Clear Sky Science · en
PiR48444 inhibits MSC osteogenic differentiation and bone regeneration via targeting METTL7A/eIF4E-mediated BMP2 m6A methylation
Why tiny molecules matter for broken bones
Broken bones and age‑related bone loss are common problems, and doctors are keen to use stem cells to help the body rebuild damaged skeleton. Yet these stem cells do not always form new bone as efficiently as we would like, especially in older people or in inflamed tissue. This study uncovers a tiny RNA molecule, called piR48444, that acts like a molecular brake on bone‑forming stem cells. By releasing that brake, the researchers show it may be possible to boost bone repair and protect against bone loss.
A hidden brake inside many kinds of stem cells
The team focused on mesenchymal stem cells, versatile cells that can become bone, cartilage, fat, and more, and that are isolated from many tissues such as bone marrow, baby teeth, and fat. When these cells are nudged to become bone in the lab, thousands of genes and small RNAs change their activity. By sequencing these molecules in stem cells from exfoliated baby teeth, the researchers found that one particular small RNA, piR48444, steadily dropped as the cells shifted toward bone. They then checked other stem cell types and saw the same pattern: cells making more bone had less piR48444, hinting that this molecule might normally restrain their bone‑forming ability.
From lab dishes to living bones
To test this idea, the scientists altered piR48444 levels in several stem cell types. When they reduced piR48444, the cells deposited more calcium, switched on genes linked to bone, and looked more like immature bone tissue under the microscope. When they forced cells to make extra piR48444, the opposite happened: bone‑related activity fell. The team then moved into animal models. Stem cells with piR48444 switched off were used to fill small skull defects in mice. These modified cells produced far more new bone than control cells, as shown by 3D X‑ray scans and tissue staining, suggesting that blocking piR48444 can materially improve bone repair in living organisms.
Protecting fragile bones in disease and aging
Bone loss is driven not only by injuries but also by inflammation and aging. In bone marrow stem cells exposed to bacterial toxins, and in stem cells from older animals, piR48444 levels were unusually high while bone‑building markers were low. The researchers created an "antagomir"—a short molecule designed to neutralize piR48444 in the bloodstream—and injected it into mice with inflammation‑induced bone loss or naturally aged mice. In both models, treated animals retained much more of their spongy inner bone, with denser and more numerous tiny bone struts, while the hard outer shell of bone remained unchanged. This indicates that blocking piR48444 can selectively shield the fragile, metabolically active part of the skeleton that is most vulnerable during aging and chronic inflammation.
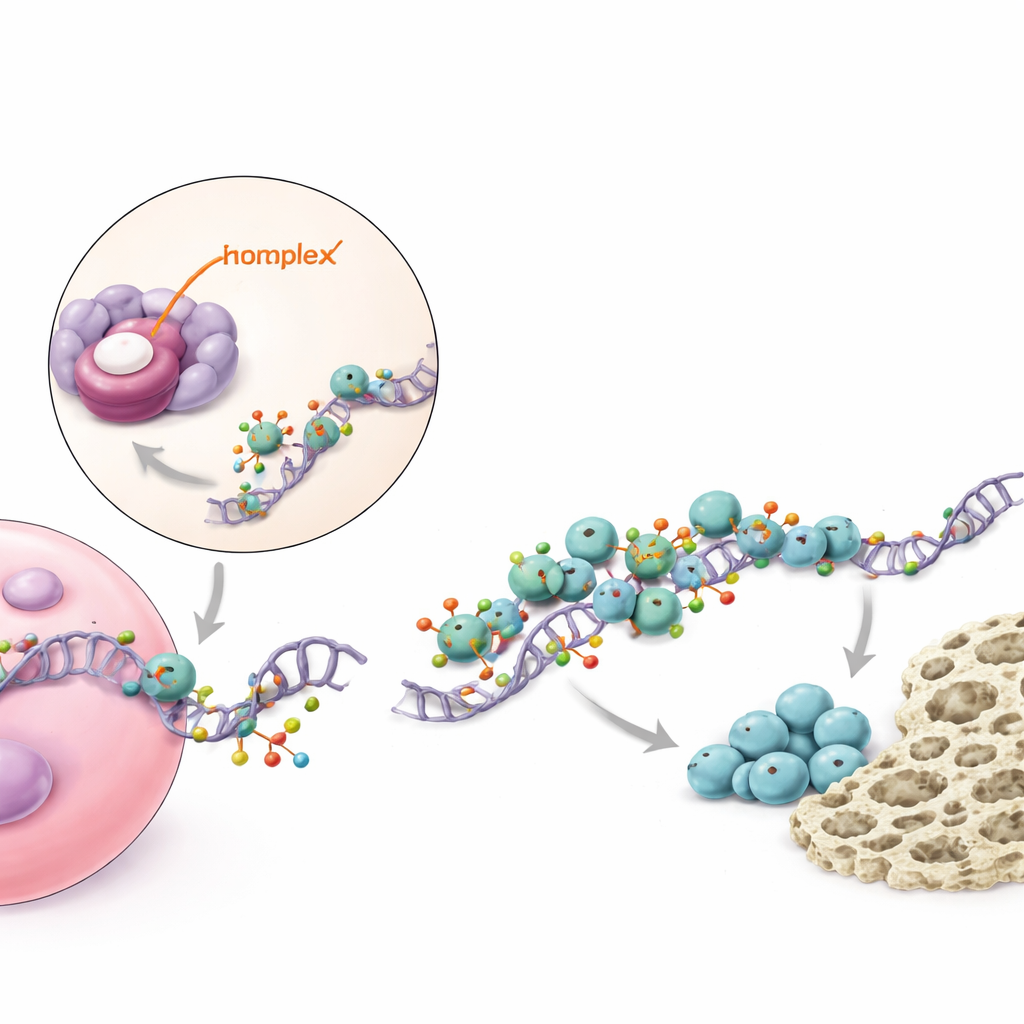
A molecular relay that boosts a key bone signal
Digging deeper, the study mapped how piR48444 exerts its effects inside cells. The small RNA binds to and suppresses a protein called METTL7A, which acts as an enzyme that modifies messenger molecules carrying the blueprint for BMP2, a well‑known growth factor that strongly encourages bone formation. METTL7A adds small chemical marks to BMP2 messages, making them more stable and easier for the cell’s protein‑making machinery to read. METTL7A also works together with another protein, eIF4E, that helps initiate protein production. When piR48444 is abundant, METTL7A is held down, fewer BMP2 messages are marked and translated, and less BMP2 protein is made, so stem cells are less likely to become bone. When piR48444 is blocked, METTL7A and BMP2 rise, pushing cells toward bone formation.
What this means for future bone repair
Put simply, the authors reveal a chain of control in which a tiny RNA (piR48444) dampens a helper enzyme (METTL7A), which in turn limits a powerful bone‑building signal (BMP2). By cutting this chain at the level of piR48444, they can make various stem cells better at forming bone and can lessen bone loss in mice. While safety, off‑target effects, and long‑term outcomes still need careful testing, the work points to piR48444 as both a potential blood marker of osteoporosis risk and a promising target for drugs meant to enhance stem cell–based bone regeneration in the clinic.
Citation: Zheng, Z., Li, X., Qin, W. et al. PiR48444 inhibits MSC osteogenic differentiation and bone regeneration via targeting METTL7A/eIF4E-mediated BMP2 m6A methylation. Commun Biol 9, 337 (2026). https://doi.org/10.1038/s42003-026-09583-1
Keywords: bone regeneration, mesenchymal stem cells, osteoporosis, non-coding RNA, BMP2 signaling