Clear Sky Science · en
Transient proliferation by reversible YAP and mitogen control of the cyclin D1/p27 ratio
How Tissues Know When to Grow
When you cut your skin or damage an organ, nearby cells briefly switch from a resting state to a repair mode, multiplying just enough to patch the wound before stopping again. This paper asks a deceptively simple question: how do cells know when to start and, just as importantly, when to stop dividing so that healing occurs without tipping into cancer-like overgrowth? The researchers uncover a built-in balancing act inside cells that turns a powerful growth signal on only temporarily and then reliably shuts it down.
A Traffic Light for Cell Division
Inside many tissues, a molecular system called Hippo–YAP acts like a traffic controller for cell growth. When tissues are crowded and intact, Hippo keeps a protein called YAP out of the nucleus, and cells remain quiescent. When tissue is injured or stretched, Hippo is relaxed, YAP moves into the nucleus, and cells are nudged to divide. The authors studied thousands of individual epithelial cells grown as flat sheets and exposed them to different combinations of growth factors (mitogens), drugs, and cell densities. They found that growth-factor signals, physical crowding, and YAP activity do not work in isolation; instead, they converge on a single internal decision point that determines whether each cell will reenter the cell cycle.
The Key Ratio Inside the Cell
The central finding is that cells decide to divide based on a ratio of two proteins in the nucleus during the early growth phase known as G1. One protein, cyclin D1, pushes the cell toward division; the other, p27, acts as a brake. Rather than the absolute amount of either protein, it is the cyclin D1/p27 ratio that matters. When this ratio rises above a critical threshold, a gatekeeping protein called Rb becomes phosphorylated and releases the machinery needed for DNA replication, allowing the cell to move forward. Below that threshold, the cell stays in a resting state. The team showed that artificially activating YAP, or blocking upstream brakes in the Hippo pathway, consistently raised the cyclin D1/p27 ratio and drove cells past this threshold, even under conditions that normally keep cells quiet.
How Signals from Outside Are Amplified and Then Faded
YAP does not act alone: it boosts the cell’s responsiveness to growth signals coming from outside. The researchers used RNA sequencing and protein measurements to show that YAP increases the number and activity of receptor proteins on the cell surface, including members of the EGFR family and other receptor tyrosine kinases. These receptors feed into classic growth pathways such as MEK–ERK and, to a lesser extent, mTOR, which raise cyclin D1 and lower p27 to tilt the internal ratio toward division. Importantly, this boost is not a quick on–off switch. Even after YAP activity is blocked, the heightened receptor signaling and elevated cyclin D1/p27 ratio decay only gradually over many hours, giving cells time to complete a limited round of division before the brakes are fully reapplied.
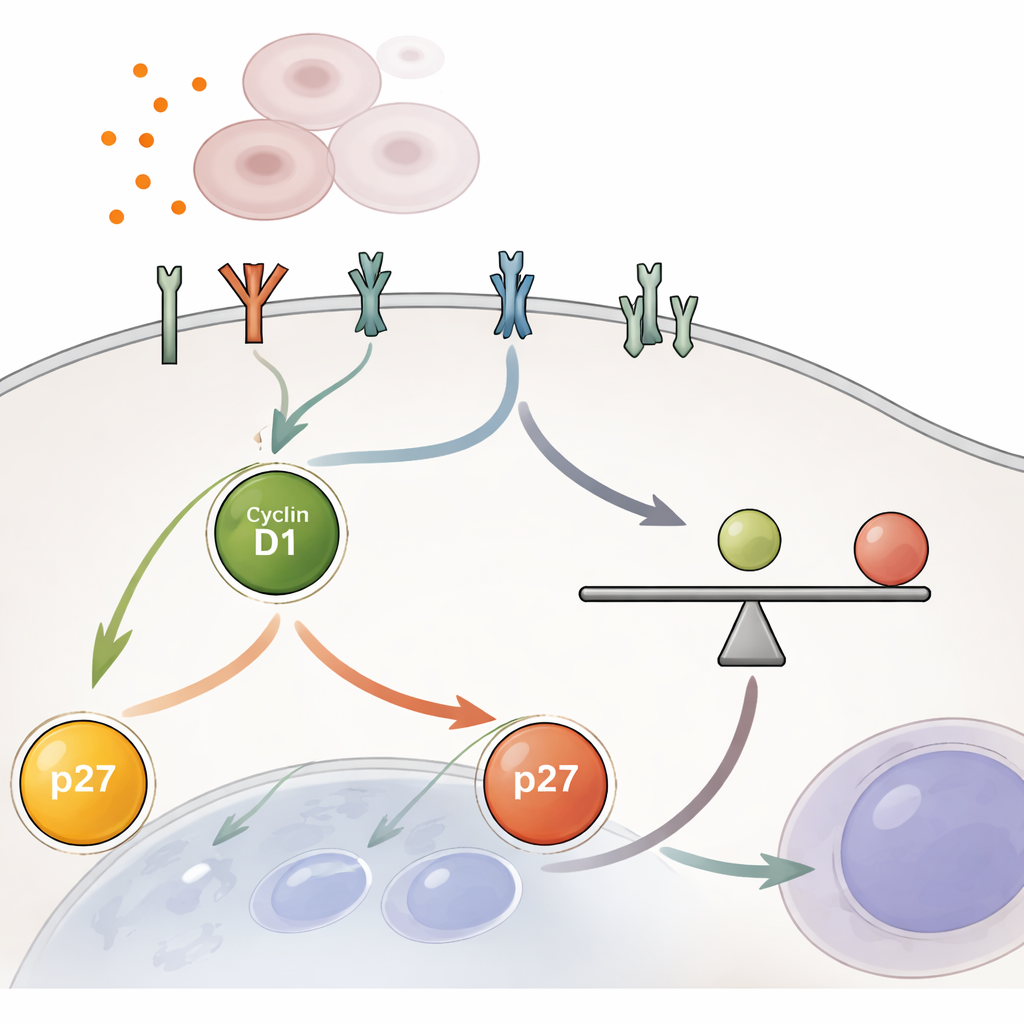
Built-In Safeguards Against Runaway Growth
The study also explores what happens when natural brakes on YAP are weakened, as occurs in some tumors. Knocking out a junctional protein called Merlin, which normally helps activate Hippo, partially unleashed YAP and made cells more sensitive to growth factors. Yet even these cells could still be shut down by very high local cell density, which restored contact inhibition and lowered the cyclin D1/p27 ratio. This shows that the system has multiple layers of control: surface receptors, Hippo–YAP, and the cyclin D1/p27 ratio all contribute to deciding whether cells proceed with division.
Why This Matters for Healing and Cancer
For a general reader, the important message is that tissue repair is driven by a transient, self-limiting growth program. YAP activation and increased receptor signaling raise the cyclin D1/p27 ratio above a threshold so cells can divide and repair damage. As cells divide and the tissue becomes crowded again, contact inhibition gradually switches YAP and receptor signaling off, the ratio falls, and division stops. When this timing mechanism is broken—for example, by persistent YAP activation or loss of upstream brakes—cells can become insensitive to crowding and continue dividing, a step toward cancer. Understanding this ratio-based decision system could help design regenerative therapies that safely boost repair without unleashing uncontrolled growth.
Citation: Ferrick, K.R., Upadhya, S.W., Fan, Y. et al. Transient proliferation by reversible YAP and mitogen control of the cyclin D1/p27 ratio. Commun Biol 9, 340 (2026). https://doi.org/10.1038/s42003-026-09590-2
Keywords: YAP signaling, cell cycle control, tissue regeneration, contact inhibition, cyclin D1 p27 balance