Clear Sky Science · en
Epithelial-mesenchymal plasticity and immunosuppression in canine carcinomas reveals cross-species upregulation of CD109
Cancer Clues Hidden in Dogs
Many family dogs develop mammary (breast) tumors that strikingly resemble human breast cancers. This study uses those naturally occurring canine cancers to answer a pressing question in oncology: why some tumors successfully shut down the immune system’s attack while others remain vulnerable. By watching how dog tumors change their cell shape and behavior, and how immune cells respond, the researchers uncover a shared, cross-species mechanism of cancer immune evasion with direct implications for human patients.
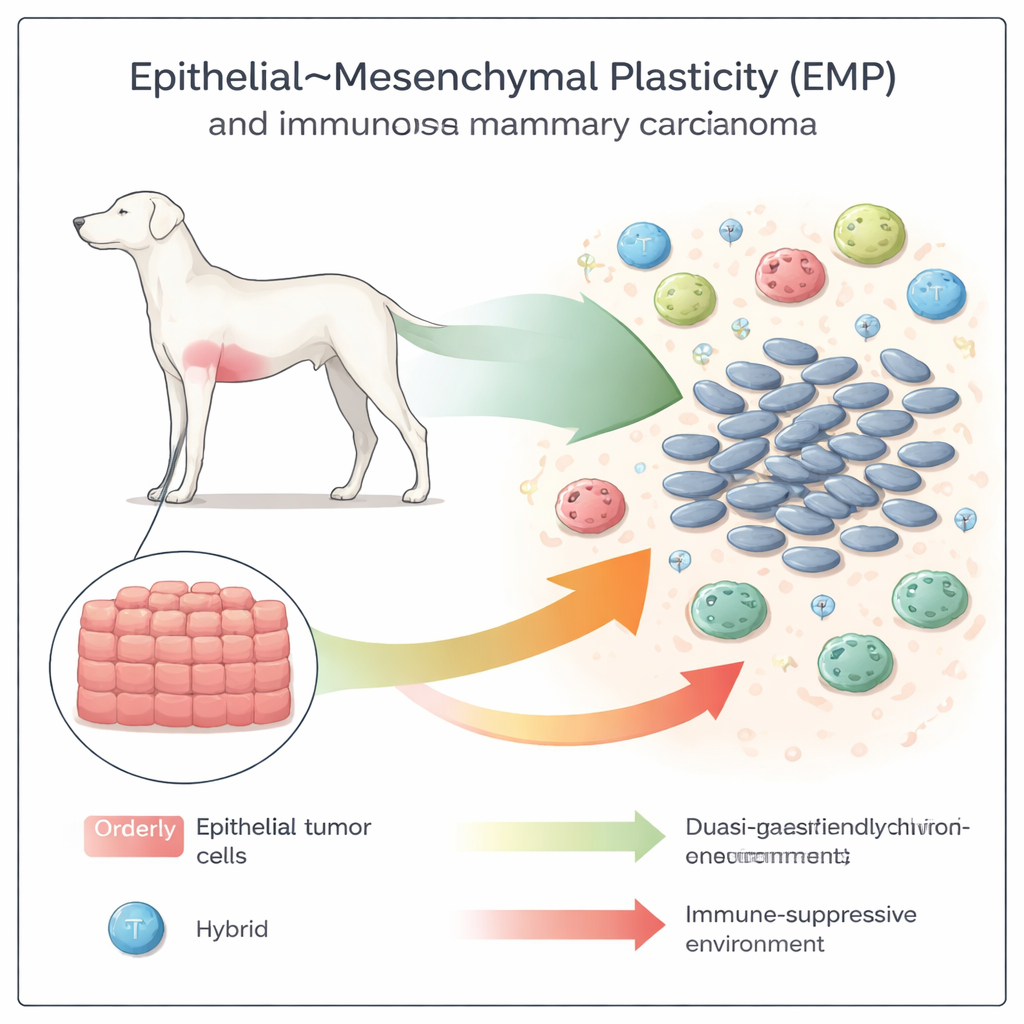
Shape-Shifting Tumor Cells
Cancer cells are not locked into one identity. They can shift from a tidy, brick-like “epithelial” form to a looser, more mobile “mesenchymal” form, and often settle in between. This flexible spectrum, called epithelial–mesenchymal plasticity (EMP), is already known to help cancers invade and spread. In canine mammary carcinomas, the team examined over 50 tumors and found that high-grade, more dangerous cancers showed stronger signs of this shape-shifting program: they lost orderly structures, gained spindle-like cells, and altered key surface proteins that normally keep cells stuck together.
How Tumors Turn Off Immune Attack
The researchers then asked whether this cellular makeover coincides with changes in the tumor’s immune “neighborhood.” In low-grade, more epithelial tumors, T cells—particularly the killer types that can destroy cancer—were relatively abundant. As tumors shifted toward quasi-mesenchymal states and higher histologic grades, overall T-cell numbers fell, while the proportion of regulatory T cells and pro-tumor M2-like macrophages rose. In other words, as cells became more plastic and invasive, the surrounding microenvironment shifted from immune-friendly to immune-suppressive, mirroring patterns previously seen only in mouse models.
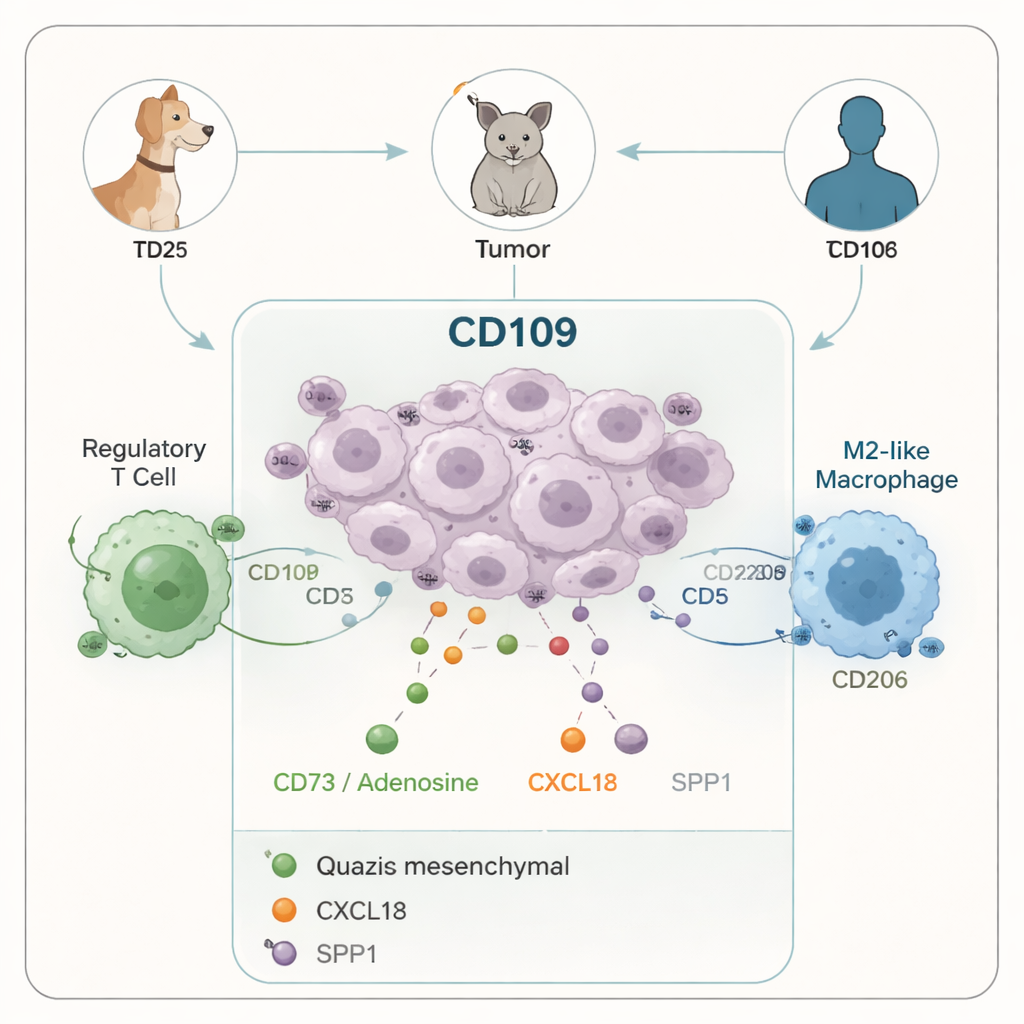
Signals That Invite the Wrong Help
To uncover how these tumors reprogram their surroundings, the team sequenced RNA from a focused set of canine tumors spanning epithelial, mixed, and quasi-mesenchymal states. Heterogeneous and quasi-mesenchymal tumors turned on a suite of secreted factors known to dampen immune responses and attract suppressive cells. These included molecules such as CD73, which generates adenosine that can put immune cells to sleep, and chemokines like CXCL12 and SPP1, which help recruit macrophages and other helpers that end up supporting tumor growth. Notably, even tumors with only a fraction of quasi-mesenchymal cells showed strong immunosuppressive signatures, suggesting a small, aggressive subpopulation can dictate the behavior of the entire tumor.
A New Player: CD109 Across Dogs, Mice, and Humans
Among the many molecules that rose with EMP, one stood out: a surface glycoprotein called CD109. In canine mammary tumors, CD109 levels were higher in more mesenchymal, higher-grade, and basal-like cancers, and tended to be associated with poorer outcomes. Mining existing mouse and human datasets, the authors showed that CD109 is also elevated in quasi-mesenchymal mouse breast tumors and in aggressive, basal-like human breast cancer cells. A key regulator of cell-state change, the transcription factor Snail, binds directly near the CD109 gene, linking it mechanistically to the EMP program. Single-cell analyses confirmed that CD109 expression is concentrated in the most mesenchymal-like cancer cells and co-occurs with other immunosuppressive signals.
Shared Immune Escape Strategies in Many Cancers
To test how general these patterns are, the team reanalyzed RNA data from several other canine cancers of epithelial origin, including oral squamous cell, invasive urothelial, and pulmonary carcinomas. Across these tumor types, EMP markers consistently correlated with higher expression of immunosuppressive paracrine factors—especially CD73, SPP1, and CXCL12—though CD109’s link to EMP appeared strongest and most specific in mammary carcinomas. This suggests a common playbook: as tumors adopt more plastic, invasive states, they simultaneously switch on chemical signals that reshape the immune landscape in their favor.
What This Means for Dogs and People
For a lay reader, the takeaway is that when cancer cells in dogs learn to change shape and identity, they also learn to quiet the immune system and resist modern immunotherapies. Because canine tumors closely mirror human disease, these findings point to shared targets—such as CD73 and the newly highlighted CD109—that might be blocked to reawaken immune attack in both species. In the future, measuring these molecules could help identify high-risk tumors and guide combination treatments that not only attack the cancer cells themselves but also dismantle the protective shield they build around them.
Citation: Bakhle, K., Nelissen, S., Li, L. et al. Epithelial-mesenchymal plasticity and immunosuppression in canine carcinomas reveals cross-species upregulation of CD109. Commun Biol 9, 303 (2026). https://doi.org/10.1038/s42003-026-09587-x
Keywords: canine mammary carcinoma, epithelial mesenchymal plasticity, tumor immune microenvironment, CD109, cancer immunotherapy resistance