Clear Sky Science · en
DTX1-mediated degradation of TUBB3 in Kupffer cells mitigates hepatocellular carcinoma progression by regulating M1/M2 polarization
Why liver cancer’s immune “neighbors” matter
Most people think of cancer as a disease of rogue cells growing out of control. But tumors live in a bustling neighborhood of immune cells, blood vessels, and supporting tissue that can either fight the cancer or quietly help it grow. This study looks at liver cancer—specifically hepatocellular carcinoma—and uncovers how a protein inside specialized liver immune cells can tilt the balance between an anti-cancer response and a tumor-friendly environment. Understanding this hidden control switch could open new ways to improve current immunotherapies.
A liver cancer driven by its surroundings
The liver is rich in immune cells, including a resident population called Kupffer cells that normally help keep the organ healthy. In liver cancer, many of these cells transform into tumor-associated macrophages that can either attack cancer (an M1-like state) or protect it (an M2-like state). Patients whose tumors are packed with M2-type macrophages usually do worse and often respond poorly to drugs that unblock immune defenses, such as PD-1 inhibitors. The authors began by mining public gene databases and patient samples to look for molecules linked both to liver cancer and to these macrophages, homing in on a structural protein called TUBB3 that was unexpectedly abundant in tumor tissue.
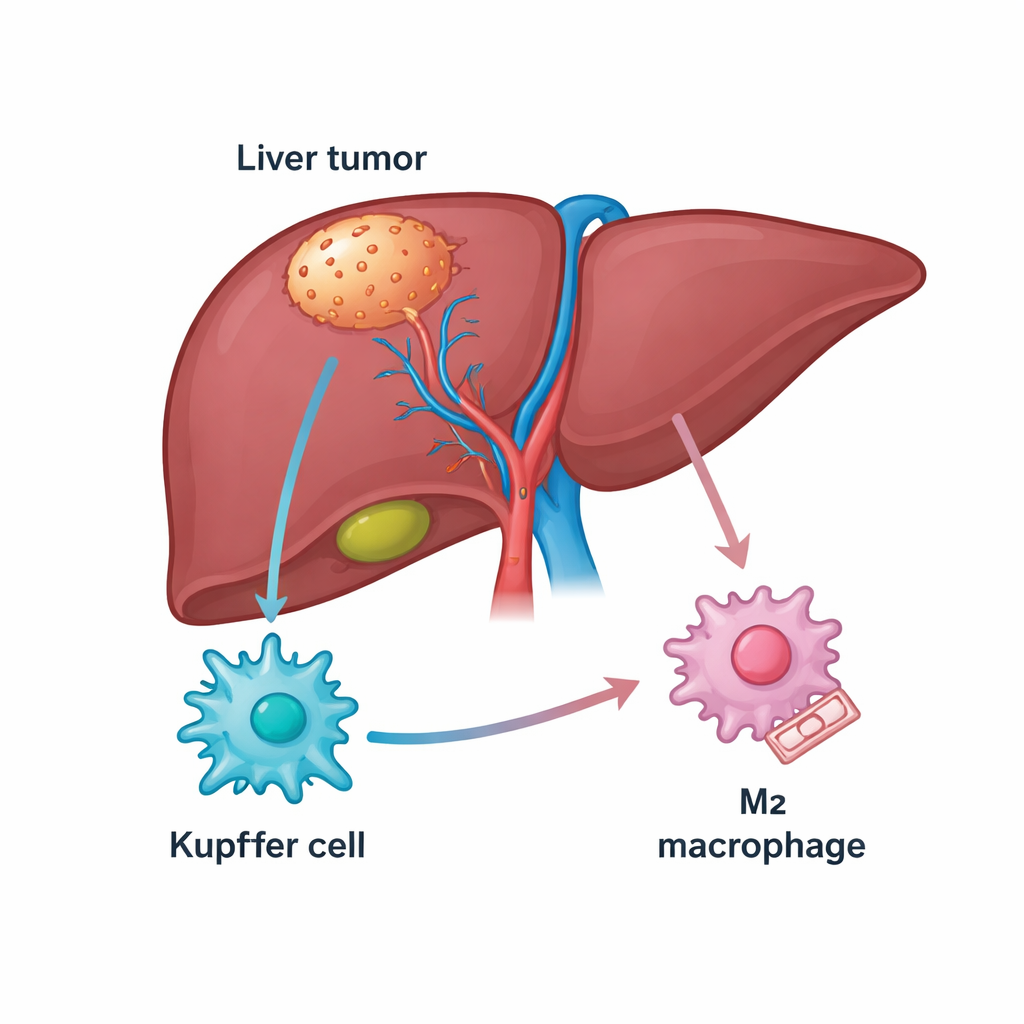
A surprising protein that pushes immune cells to help tumors
TUBB3 is best known as a building block of microscopic “rails” inside cells, but it has also been linked to aggressive behavior and drug resistance in several cancers. Here, the researchers found that TUBB3 levels were much higher in liver tumors than in nearby healthy tissue, and that patients with more TUBB3 tended to have shorter survival. Using staining techniques, they showed that TUBB3 was especially enriched in Kupffer cells inside tumors, and its presence strongly tracked with markers of the tumor-helping M2 state. In other words, Kupffer cells packed with TUBB3 were more likely to behave in ways that dampen immune attack and support cancer growth.
Reprogramming macrophages to restrain liver tumors
To test cause and effect, the team reduced TUBB3 levels in Kupffer cells in laboratory experiments. When TUBB3 was knocked down, these cells produced fewer M2-type signals, more M1-type signals, and secreted fewer molecules that normally suppress immune activity. Liver cancer cells exposed to these reprogrammed macrophages grew more slowly, divided less, and were less able to migrate and invade. In mice, mixing liver cancer cells with TUBB3-deficient Kupffer cells led to smaller tumors, more cancer cell death, and a greater influx of killer CD8 T cells armed with destructive proteins. If macrophages were removed from the animals altogether, the benefit of blocking TUBB3 largely disappeared, underscoring that the effect runs through these immune cells rather than the cancer cells alone.
Uncovering an internal brake on a key signaling pathway
The study also probed how TUBB3 exerts this influence. The authors focused on a well-known growth and survival pathway controlled by the proteins PTEN and AKT, which also shapes whether macrophages adopt an M1 or M2 identity. They found that lowering TUBB3 boosted PTEN, which in turn reduced the activating “phosphate tags” on AKT, nudging macrophages toward the tumor-fighting state. When they artificially re-activated AKT, macrophages reverted to the M2 pattern and cancer cells regained their aggressive behavior, even in the absence of TUBB3. This placed TUBB3 upstream of PTEN and AKT as a kind of internal dial for macrophage behavior.
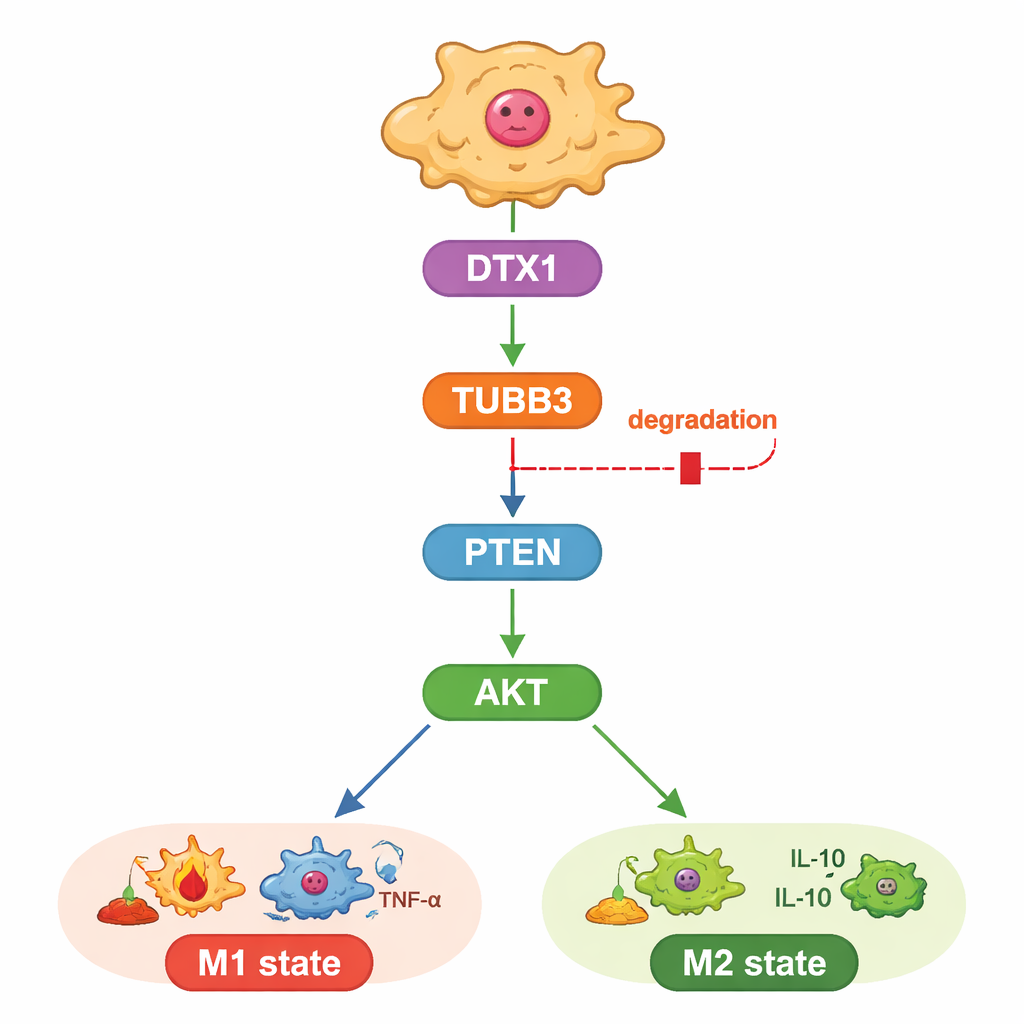
A built-in trash tag that could be turned back on
Finally, the researchers asked why TUBB3 is so abundant in liver tumors. By combining bioinformatic predictions with expression data, they identified an enzyme called DTX1, part of a family that tags proteins for disposal, as a likely regulator. DTX1 levels were lower in liver tumors than in healthy tissue and were linked to better patient outcomes and to a greater presence of M1-type macrophages. In cells, boosting DTX1 accelerated TUBB3 breakdown, dampened AKT activity, and pushed macrophages toward the tumor-fighting state, curbing cancer cell growth. Overriding this by re-introducing extra TUBB3 erased the benefits of DTX1, both in dishes and in mouse tumors. Together, these results draw a clear line from DTX1 through TUBB3 and PTEN/AKT to the immune tone of the tumor.
What this means for future liver cancer treatment
For non-specialists, the key message is that this work identifies a previously hidden control system inside liver-resident immune cells that decides whether they help or hinder cancer. When the trash-tagging enzyme DTX1 is low, TUBB3 accumulates, a growth pathway turns on, and Kupffer cells slide into a tumor-supporting mode that blunts the effect of immunotherapy. Restoring this pathway—by lowering TUBB3, boosting DTX1, or carefully tuning the PTEN/AKT signal—could reawaken local immune defenses and make treatments like PD-1 blockers more effective. While such strategies are still at the experimental stage, they highlight the promise of treating not just the tumor cells, but also the surrounding immune “neighbors” that strongly influence how liver cancer behaves.
Citation: Sun, J., Sun, T., Zhang, Y. et al. DTX1-mediated degradation of TUBB3 in Kupffer cells mitigates hepatocellular carcinoma progression by regulating M1/M2 polarization. Commun Biol 9, 311 (2026). https://doi.org/10.1038/s42003-026-09593-z
Keywords: hepatocellular carcinoma, tumor-associated macrophages, Kupffer cells, immunotherapy, AKT signaling