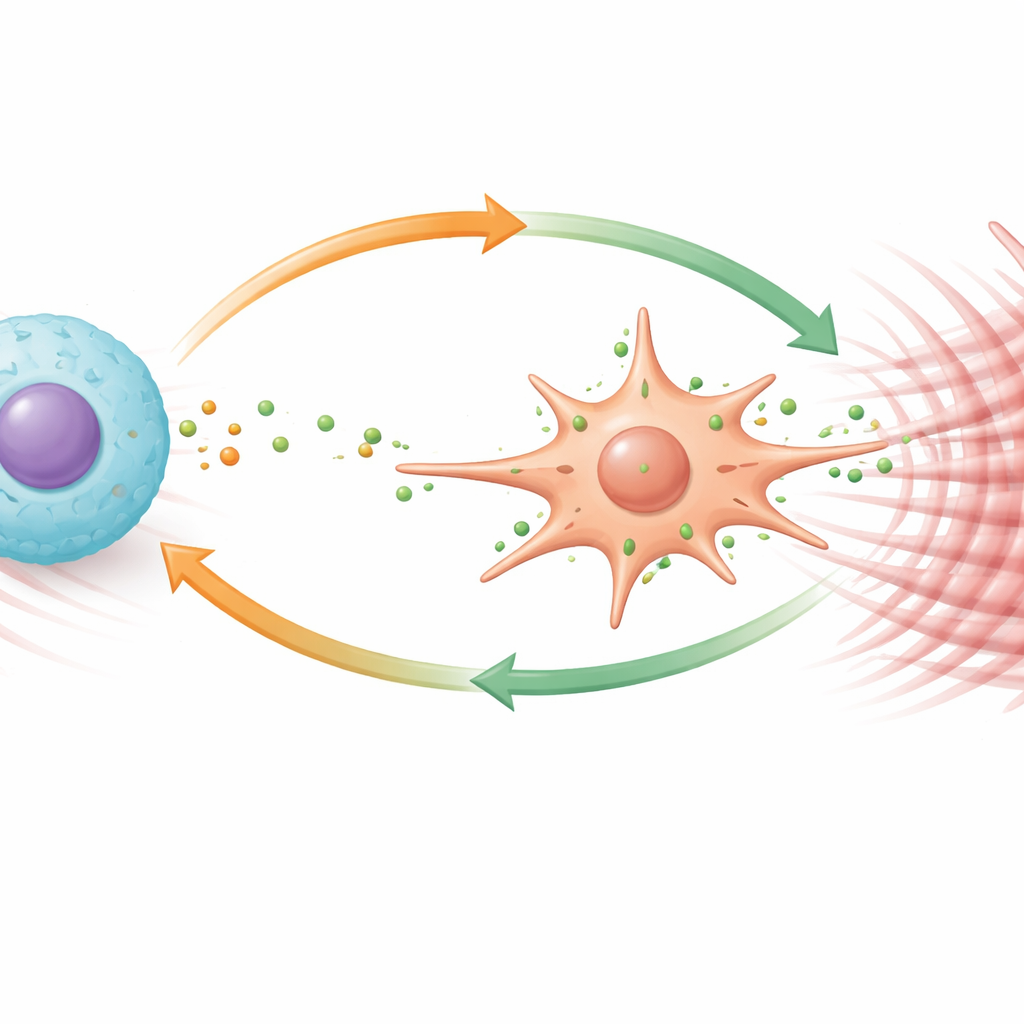
How immune cells and TGFBI reinforce the damage
Macrophages, the liver’s cleanup and defense cells, also proved highly sensitive to TGFBI. When exposed to the protein, these cells proliferated, migrated more readily, and shifted into a specialized subset previously linked to fibrosis. TGFBI made macrophages produce more of a potent signaling molecule called PDGF‑B, which in turn stimulates stellate cells via PDGFRβ. Importantly, PDGF‑B also fed back on macrophages themselves, driving them to make even more TGFBI through another signaling route. This creates a positive feedback loop: macrophages secrete TGFBI, TGFBI makes them release more PDGF‑B and become more profibrotic, and both factors together strongly activate stellate cells. In mouse models, drugs that block PDGFRβ signaling reduced liver inflammation and scarring, underscoring the therapeutic potential of targeting this loop.
What this means for future treatments
In plain terms, the study shows that TGFBI helps set up and maintain a harmful neighborhood inside the injured liver. It pushes support cells to turn into scar‑producing factories and reprograms immune cells into a scarring‑friendly state, all while locking these cells into a self‑amplifying cycle. Interrupting this cycle—by blocking TGFBI itself, its integrin connection, or the PDGF‑B/PDGFRβ pathway—could slow or even reverse fibrosis. While these findings are currently based on animal models and cell experiments, they identify a concrete molecular circuit that drug developers can now target in the quest for better antifibrotic therapies.
Citation: Wu, H., Yan, X., Kuang, L. et al. TGFBI promotes liver fibrosis through remodeling the profibrotic microenvironment by a positive feedback regulatory loop. Commun Biol 9, 355 (2026). https://doi.org/10.1038/s42003-026-09601-2
Keywords: liver fibrosis, TGFBI, hepatic stellate cells, macrophages, PDGFRβ