Clear Sky Science · en
Allergen-specific human IgE isolated through an allergen-agnostic pipeline—understanding immune response and allergen recognition
Why this matters for allergy sufferers
Seasonal sneezing and itchy eyes may feel simple, but the molecules that cause these reactions are anything but. This study reveals a new way to capture and study the exact human antibodies that drive grass pollen allergy, straight from allergic patients. By mapping these antibodies in unprecedented detail, the work opens doors to sharper diagnostics, smarter allergy shots, and future drugs that could block symptoms at their molecular source. 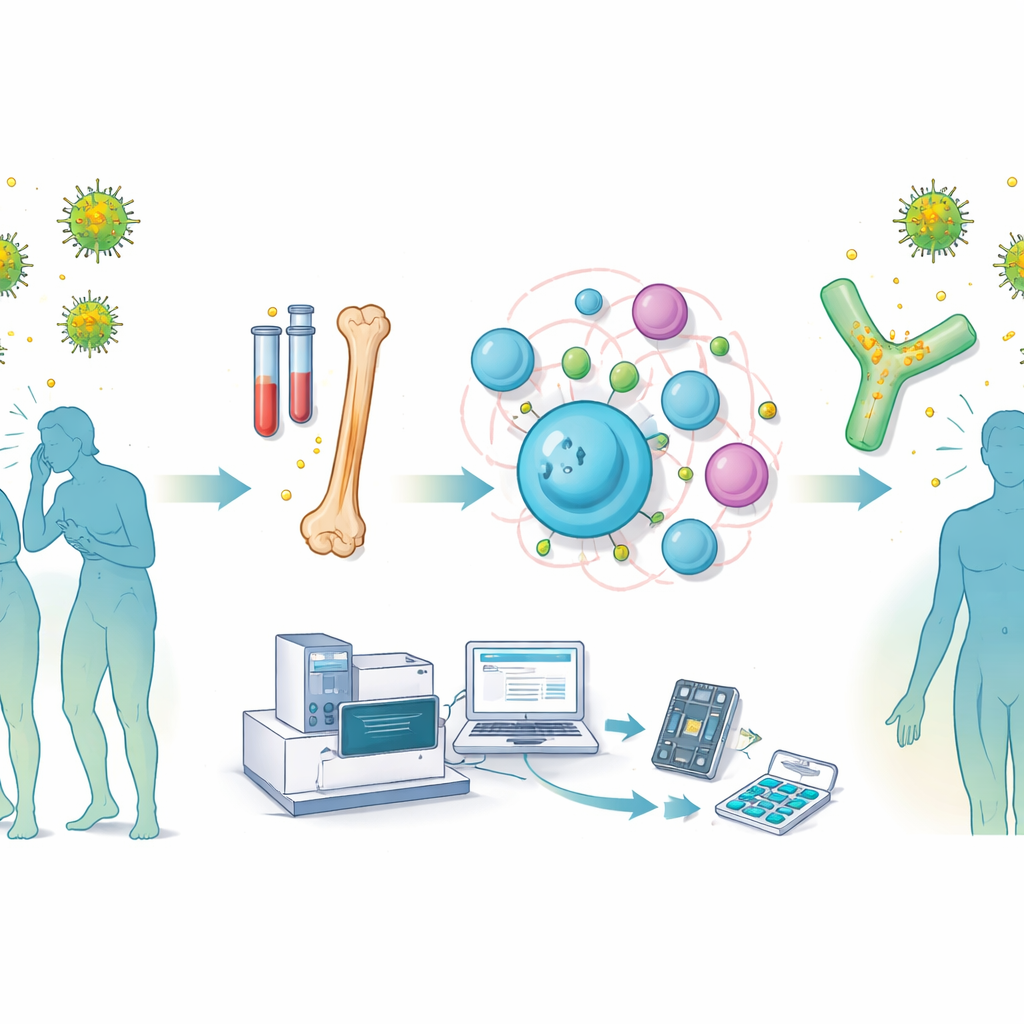
A new window into allergy molecules
Allergic reactions to pollen are powered by a special class of antibodies called IgE, which sit on immune cells and trigger inflammation when they meet an allergen. Yet IgE-producing cells are rare, and scientists have had surprisingly few fully human IgE antibodies to study. The researchers built a "pipeline" that overcomes this problem. They collected blood and bone marrow from six people with grass pollen–induced hay fever, then used single-cell sequencing to read, one cell at a time, the paired heavy and light chain genes that make up each antibody. At the same time, they used deep sequencing of all antibody genes in each person to see which families of antibodies included IgE versions.
Fishing out allergen-specific antibodies
Instead of starting with a particular allergen in mind, the team took an allergen-agnostic approach. They first looked for antibody families that included IgE members in the bulk sequencing data, then matched those families to complete heavy–light chain pairs from the single-cell data. Using recombinant DNA methods, they rebuilt these antibodies in the lab, mostly as the more stable IgG form and, for some, also as IgE. Then came the detective work: a suite of binding tests with purified allergens and complex pollen extracts, along with immunoprecipitation followed by mass spectrometry, to see which pollen proteins each antibody could pull out and recognize. 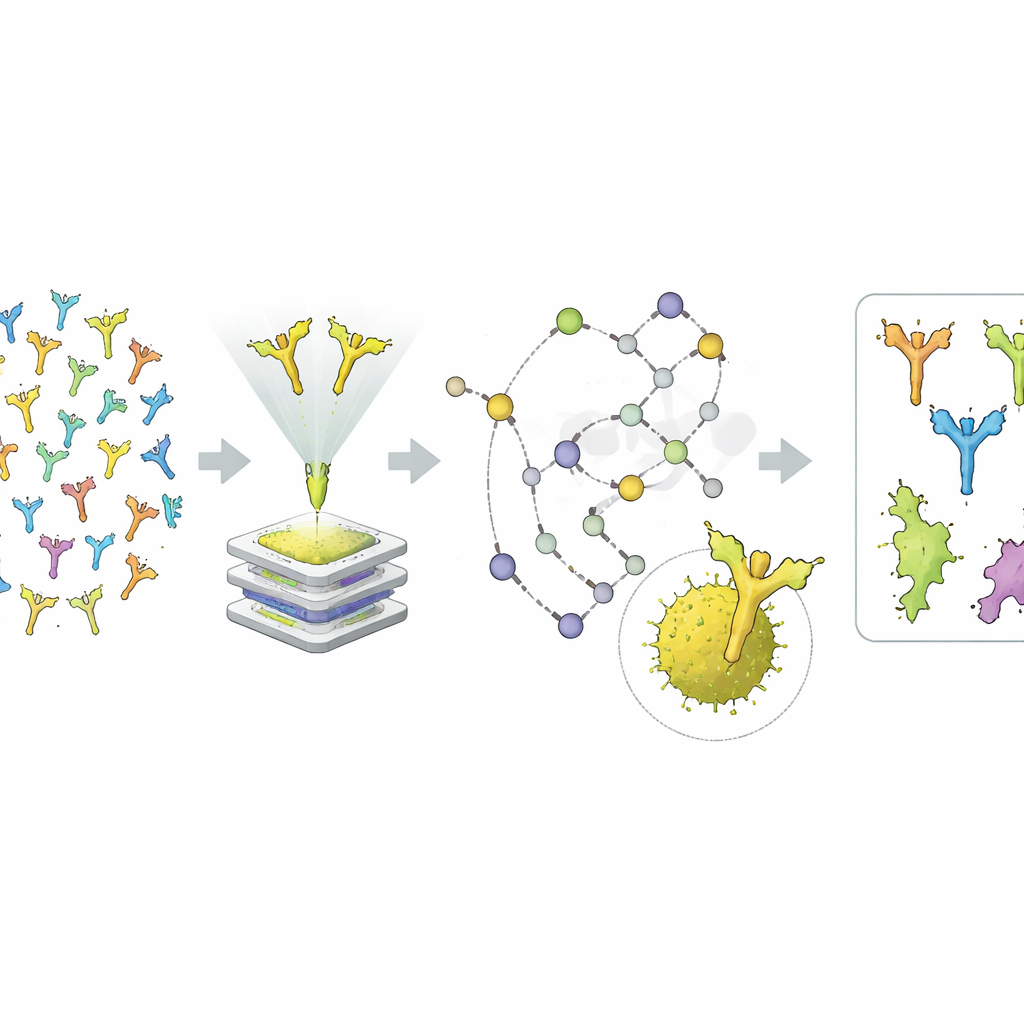
Four key grass pollen targets uncovered
From many candidates, the pipeline delivered four fully human antibodies that clearly recognized distinct grass pollen components. One antibody locked onto group 5 allergens from timothy grass, another onto group 11, a third onto group 3, and a fourth onto group 4. All four bound their targets with remarkably high affinity, in the sub-nanomolar range, meaning they cling tightly to their allergens and dissociate only very slowly. The group 3 antibody proved especially informative: it bound pollen extracts from grasses in one major botanical branch (the BOP clade) but not from another (the PACMAD clade), revealing that this allergen is unevenly distributed across grass species. The group 4 antibody showed that some clinically important allergen components may be underrepresented in standard extract-based diagnostic tests.
How allergy antibodies evolve in the body
Because each antibody family contained multiple sequence variants, the team could reconstruct “family trees” that traced how these antibodies changed over time. For the group 5–specific antibody, they found both IgG1 and IgE versions from the same lineage. Strikingly, the IgG1 variant was only lightly mutated yet already showed very high affinity, suggesting that potent allergy antibodies can arise from near-naïve cells with minimal editing. The IgE variant carried more changes but did not gain dramatically higher affinity, hinting that switching to the IgE class can occur after a strong binder has already been established. Other antibody families appeared in both blood and bone marrow, consistent with long-lived cells that help maintain allergic memory over years.
From lab discovery to future treatments
Beyond basic understanding, the authors examined whether these native human antibodies would be suitable as starting points for drugs. A computational “developability” screen found that most had favorable properties, with only minor sequence features that might need tuning. Together, the results show that combining single-cell sequencing, bulk repertoire analysis, and protein-level assays can reliably isolate natural, high-affinity human IgE-related antibodies without preselecting by allergen. For people living with hay fever and related conditions, this means scientists can now more precisely chart which pollen molecules matter, how the immune system learns to recognize them, and how to design diagnostics, vaccines, or antibody-based therapies that calm allergies by targeting the disease at its molecular roots.
Citation: Thörnqvist, L., Franciskovic, E., Godzwon, M. et al. Allergen-specific human IgE isolated through an allergen-agnostic pipeline—understanding immune response and allergen recognition. Commun Biol 9, 332 (2026). https://doi.org/10.1038/s42003-026-09600-3
Keywords: grass pollen allergy, IgE antibodies, single-cell sequencing, allergen immunotherapy, monoclonal antibodies