Clear Sky Science · en
Sparsification and decorrelation of granule cell activity in the dentate gyrus by noradrenaline
Why a burst of arousal can sharpen memories
Moments that jolt us awake—a near-miss in traffic, an unexpected comment, a surprising twist in a film—often stick in memory far better than an ordinary day. This study explores a key reason why: a brain chemical linked to arousal, noradrenaline, quietly reshapes how a critical memory gateway in the hippocampus filters and separates experiences, helping similar events become easier to tell apart later.
The brain’s gatekeeper for similar experiences
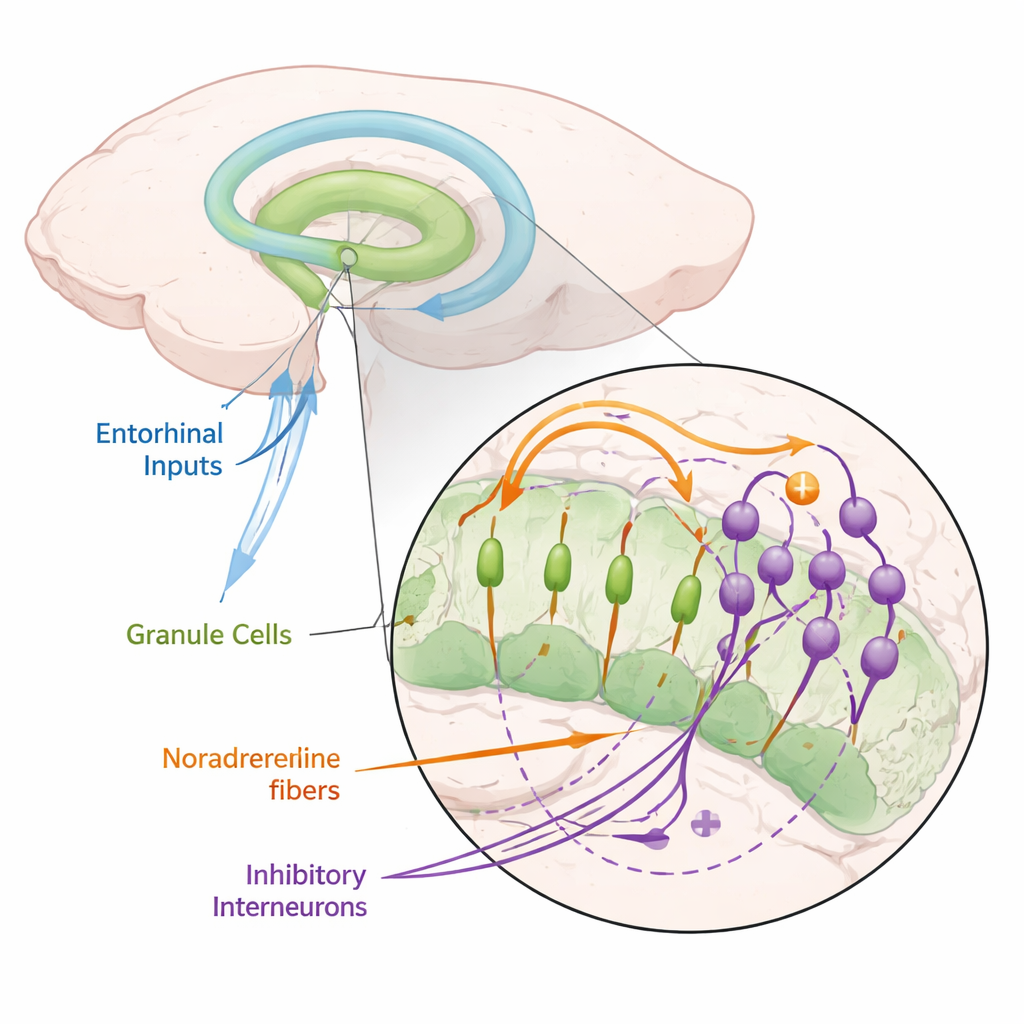
Inside the hippocampus lies the dentate gyrus, a region that acts like a gatekeeper for new memories. It receives rich information from the entorhinal cortex—signals about where we are and what is happening around us—and converts them into activity patterns in granule cells, its main neurons. Theory and experiments suggest these patterns must be “sparse” (only a few cells active at once) and “decorrelated” (different experiences activate different sets of cells) so that memories don’t blend together. Yet, how this transformation happens at the level of specific cells and circuits has remained unclear.
Arousal chemical that quiets key memory cells
The authors focused on noradrenaline, a neuromodulator released by neurons in a small brainstem area called the locus coeruleus, which becomes active during attention, novelty, and stress. Using mice, they expressed light-sensitive proteins in these noradrenergic neurons, allowing them to release noradrenaline on demand with flashes of light. When they stimulated the main input pathway to the dentate gyrus and recorded from granule cells, they found that releasing noradrenaline strongly reduced the cells’ tendency to fire. This suppression appeared both at the level of single neurons and in population signals, and it was reproduced by simply bathing slices in noradrenaline. Blocking noradrenaline receptors removed the effect, showing that it truly depended on this chemical messenger.
Not weaker excitation, but stronger brakes
To understand how noradrenaline silenced granule cells, the team checked the obvious possibilities. It did not significantly change the resting voltage or input resistance of granule cells, meaning their basic excitability stayed roughly the same. Nor did it weaken the excitatory currents these cells receive from the entorhinal cortex. Instead, when they blocked GABAA receptors, which mediate inhibition, noradrenaline could no longer suppress granule cell firing. Detailed current measurements showed that noradrenaline selectively boosted a fast, feedforward form of inhibition: incoming excitatory signals first drove a set of interneurons, which then rapidly inhibited granule cells before those cells could spike. Timing analyses revealed that this noradrenaline-sensitive inhibitory current arrived just after the direct excitation, but before the main granule cell population fired, a hallmark of feedforward brakes.
Specialized inhibitory cells that enforce timing
Which interneurons provided this crucial inhibition? Surprisingly, parvalbumin-expressing cells, long thought to dominate fast feedforward control, were not responsible—noradrenaline actually made them less active. Instead, the key players were cholecystokinin-expressing interneurons (CCK cells). These cells receive direct input from the same cortical fibers that excite granule cells and fire just before granule cells do, indicating a feedforward role. Noradrenaline depolarized CCK cells, making them easier to recruit, and increased how often input signals triggered them, without changing the strength of each individual inhibitory connection. When the researchers pharmacologically blocked output from CCK cells, noradrenaline could no longer suppress granule cell activity. In effect, noradrenaline turns up a circuit of CCK interneurons that imposes a very narrow time window during which incoming excitatory spikes can successfully drive granule cells.
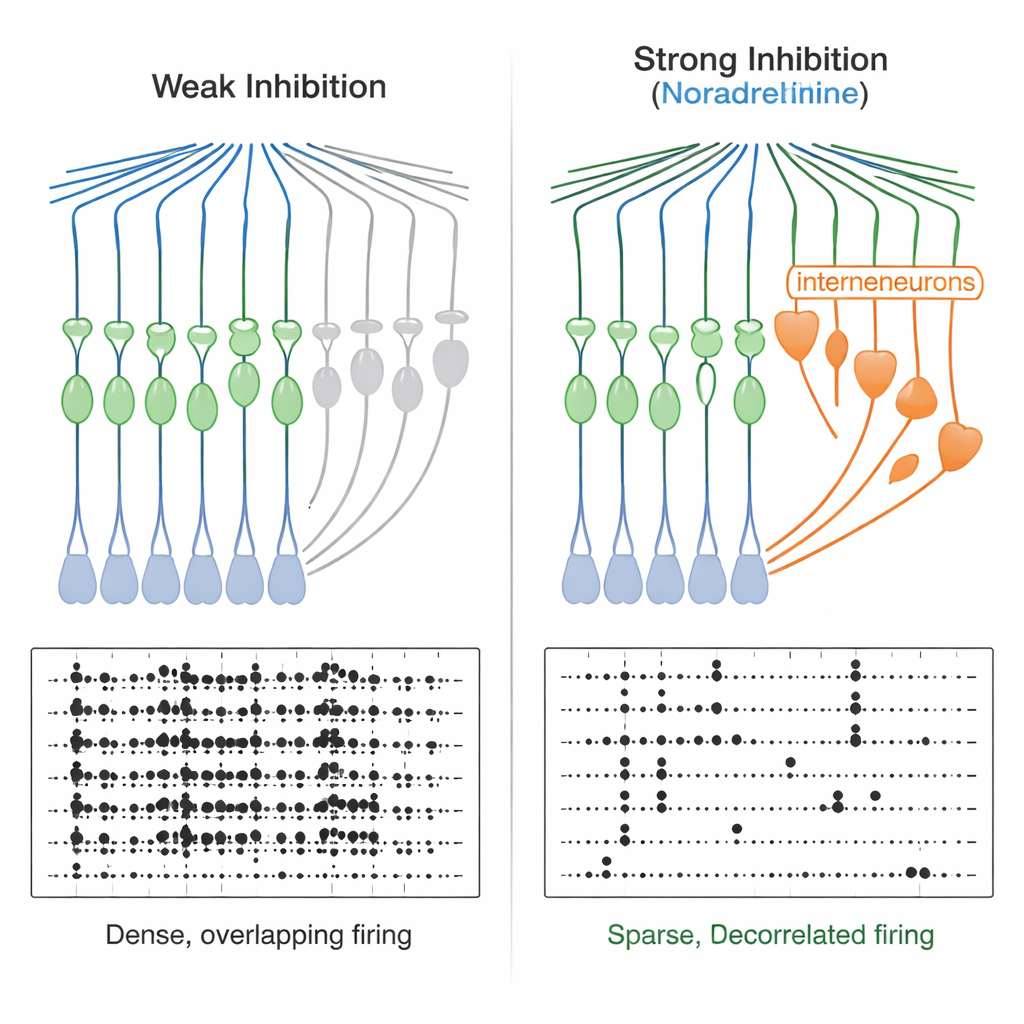
From narrow windows to cleaner memory codes
This sharpened timing has powerful consequences. When the team delivered pairs of brief excitatory inputs, they found that under normal conditions granule cells could integrate inputs spaced tens of milliseconds apart into a spike. With noradrenaline present, the window shrank to only a few milliseconds—granule cells now responded almost exclusively to highly synchronized inputs. Computational network models confirmed that making feedforward inhibition stronger and faster produced sparser output and reduced overlaps between activity patterns, improving “decorrelation.” Experimentally, when the researchers drove two similar, but not identical, input patterns into the dentate gyrus, granule cells responded with more distinct firing patterns in the presence of noradrenaline, both in single-cell recordings and in calcium imaging across many cells. At the same time, overall granule cell activity became sparser.
How arousal may help us separate similar memories
For a lay reader, the takeaway is that noradrenaline, released when we are alert or emotionally engaged, helps a key memory filter in the hippocampus become more selective. By energizing a specific class of inhibitory nerve cells, it narrows the time window during which inputs can trigger granule cells, so only tightly synchronized, meaningful signals get through. This reduces overall firing, makes patterns of activity less overlapping, and helps the brain store similar experiences—like two classrooms or two conversations—as separate memories instead of a blur. The work reveals a concrete circuit mechanism linking momentary arousal to more precise, less confusable memories.
Citation: Glovaci, I., Mihály, A., Vervaeke, K. et al. Sparsification and decorrelation of granule cell activity in the dentate gyrus by noradrenaline. Commun Biol 9, 323 (2026). https://doi.org/10.1038/s42003-026-09592-0
Keywords: noradrenaline, dentate gyrus, inhibitory interneurons, pattern separation, episodic memory